Primary hepatic schwannoma: imaging and histological findings
Hong
Kong Med J 2020 Oct;26(5):449.e1–4
Hong Kong Academy of Medicine. CC BY-NC-ND 4.0
PICTORIAL MEDICINE
Primary hepatic schwannoma: imaging and histological findings
HL Tsui, MB, ChB, FRCR1; SM Yu, MB, ChB, FHKAM (Radiology)1; CH Lau, MB, ChB2; Sherman SM Lam, MB, BS, FHKAM (Surgery)3; PY Chu, MB, ChB, FHKAM (Radiology)1; YH Hui, MB, BS, FHKAM (Radiology)1; KL Lo, MB, ChB, FHKAM (Radiology)1
1 Department of Radiology and Organ Imaging, United Christian Hospital, Hong Kong
2 Department of Pathology, United Christian Hospital, Hong Kong
3 Department of Surgery, United Christian Hospital, Hong Kong
Corresponding author: Dr HL Tsui (karen.tsuihl@gmail.com)
Schwannoma is a rare tumour in the liver. It is likely to arise from the hepatobiliary nerves among the hepatic plexus in the liver hilum as well as interlobular connective tissues and hepatic arteries. To the best of our knowledge, no prior publications have reported cases in Hong Kong.
We report the case of a 64-year-old man with a history of nasopharyngeal carcinoma and colon carcinoma and a new liver lesion detected on follow-up imaging for surveillance in 2018 following detection of a slightly elevated serum carcinoembryonic antigen level (4.7 μg/L). Alpha fetoprotein level was within the normal range
(3.9 μg/L). Liver function tests were normal and he was asymptomatic with no history of neurofibromatosis.
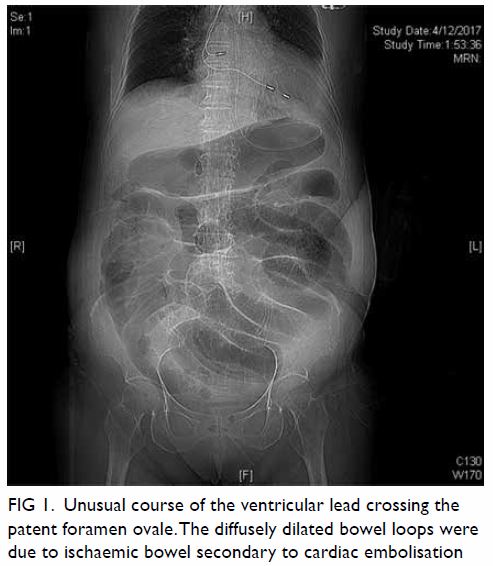
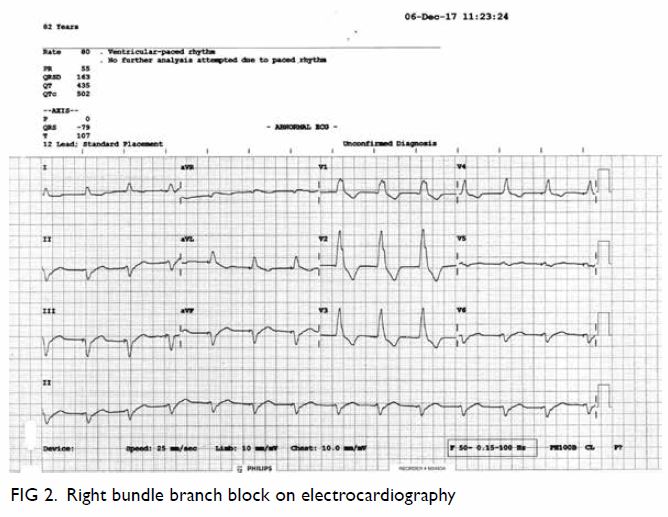
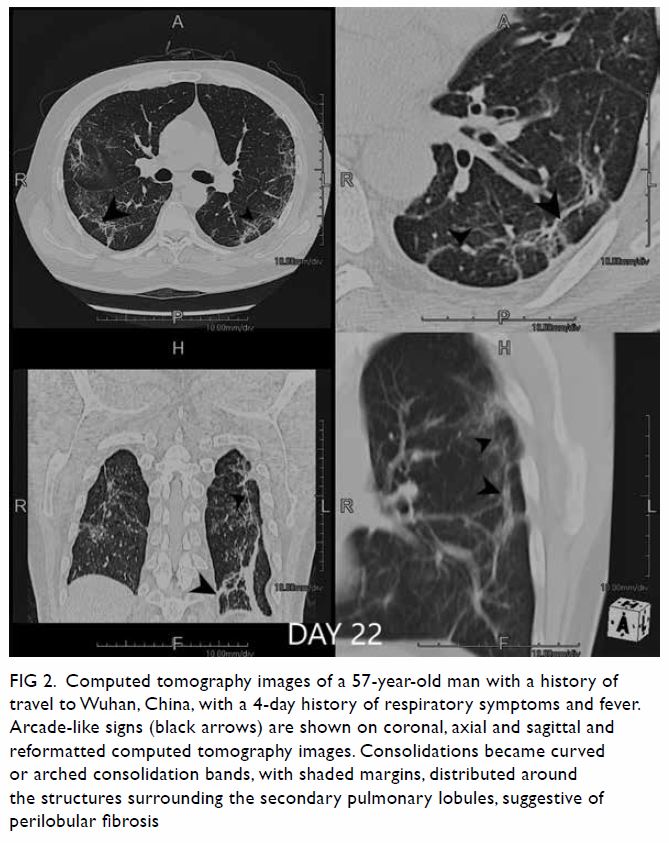
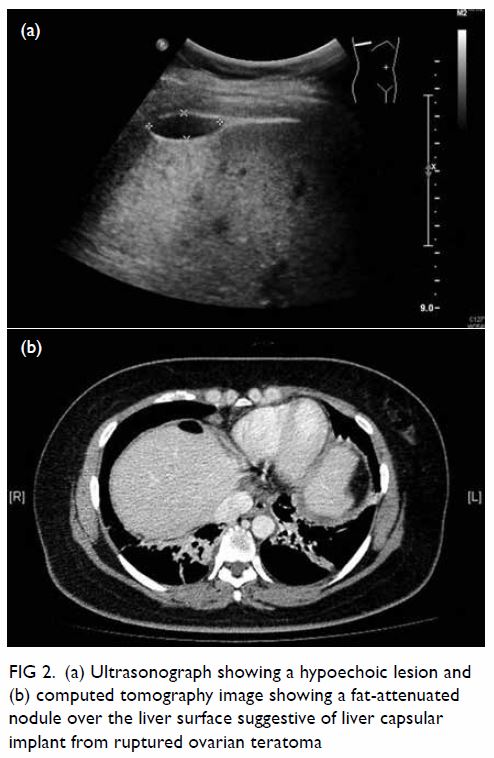
Triphasic contrast computed tomography (CT) of the liver revealed a 5.2-cm ovoid hypodense lesion with heterogeneous enhancement in the caudate lobe of the liver (Fig 1). No washout of contrast was evident in the portal venous or delayed phases. Fluorodeoxyglucose-18 positron emission tomography–CT showed a moderately
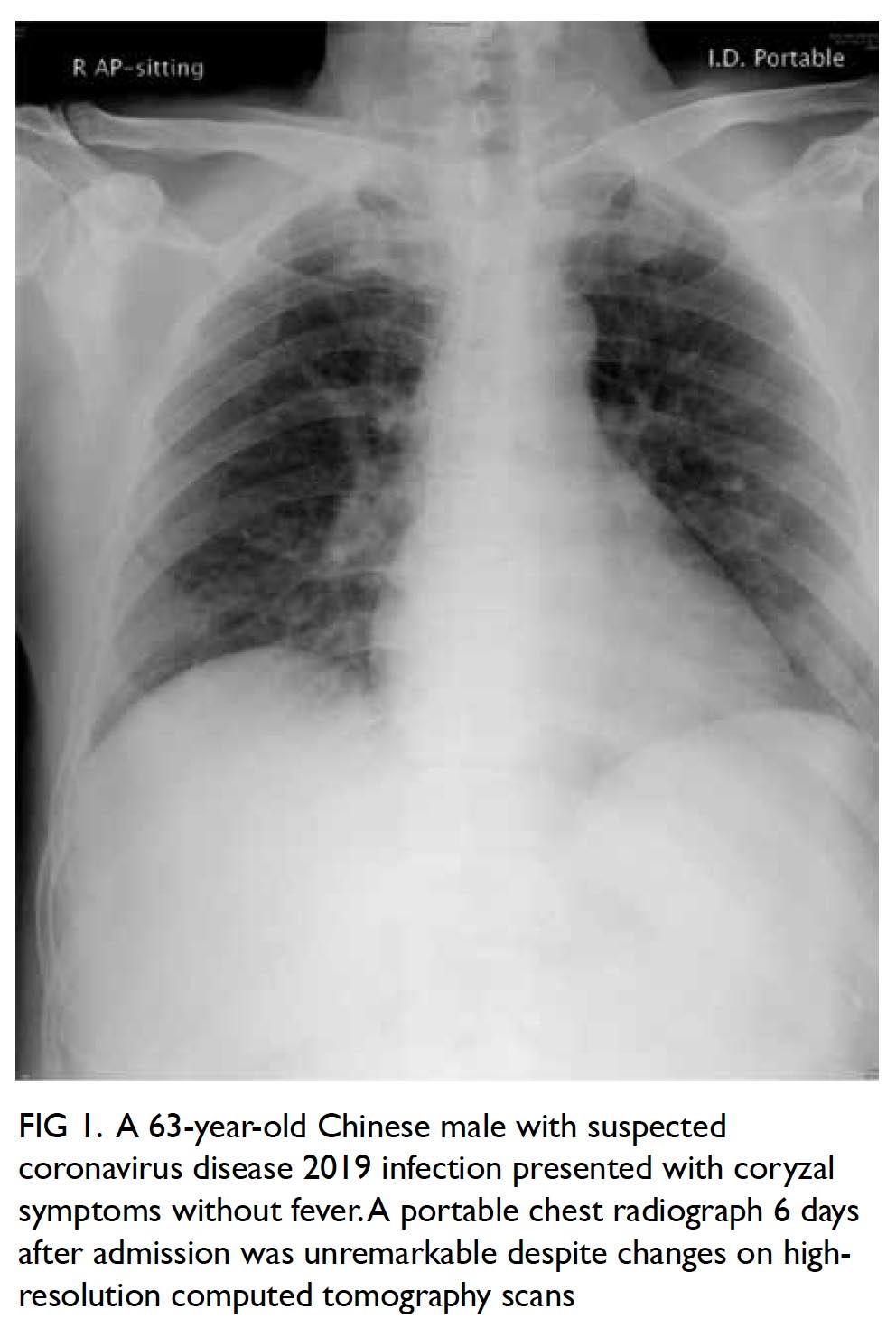
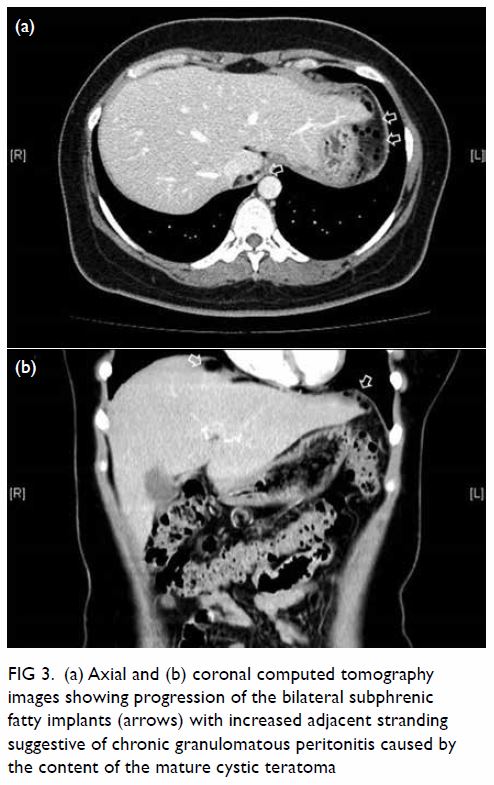
hypermetabolic lesion at the caudate lobe with a
maximum standardised update value of 5.8 (Fig 2).
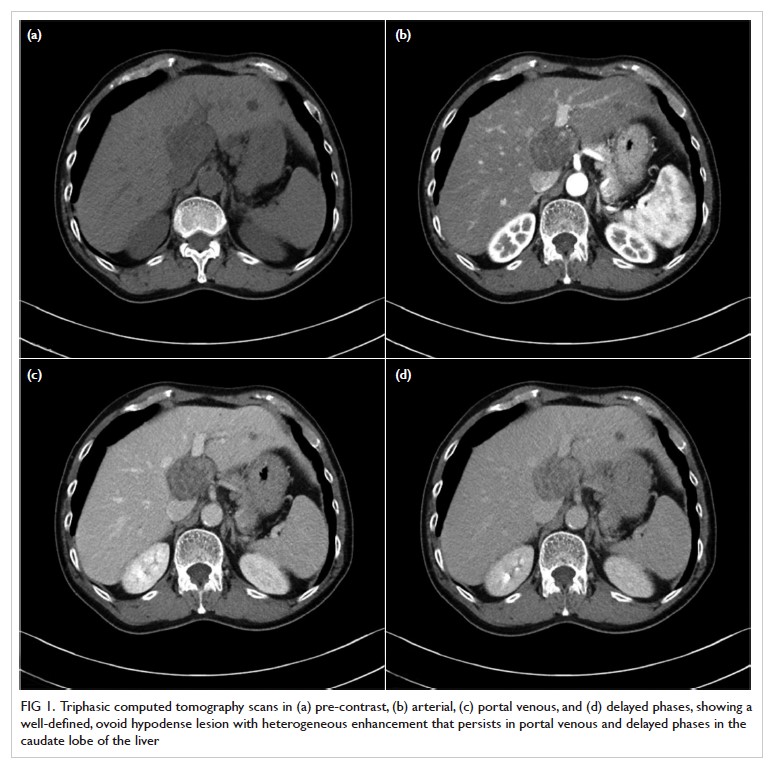
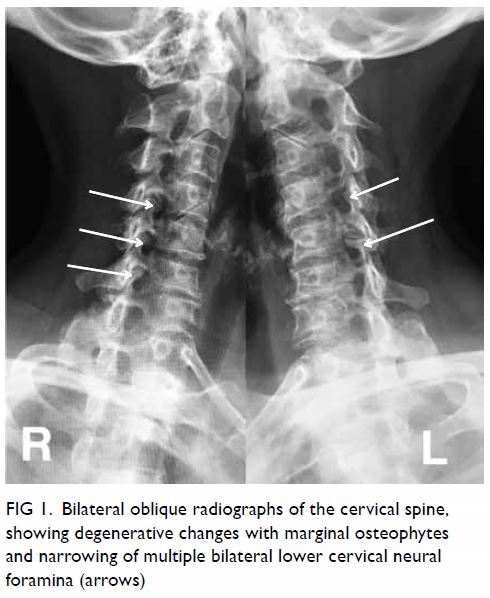
Figure 1. Triphasic computed tomography scans in (a) pre-contrast, (b) arterial, (c) portal venous, and (d) delayed phases, showing a well-defined, ovoid hypodense lesion with heterogeneous enhancement that persists in portal venous and delayed phases in the caudate lobe of the liver
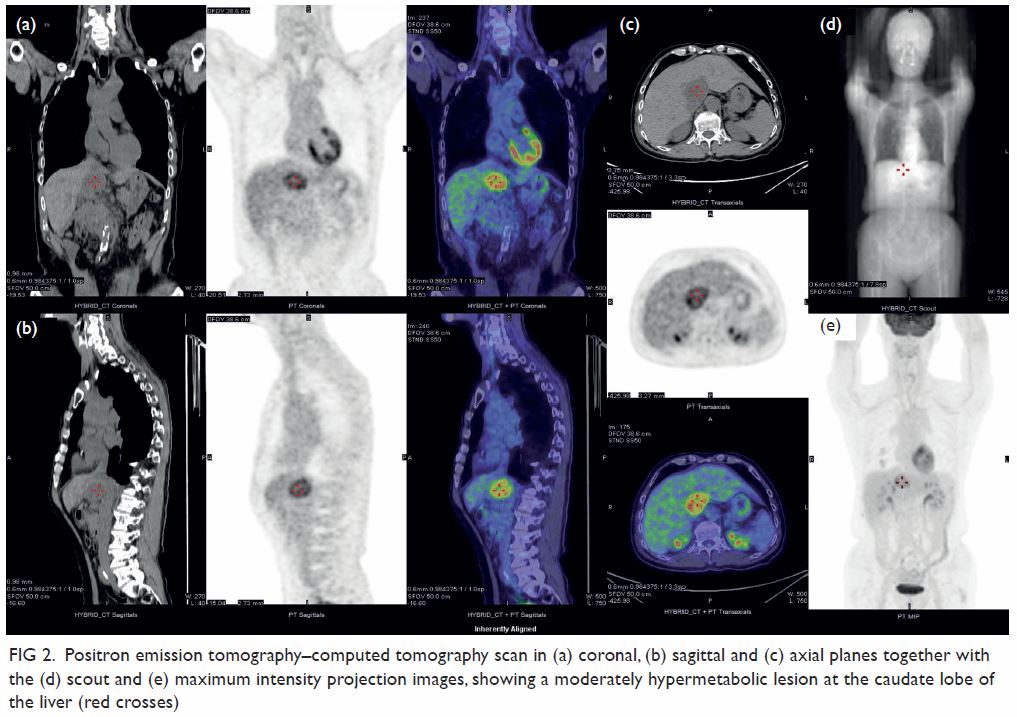
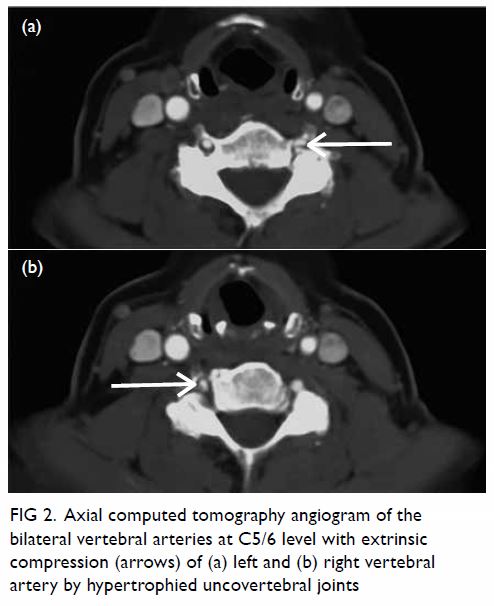
Figure 2. Positron emission tomography–computed tomography scan in (a) coronal, (b) sagittal and (c) axial planes together with the (d) scout and (e) maximum intensity projection images, showing a moderately hypermetabolic lesion at the caudate lobe of the liver (red crosses)
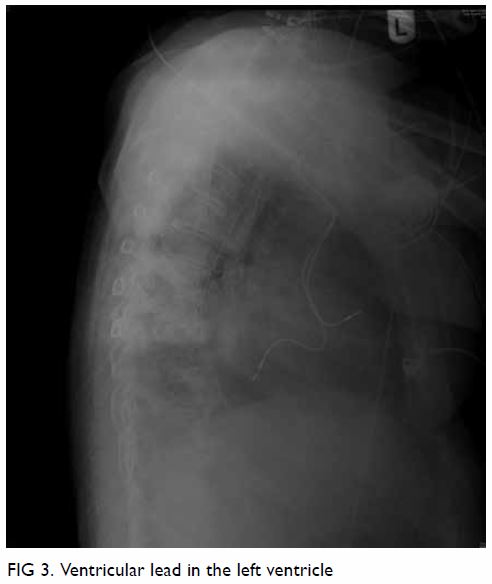
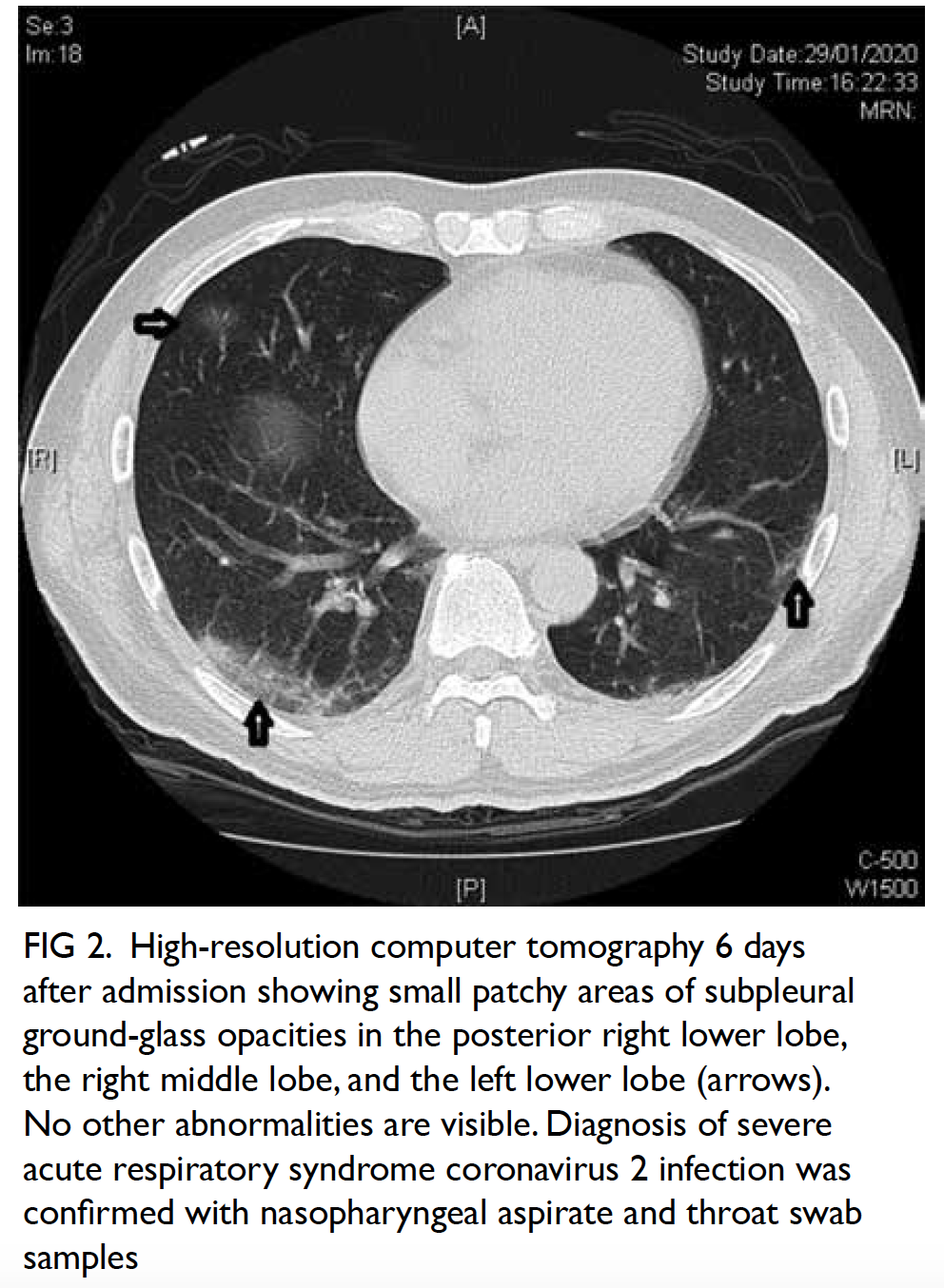
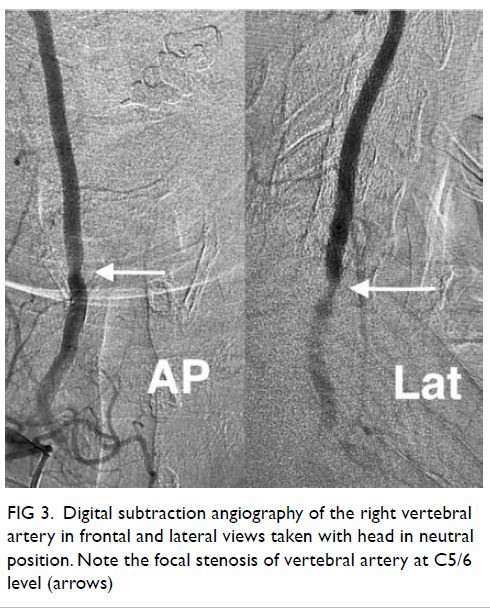
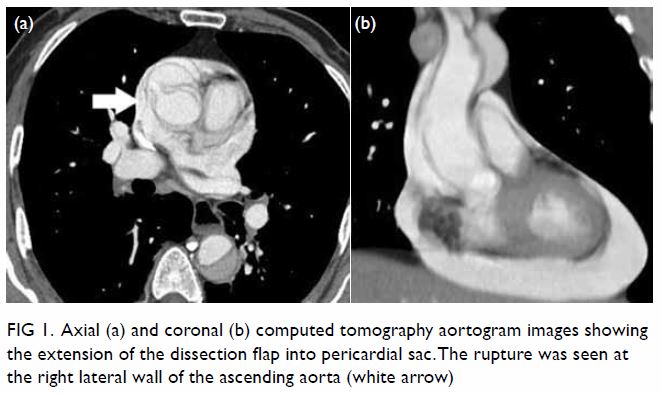
Surgical resection of the lesion was performed
(Fig 3). Pathology showed a schwannoma and
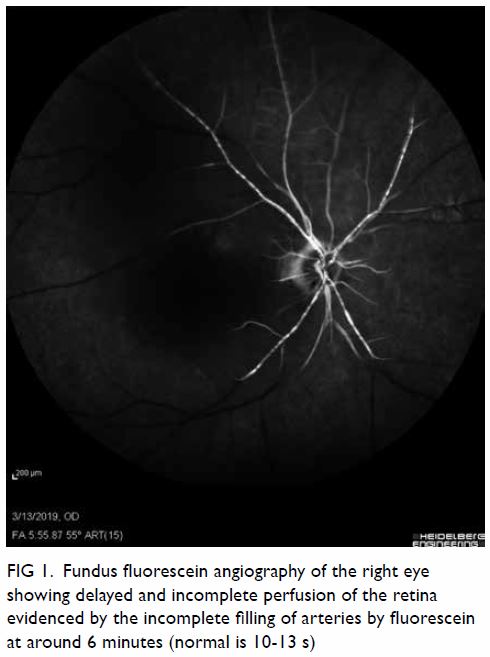
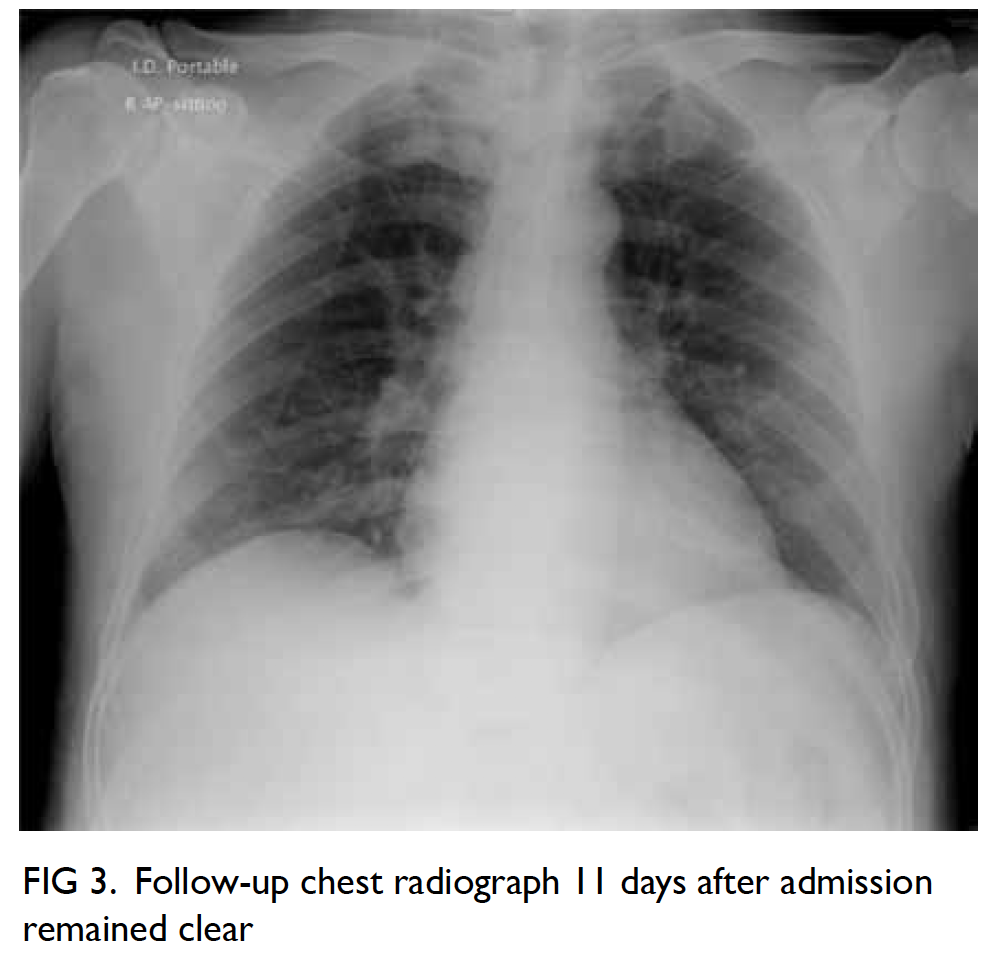
degenerative changes. Histological examination
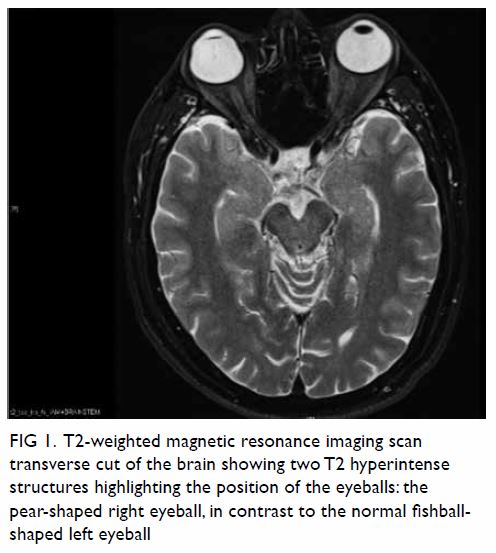
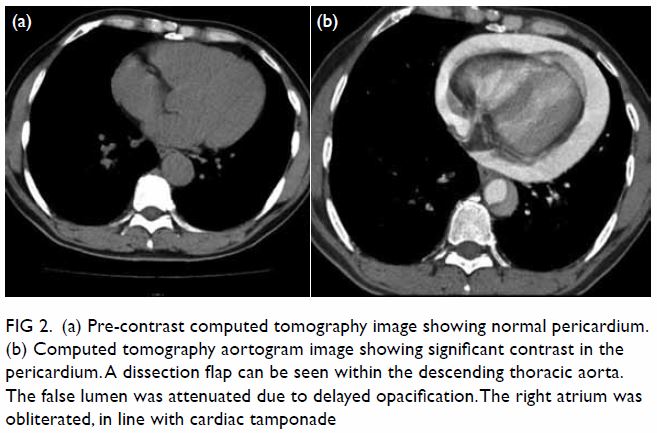
revealed an encapsulated tumour consisting of
highly ordered Antoni type A and B areas (Fig 4).
Immunohistochemical analysis showed the tumour
cells to be diffusely positive for S100, consistent
with neural differentiation (Fig 4). HerPar1, a
mitochondrial antigen of hepatocytes, was negative.
The CD34, a cell surface glycoprotein that is positive
in gastrointestinal stromal tumour, was also negative.
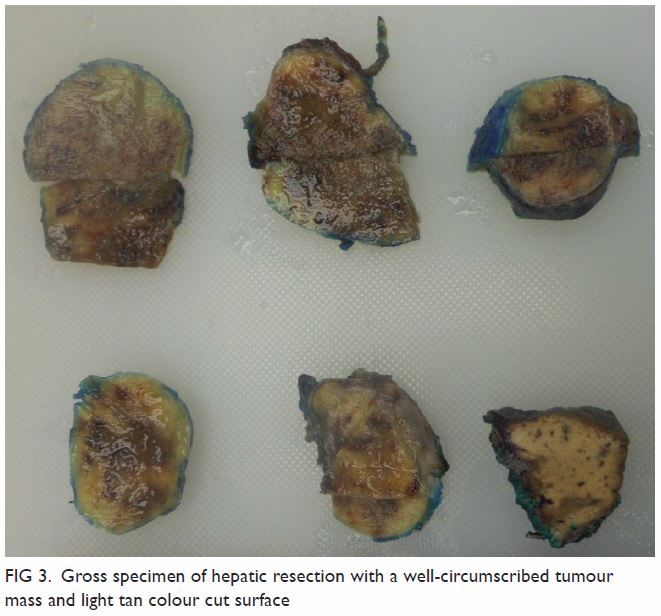
Figure 3. Gross specimen of hepatic resection with a well-circumscribed tumour mass and light tan colour cut surface
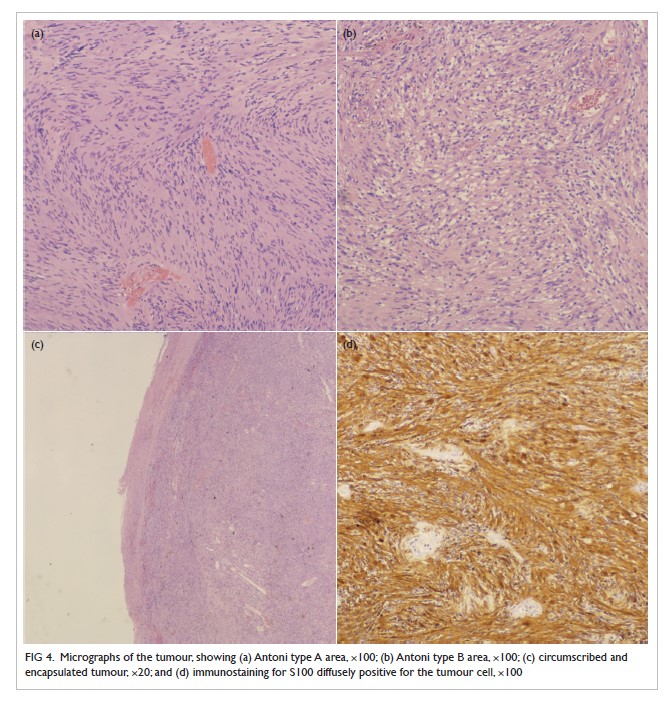
Figure 4. Micrographs of the tumour, showing (a) Antoni type A area, ×100; (b) Antoni type B area, ×100; (c) circumscribed and encapsulated tumour, ×20; and (d) immunostaining for S100 diffusely positive for the tumour cell, ×100
Schwannoma is most commonly found in the
limbs and the head and neck region. A fifth of cases
shows association with neurofibromatosis type 1.
The mediastinum and retroperitoneum are other
possible sites. It is uncommon in the gastrointestinal
tract and extremely rare in the liver.1 It was first
reported in 1978 by Pereira et al.2 A literature
search through PubMed and MEDLINE revealed
32 reported cases. No cases have been published in
Hong Kong.
The origin of hepatic schwannoma is the
hepatobiliary nerves among the hepatic plexus in
the liver hilum as well as interlobular connective
tissues and the hepatic arteries.1 3 They are usually
well-encapsulated and grow very slowly, usually
smaller than 5 cm at the time of diagnosis. Larger
schwannomas may undergo secondary degeneration
with consequent pseudocystic regression, haemorrhage, and calcification. Malignant
transformation is very rare.3
Pathologically, a schwannoma is an
encapsulated tumour that arises within the nerve
sheaths. It consists of a highly ordered cellular
component (Antoni type A area) characterised by
spindle cells with twisted nuclei arranged in short
bundles, and a hypocellular area in a loose myxoid
stroma (Antoni type B area) that comprises a loose
meshwork of gelatinous and microcystic tissue.4
On imaging, hepatic schwannoma is
usually well circumscribed with various signal
characteristics, depending on the distribution of
Antoni A and Antoni B areas.1 It is commonly of
low density with heterogeneous enhancement on CT, hypointense on T1-weighted, and hyperintense
on T2-weighted magnetic resonance imaging.5
There have also been reports of malignant tumours
but there are no distinct radiological features that
differentiate them from benign tumours.3 A hepatic
schwannoma may be fluorodeoxyglucose-avid
depending on inflammatory activity and cellularity.
Fluorodeoxyglucose-18 positron emission
tomography–CT alone may enable differentiation
of a schwannoma from malignant lesions of the
liver.1
Hepatic schwannoma is an extremely rare
tumour and preoperative diagnosis with imaging is
challenging. Biopsy or surgical resection is usually
required for definitive diagnosis.
Author contributions
Concept or design: All authors.
Acquisition of data: HL Tsui and CH Lau.
Analysis or interpretation of data: HL Tsui, SM Yu, and CH Lau.
Drafting of the manuscript: HL Tsui, SM Yu, and CH Lau.
Critical revision of the manuscript for important intellectual content: All authors.
Acquisition of data: HL Tsui and CH Lau.
Analysis or interpretation of data: HL Tsui, SM Yu, and CH Lau.
Drafting of the manuscript: HL Tsui, SM Yu, and CH Lau.
Critical revision of the manuscript for important intellectual content: All authors.
All authors had full access to the data, contributed to the
study, approved the final version for publication, and take
responsibility for its accuracy and integrity.
Conflicts of interest
All authors have disclosed no conflicts of interest.
Funding/support
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethics approval
This study is approved by the cluster Research Ethics
Committee (Ref KC/KE-19-0247/ER-3). Written patient
consent was also obtained.
References
1. Hayashi M, Takeshita A, Yamamoto K, Tanigawa N.
Primary hepatic benign schwannoma. World J Gastrointest
Surg 2012;4:73-8. Crossref
2. Pereira Filho RA, Souza SA, Oliveira Filho JA. Primary
neurilemmal tumour of the liver: case report. Arq
Gastroenterol 1978;15:136-8.
3. Ozkan EE, Guldur M, Uzunkoy A. A case report of benign
schwannoma of the liver. Intern Med 2010;49:1533-6. Crossref
4. Wan DL, Zhai ZL, Ren KW, Yang YC, Lin SZ, Zheng
SS. Hepatic schwannoma: a case report and an updated
40-year review of the literature yielding 30 cases. Mol Clin
Oncol 2016;4:959-64. Crossref
5. Yamamoto M, Hasegawa K, Arita J, et al. Primary
hepatic schwannoma: a case report. Int J Surg Case Rep
2016;29:146-50. Crossref