Breast lymphoma: a rare breast malignancy mimicking benign breast disease and carcinoma
Hong Kong Med J 2022 Oct;28(5):409.e1–4
© Hong Kong Academy of Medicine. CC BY-NC-ND 4.0
PICTORIAL MEDICINE
Breast lymphoma: a rare breast malignancy
mimicking benign breast disease and carcinoma
Carol PY Chien, MB, BS, FRCR1; Kimmy KM Kwok, FRCR, FHKAM (Radiology)2; Eliza PY Fung, FRCR, FHKAM (Radiology)2
1 Department of Radiology, Queen Mary Hospital, Hong Kong
2 Department of Diagnostic and Interventional Radiology, Kwong Wah Hospital, Hong Kong
Corresponding author: Dr Carol PY Chien (chienpyc@gmail.com)
In July 2020, a 52-year-old woman presented with
a 1-month history of palpable right breast lump.
Clinical examination revealed a hard mass at the upper
inner quadrant of the right breast. No skin changes
or nipple retraction were evident. Ultrasonography
confirmed multiple oval circumscribed hypoechoic
masses at the upper right breast measuring up to
2 cm that were considered likely benign (Fig 1). The
lesions were mammographically occult (Fig 2).
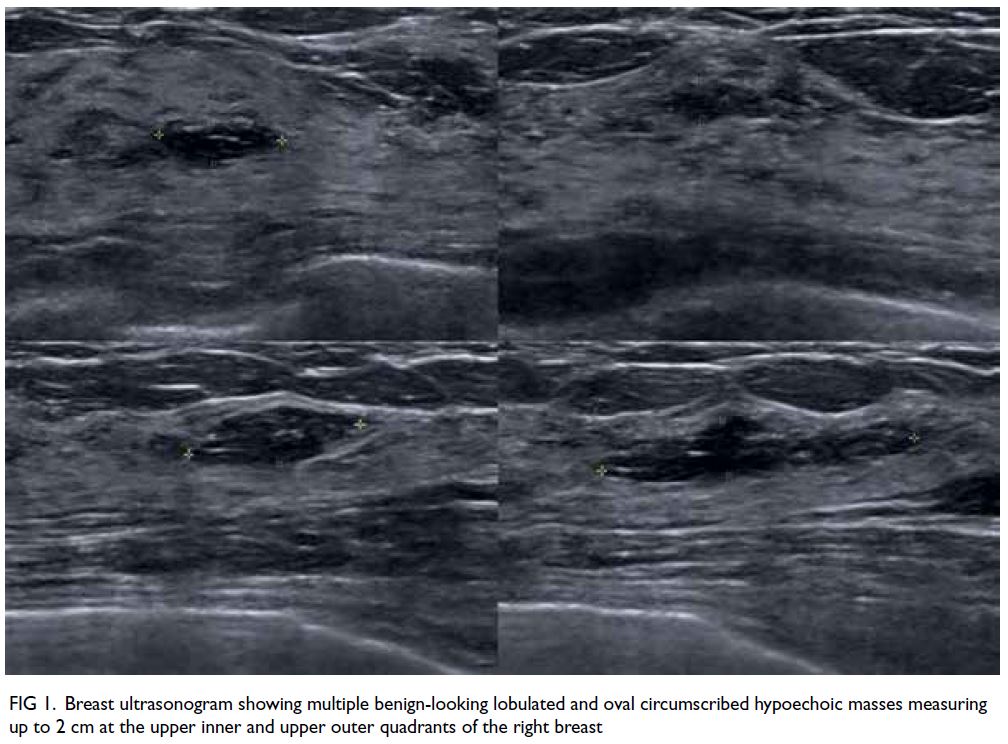
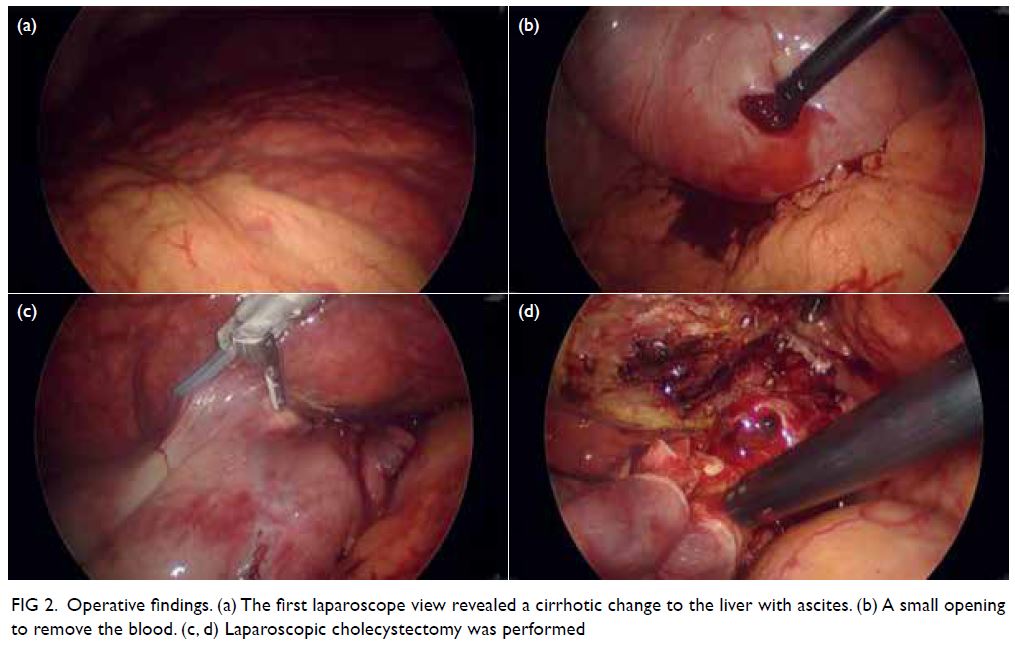
Figure 1. Breast ultrasonogram showing multiple benign-looking lobulated and oval circumscribed hypoechoic masses measuring up to 2 cm at the upper inner and upper outer quadrants of the right breast
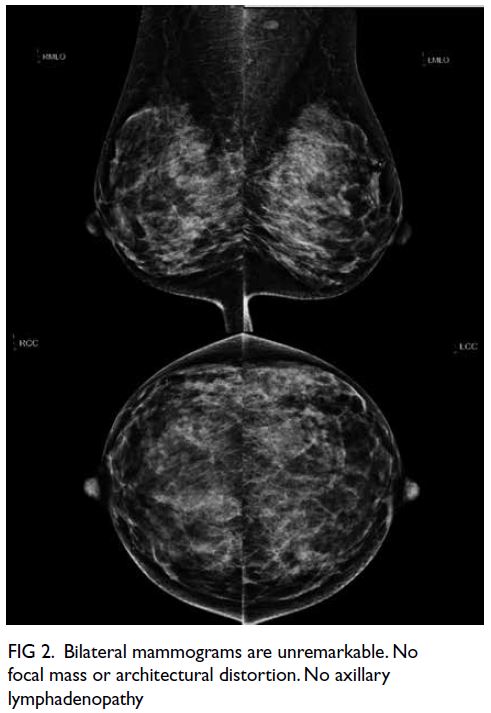
Figure 2. Bilateral mammograms are unremarkable. No focal mass or architectural distortion. No axillary lymphadenopathy
The patient also reported non-specific
abdominal pain and distension for a few weeks.
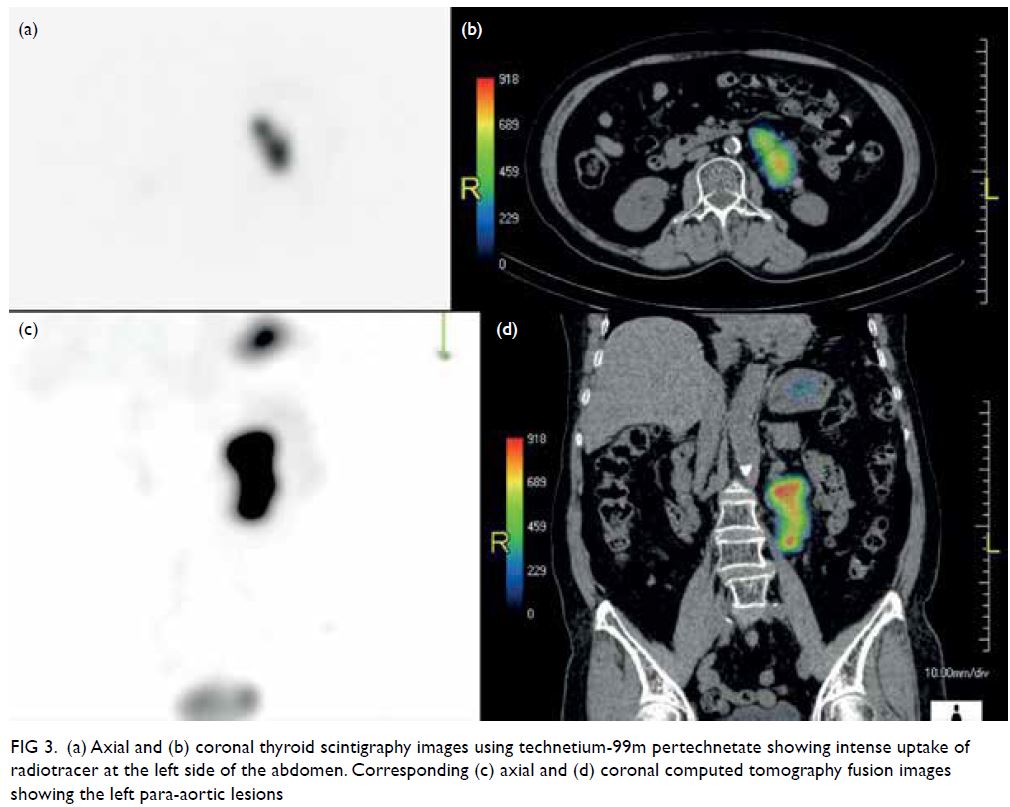
Positron emission tomography–computed
tomography (PET-CT) showed multiple
hypermetabolic masses at the right breast
(maximum standardised uptake value [SUVmax]
19.0, 16 mm) and abdominal lymphadenopathy
(Fig 3). Ultrasound-guided core biopsy of the right
breast mass confirmed diffuse large B-cell lymphoma
(Fig 4). The patient was diagnosed with stage IV lymphoma with diffuse extranodal involvement of
the right breast. Reassessment PET-CT showed
progressive disease despite R-CEOP chemotherapy
with enlargement and increased metabolic activity
of the right breast lymphoma (SUVmax 23.7, 41 mm)
and abdominal lymphadenopathy (Fig 5). Second-line
chemotherapy and radiotherapy of the breast
was administered.
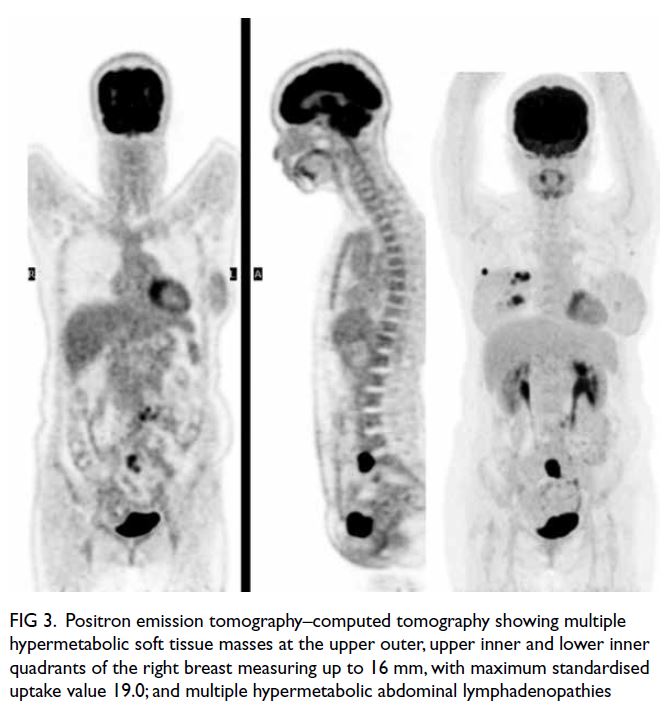
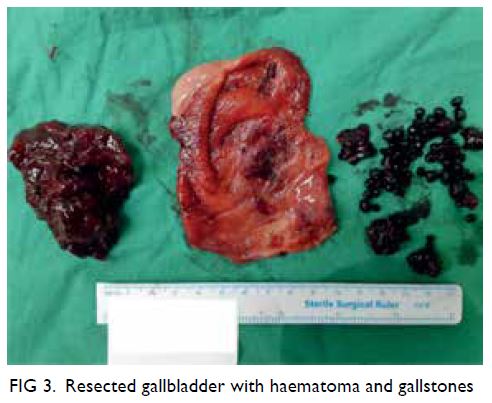
Figure 3. Positron emission tomography–computed tomography showing multiple hypermetabolic soft tissue masses at the upper outer, upper inner and lower inner quadrants of the right breast measuring up to 16 mm, with maximum standardised uptake value 19.0; and multiple hypermetabolic abdominal lymphadenopathies
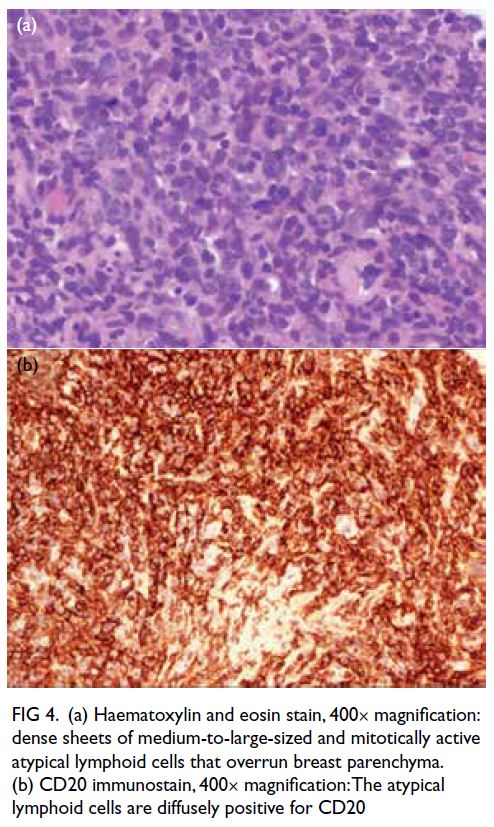
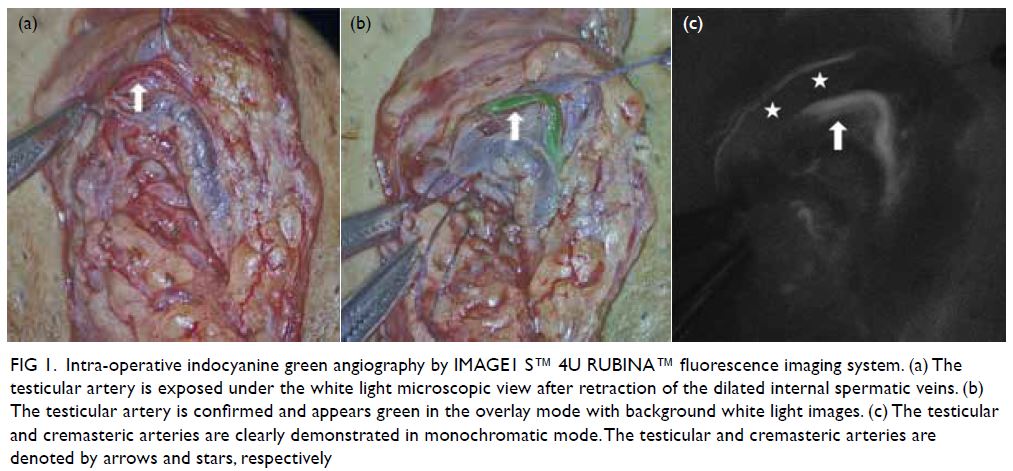
Figure 4. (a) Haematoxylin and eosin stain, 400× magnification: dense sheets of medium-to-large-sized and mitotically active atypical lymphoid cells that overrun breast parenchyma. (b) CD20 immunostain, 400× magnification: The atypical lymphoid cells are diffusely positive for CD20
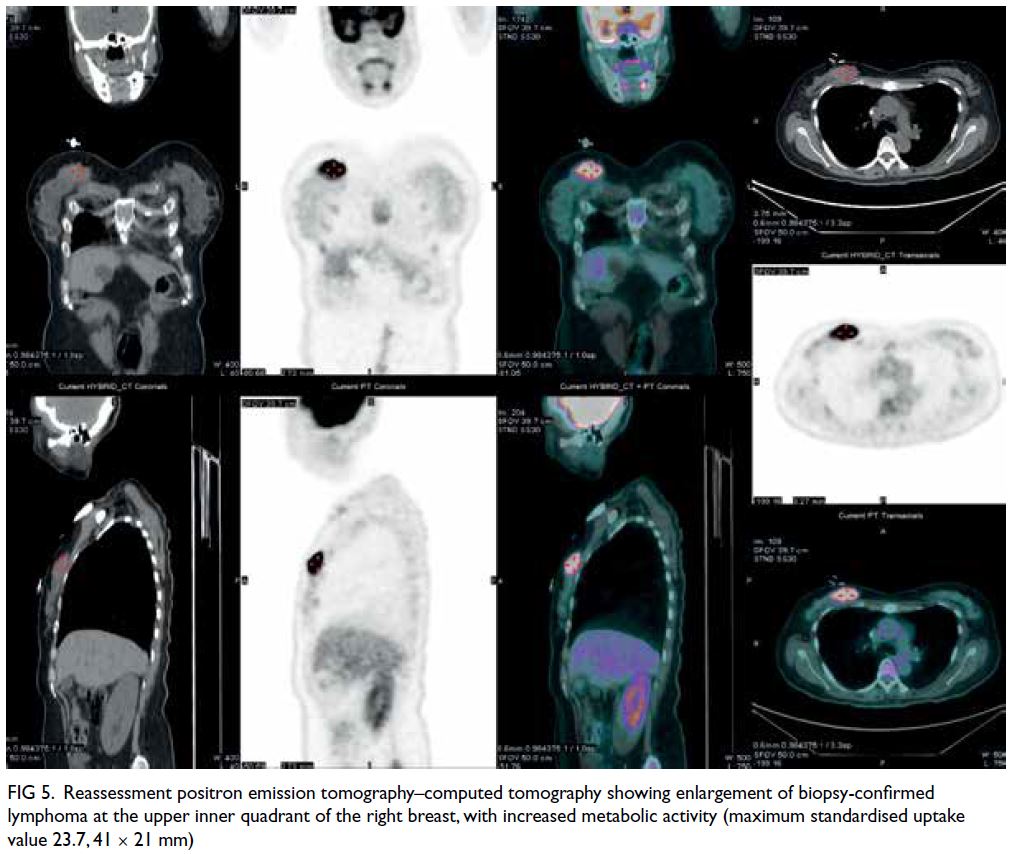
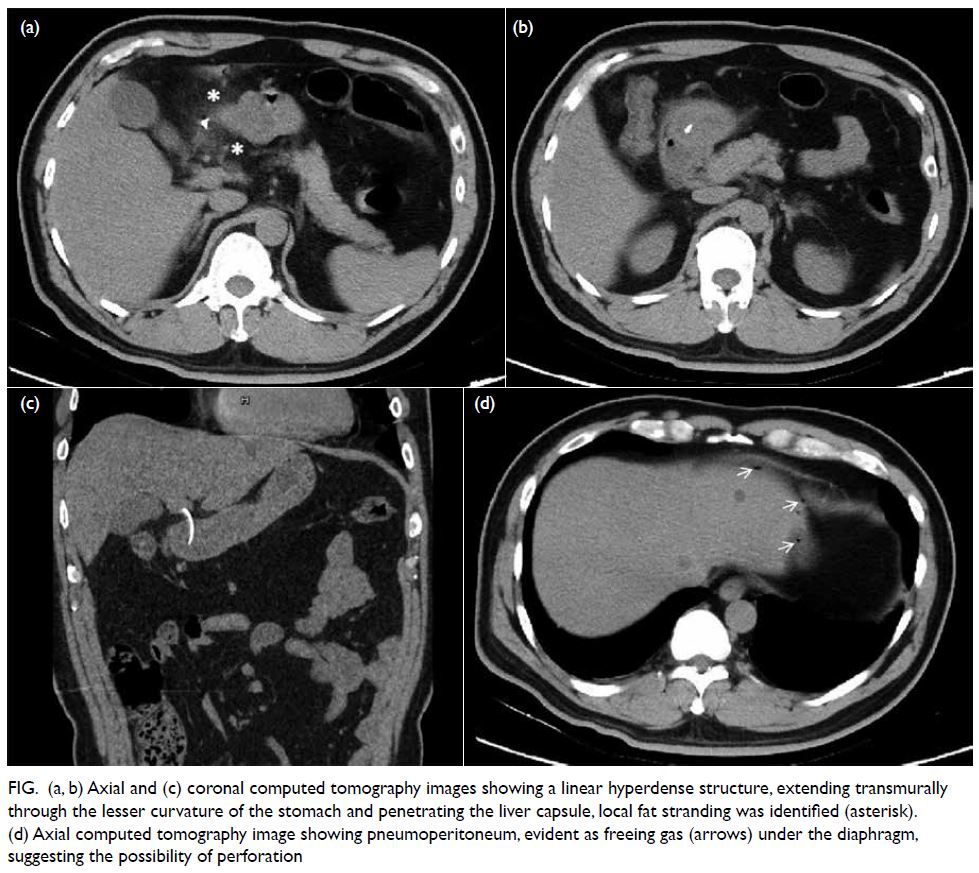
Figure 5. Reassessment positron emission tomography–computed tomography showing enlargement of biopsy-confirmed lymphoma at the upper inner quadrant of the right breast, with increased metabolic activity (maximum standardised uptake value 23.7, 41 × 21 mm)
The breast is an uncommon extranodal site
of involvement by lymphoma because of the lack
of lymphoid tissue. Breast lymphoma is a rare
malignancy of the breast that accounts for <1% of
all breast malignancies and <2% of all extranodal
non-Hodgkin lymphoma.1 Diffuse large B-cell
lymphoma is the most common histological type in
both primary and secondary breast lymphoma with
a median age at presentation of 60 to 70 years.1
The clinical presentation of breast lymphoma
is highly variable, so diagnosis is challenging. It commonly presents as a mass that mimics carcinoma,
or skin inflammatory changes that mimic benign
inflammatory breast disease or inflammatory breast
cancer.1 2 Axillary lymphadenopathy is uncommon.1
However, unlike with breast carcinoma, nipple
retraction or discharge and other skin changes are
rare with breast lymphoma.1 2
Mammographically, breast lymphoma
commonly presents as a solitary mass (69%-76%) and asymmetry (20%). It may also present
as skin thickening, architectural distortion or
lymphedema.3 4 In contrast to breast carcinoma,
spiculations, architectural distortion and
microcalcifications are distinctively absent with
breast lymphoma. Sonographically, breast lymphoma
commonly presents as a parallel benign-looking
hypoechoic mass without malignant features.3 4
Ultrasonography alone cannot distinguish breast
lymphoma from breast carcinoma or benign breast
lesions such as fibroadenoma. However, it can guide
tissue diagnosis when mammogram is negative. This
is particularly helpful in Asian patients with dense
breast tissue that lowers mammogram sensitivity.
A recent study has demonstrated that 18F-fluorodeoxyglucose
PET-CT can potentially
differentiate between breast lymphoma and
carcinoma.5 The authors found that the median
SUVmax for breast lymphoma (10.96) was
significantly higher than that for breast carcinoma
(4.76; specificity up to 96.3%).5 Although the
sonographic features of breast masses in our patient
were absent, an SUVmax of 19.0 was unusually
high for breast carcinoma. This raised the suspicion
of secondary breast lymphoma rather than
primary breast carcinoma with distant metastases.
Histological diagnosis was required to guide
appropriate treatment.
In conclusion, PET-CT can potentially
differentiate between breast lymphoma and breast
carcinoma. Increased awareness of breast lymphoma
and correlation with radiological and pathological
findings are essential for diagnosis.
Author contributions
Concept or design: All authors.
Acquisition of data: CPY Chien.
Analysis or interpretation of data: CPY Chien.
Drafting of the manuscript: CPY Chien.
Critical revision of the manuscript for important intellectual content: All authors.
Acquisition of data: CPY Chien.
Analysis or interpretation of data: CPY Chien.
Drafting of the manuscript: CPY Chien.
Critical revision of the manuscript for important intellectual content: All authors.
All authors had full access to the data, contributed to the study, approved the final version for publication and take responsibility for its accuracy and integrity.
Conflicts of interest
All authors have disclosed no conflicts of interest.
Funding/support
This study received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethics approval
The patient was treated in accordance with the Declaration of Helsinki and provided informed consent for all investigations and procedures, and publication.
References
1. Raj SD, Shurafa M, Shah Z, Raj KM, Fishman MD,
Dialani VM. Primary and secondary breast lymphoma:
clinical, pathologic, and multimodality imaging review.
Radiographics 2019;39:610-25. Crossref
2. Sabaté JM, Gómez A, Torrubia S, et al. Lymphoma of the breast: clinical and radiologic features with pathologic correlation in 28 patients. Breast J 2002;8:294-304. Crossref
3. Yang WT, Lane DL, Le-Petross HT, Abruzzo LV, Macapinlac HA. Breast lymphoma: imaging findings of 32
tumors in 27 patients. Radiology 2007;245:692-702. Crossref
4. Surov A, Holzhausen HJ, Wienke A, et al. Primary and
secondary breast lymphoma: prevalence, clinical signs and
radiological features. Br J Radiol 2012;85:e195-205. Crossref
5. Ou X, Wang J, Zhou R, et al. Ability of 18F-FDG PET/CT
radiomic features to distinguish breast carcinoma
from breast lymphoma. Contrast Media Mol Imaging
2019;2019:4507694. Crossref



 A video clip demonstrating systematic transperineal prostate biopsy is avaialble at
A video clip demonstrating systematic transperineal prostate biopsy is avaialble at