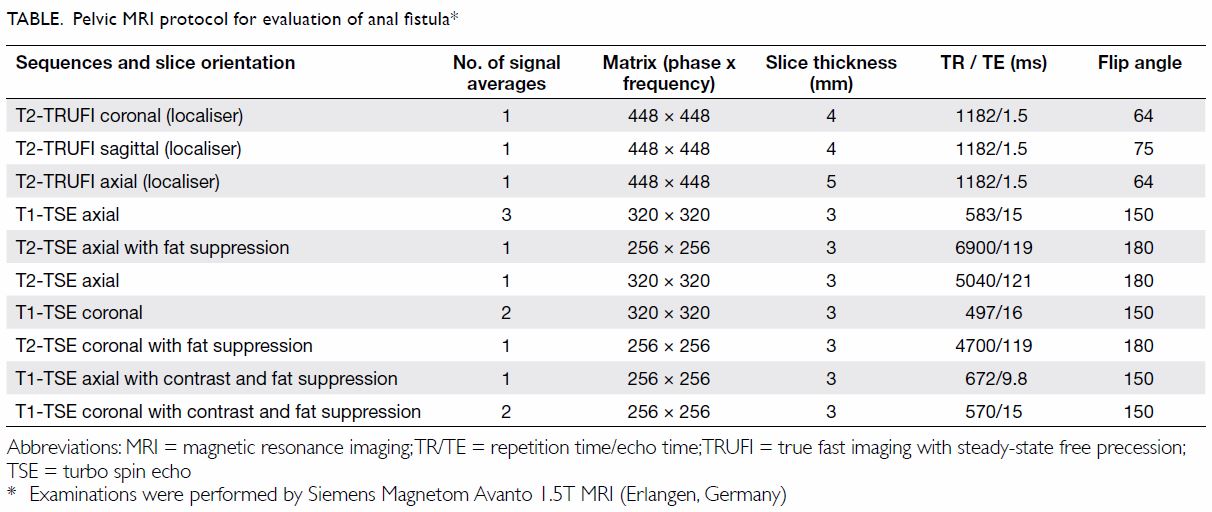
Diagnosis of Wunderlich syndrome in a patient with flank pain
Hong
Kong Med J 2019 Oct;25(5):406.e1–2
© Hong Kong Academy of Medicine. CC BY-NC-ND 4.0
PICTORIAL MEDICINE
Diagnosis of Wunderlich syndrome in a patient with
flank pain
YY Lin, MD; CW Hsu, PhD; HM Li, MD; HY Su, MD
Department of Emergency Medicine, E-Da Hospital,
I-Shou University, Kaohsiung, Taiwan
Corresponding author: Dr HY Su (hys927@hotmail.com)
In September 2018, a 62-year-old man
without underlying disease presented to the emergency
department of E-Da Hospital, Kaohsiung, Taiwan, with right flank pain for
1 day. The patient reported sharp and persistent pain radiating to the
right upper abdomen. On arrival at the emergency department, the patient
had heart rate 120 beats per minute and blood pressure 85/54 mm Hg.
Physical examination revealed right flank knocking tenderness. Laboratory
test results, including blood test and urinary analysis, were
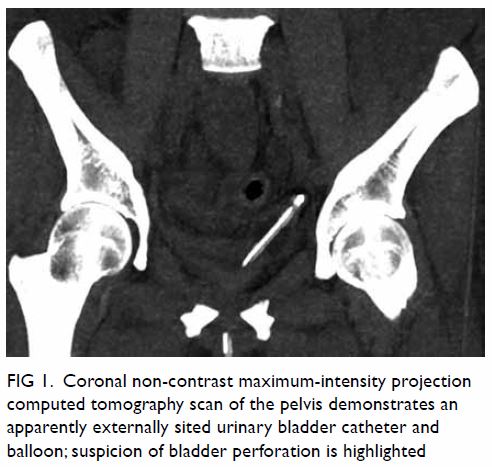
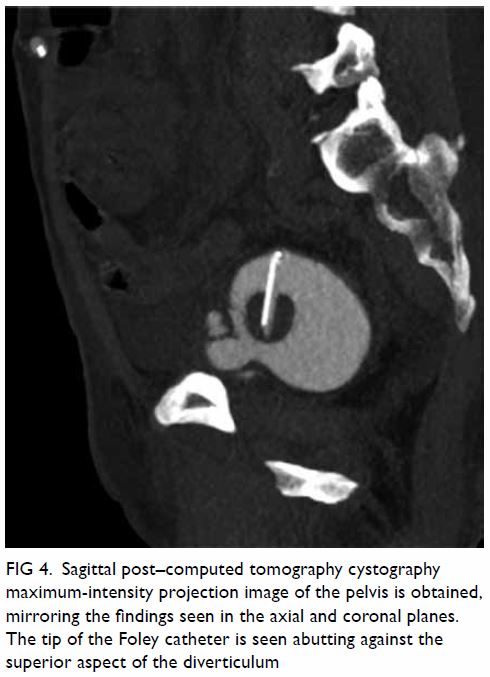
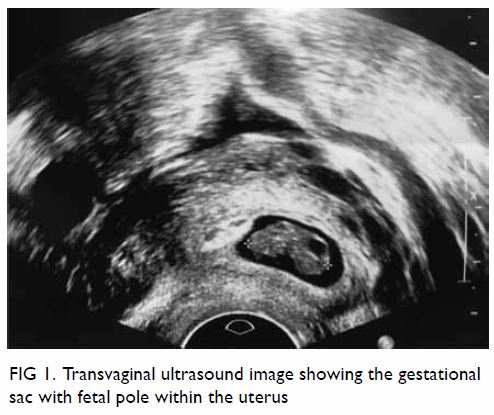
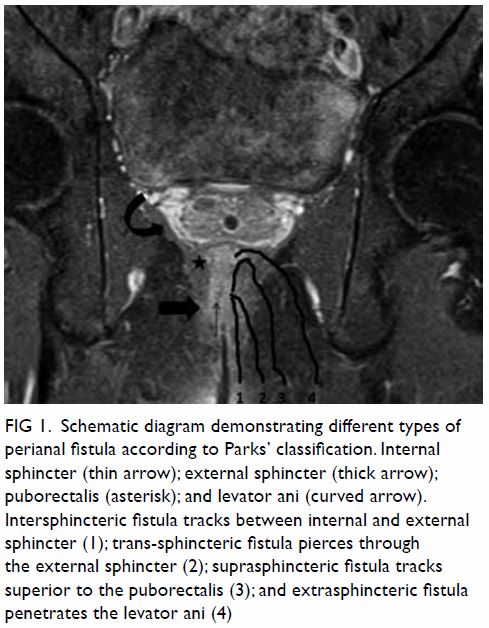
unremarkable. Abdominal plain film radiograph revealed a large right renal
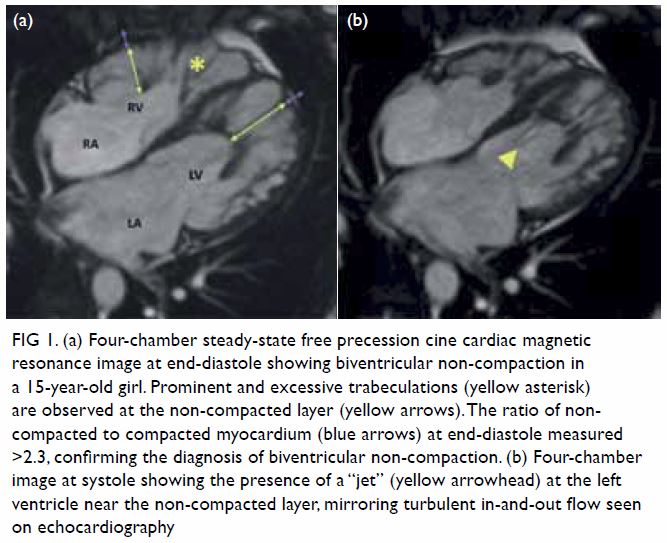
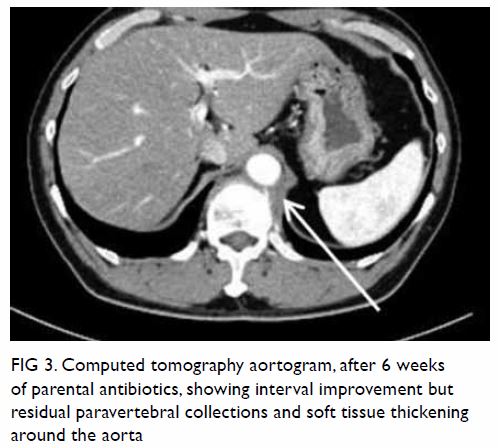
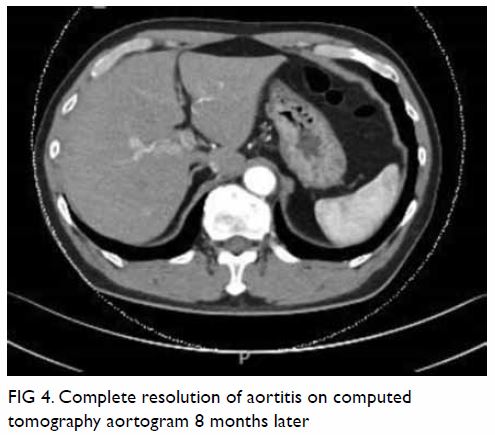
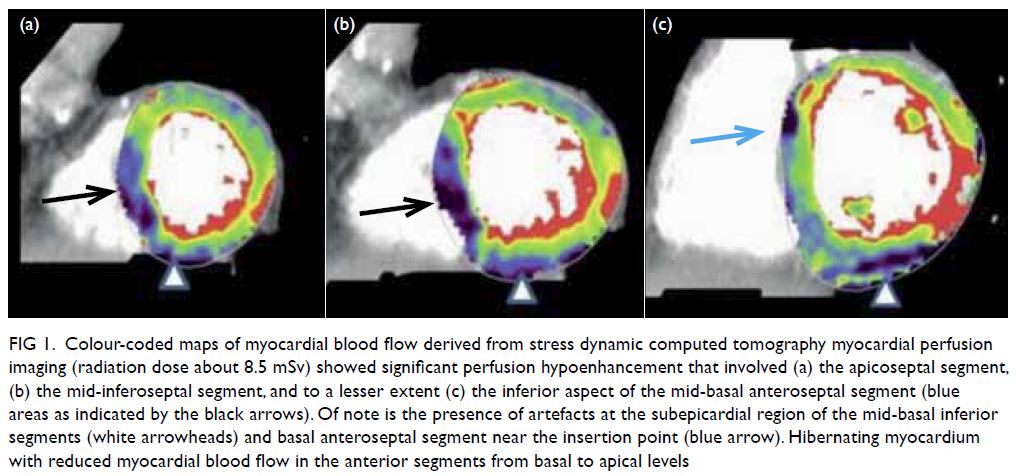
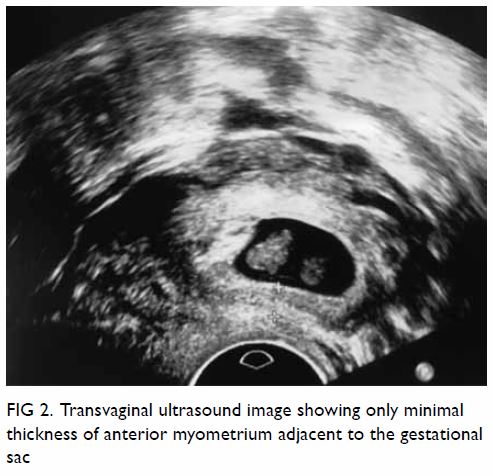
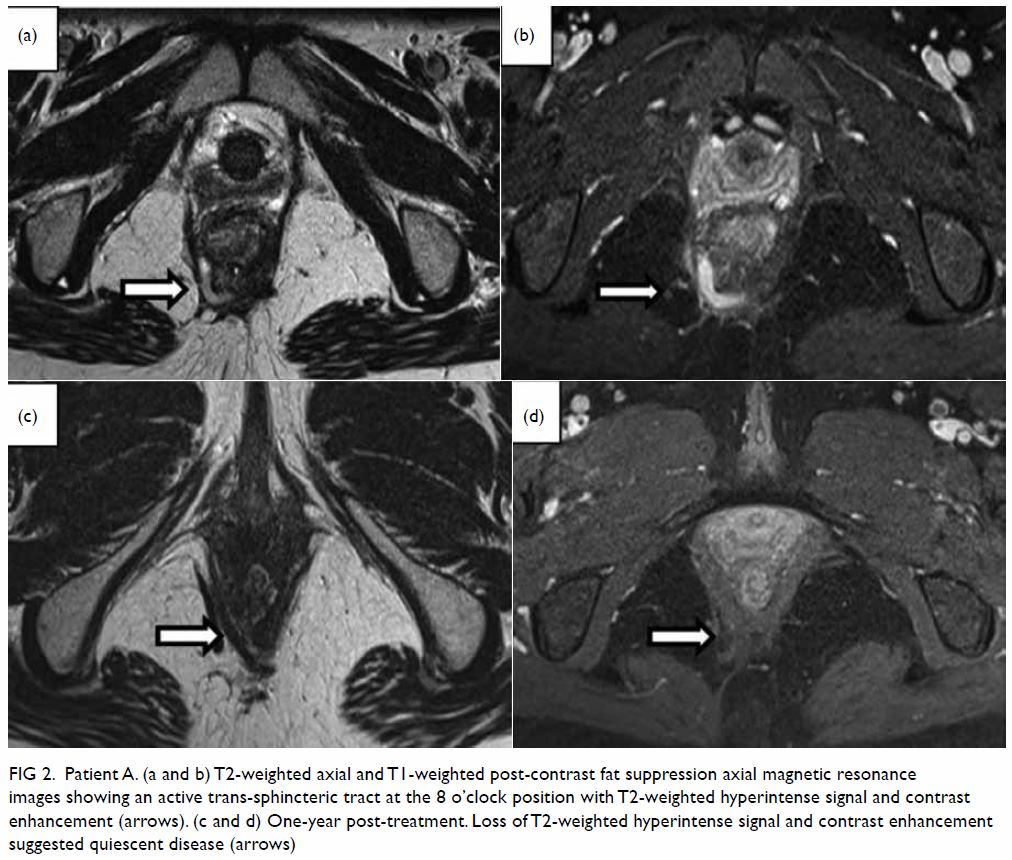
mass displacing surrounding structures (Fig 1). Point-of-care ultrasound demonstrated a
right renal mass with hyperechogenicity, which was surrounded by
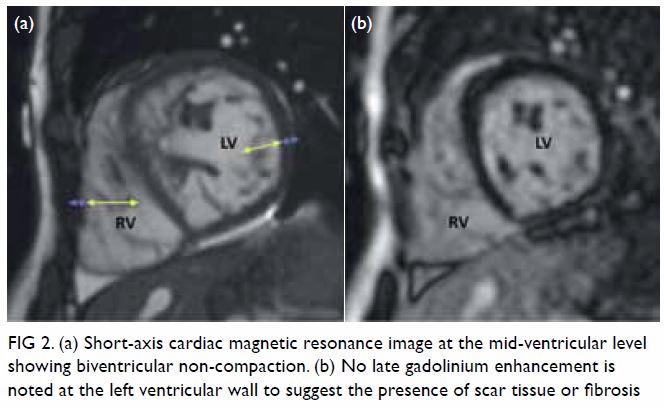
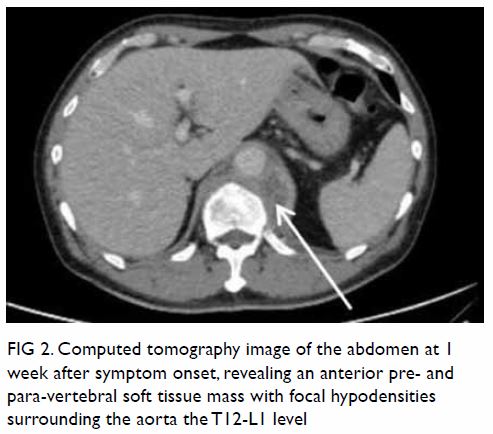
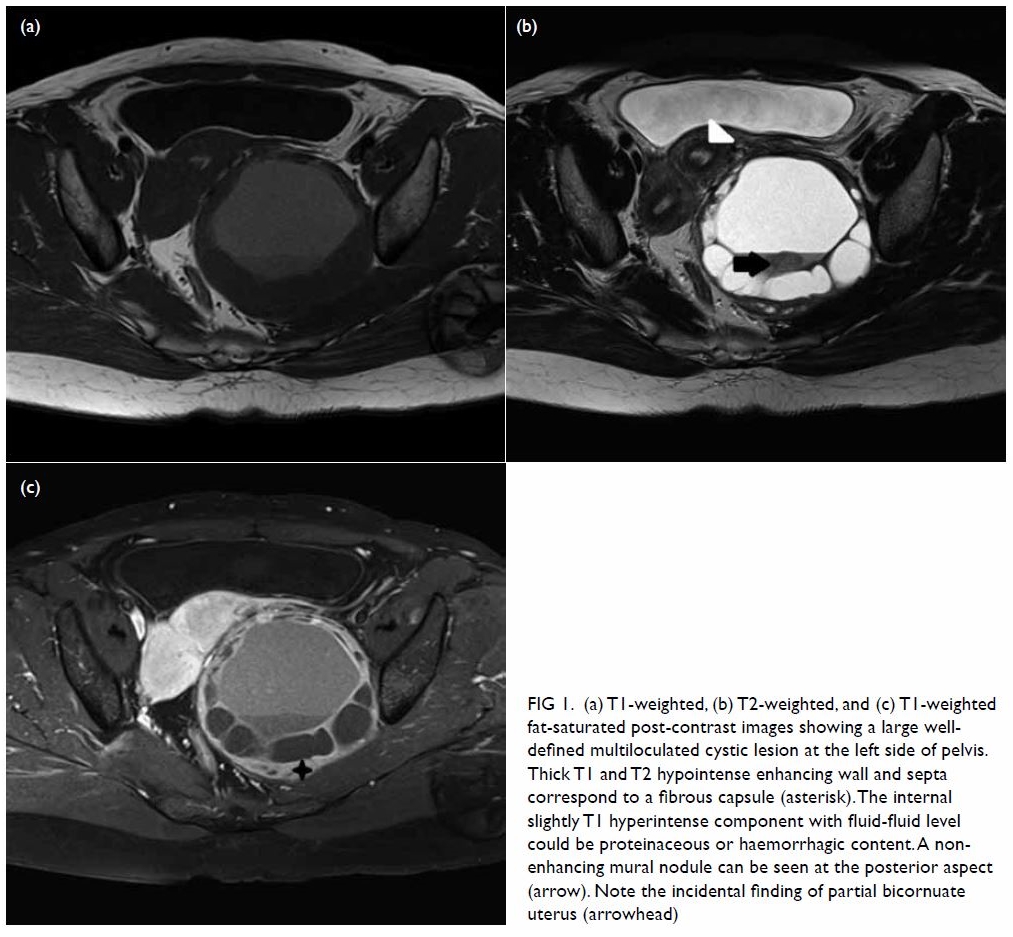
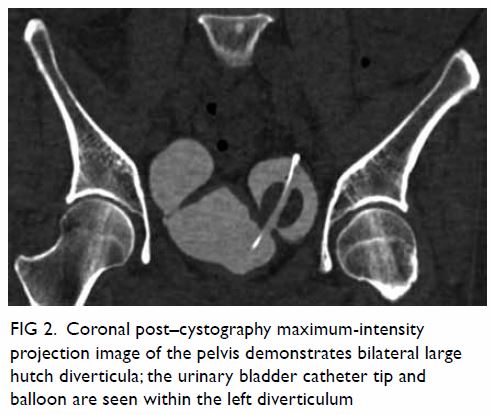
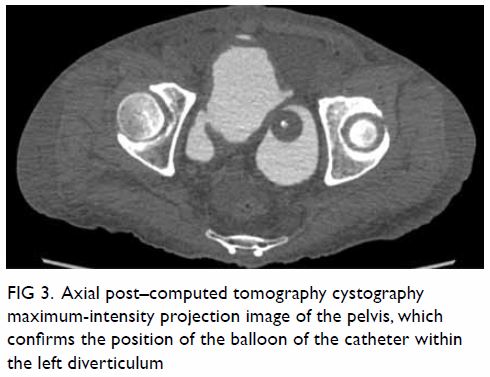
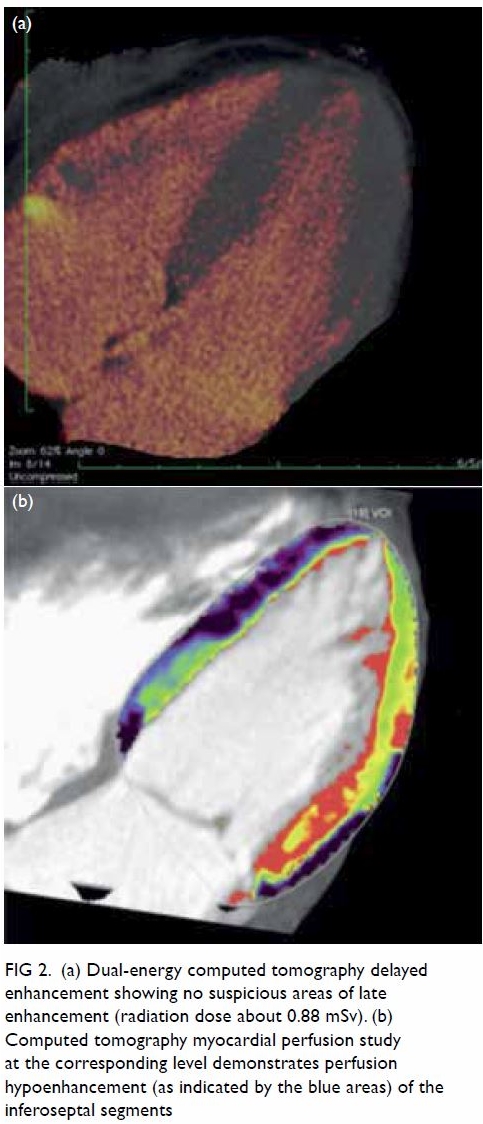
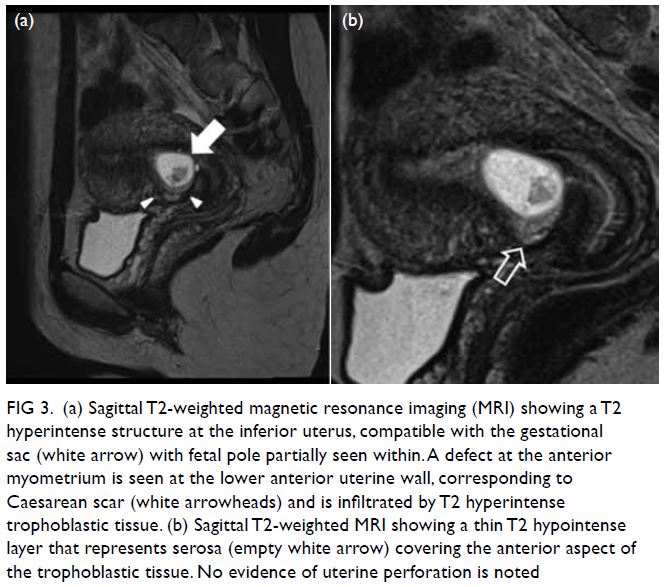
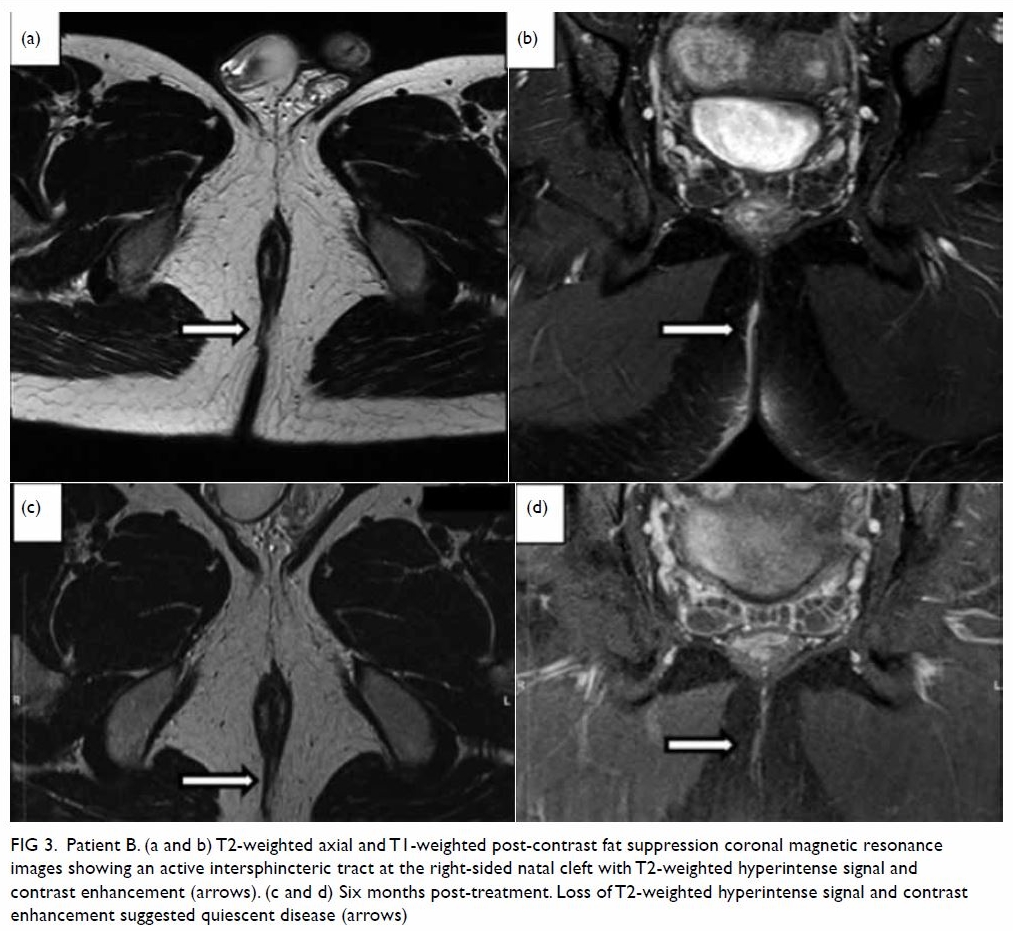
hypoechoic haematoma in the perinephric space (Fig 2). Subsequent abdominal computed tomography
(CT) revealed rupture of right renal angiomyolipoma with pericapsular
haematoma (Fig 3). Wunderlich syndrome complicated by
hypovolaemic shock was diagnosed, and proper fluid resuscitation and blood
transfusion were performed in the emergency department. The patient
received partial nephrectomy of right kidney on the next day, and was
discharged uneventfully from the hospital 2 weeks after admission.
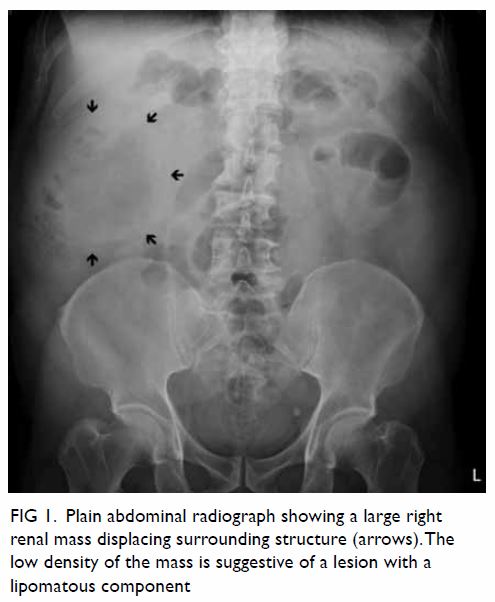
Figure 1. Plain abdominal radiograph showing a large right renal mass displacing surrounding structure (arrows). The low density of the mass is suggestive of a lesion with a lipomatous component
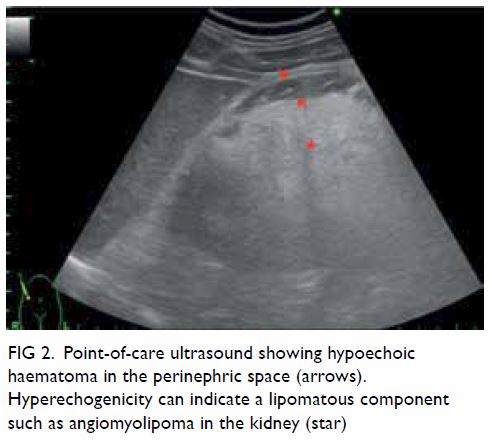
Figure 2. Point-of-care ultrasound showing hypoechoic haematoma in the perinephric space (arrows). Hyperechogenicity can indicate a lipomatous component such as angiomyolipoma in the kidney (star)
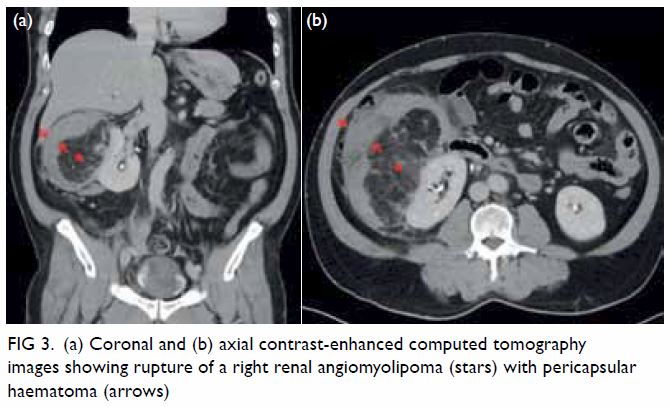
Figure 3. (a) Coronal and (b) axial contrast-enhanced computed tomography images showing rupture of a right renal angiomyolipoma (stars) with pericapsular haematoma (arrows)
Wunderlich syndrome, a rare but life-threatening
entity, is defined as spontaneous nontraumatic renal haemorrhage confined
to the subcapsular and perirenal space.1
Lenk’s triad, which consists of acute flank pain, palpable flank mass, and
hypovolemic shock, is the classical clinical feature of Wunderlich
syndrome.2 The aetiologies of
Wunderlich syndrome are classified into neoplastic and non-neoplastic
origins. Up to 60% of patients with Wunderlich syndrome are caused by
neoplasm, including benign tumours such as angiomyolipoma and malignancies
such as renal cell carcinoma.3 A
variety of diseases account for non-neoplastic origins of Wunderlich
syndrome, including vasculitis, renal artery aneurysm, arteriovenous
malformation, renal vein thrombosis, nephritis, cystic renal disease, and
coagulopathy.3 Angiomyolipoma, a
benign neoplasm composed of smooth muscle, adipose tissue, and
thick-walled blood vessels, is the most common cause of Wunderlich
syndrome.3 The risk of tumour
rupture leading to fatal internal haemorrhage increases when
angiomyolipoma grows >40 mm in diameter.4
Aneurism formation due to poor elastic vascular structure might be the
reason for angiomyolipoma rupture, especially during tumour growth.
For diagnosis of Wunderlich syndrome,
contrast-enhanced CT scan is a standard medical imaging modality with 100%
sensitivity in identifying perirenal haemorrhage.4
Computed tomography scan can present renal vascular structure, origins of
tumours and pathological change in adjacent tissues. Furthermore, CT scan
can also provide detailed vascular anatomy to provide a roadmap for
superselective renal embolisation in management of perirenal haemorrhage.
In contrast with CT scan, point-of-care ultrasound might be considered as
a prompt tool to diagnose patients with Wunderlich syndrome. Point-of-care
ultrasound can be used to screen the renal structure, quickly identify
internal bleeding, and evaluate the hemodynamic condition by measuring the
diameter of the inferior vena cava and assessing the cardiac preload and
contractility. Ultrasound can also facilitate the initial differential
diagnosis of patients with flank pain, such as renal colic, renal abscess
or acute pyelonephritis. Initial treatments for Wunderlich syndrome
include selective arterial embolisation and surgical intervention.
However, clinical guidelines for management of Wunderlich syndrome are not
yet well established.5 Selective
arterial embolisation has the advantage of minimal invasiveness, renal
preservation, and efficiency in treating acute renal haemorrhage. However,
surgical intervention can provide a delicate strategy for tumour
resection, especially if suspicious for malignancy, and prevent recurrent
tumour bleeding.5 Since Wunderlich
syndrome is a life-threatening condition, clinicians should be aware while
approaching patients presenting with flank pain and in shock to facilitate
timely emergency surgery or embolisation if needed.
Author contributions
All authors had full access to the data,
contributed to the study, approved the final version for publication, and
take responsibility for its accuracy and integrity.
Concept and design of the study: HY Su.
Acquisition of data: YY Lin.
Analysis or interpretation of data: YY Lin.
Drafting of the article: HY Su.
Critical revision for important intellectual content: HM Li, CW Hsu.
Acquisition of data: YY Lin.
Analysis or interpretation of data: YY Lin.
Drafting of the article: HY Su.
Critical revision for important intellectual content: HM Li, CW Hsu.
Conflicts of interest
All authors have disclosed no conflicts of
interest.
Funding/support
This study received no specific grant from any
funding agency in the public, commercial, or not-for-profit sectors.
Ethics approval
This study was conducted in accordance with the
principles outlined in the Declaration of Helsinki.
References
1. Medda M, Picozzi SC, Bozzini G,
Carmignani L. Wunderlich’s syndrome and hemorrhagic shock. J Emerg Trauma
Shock 2009;2:203-5. Crossref
2. Simkins A, Maiti A, Cherian SV.
Wunderlich syndrome. Am J Med 2017;130:e217-8. Crossref
3. Katabathina VS, Katre R, Prasad SR,
Surabhi VR, Shanbhogue AK, Sunnapwar A. Wunderlich syndrome:
cross-sectional imaging review. J Comput Assist Tomogr 2011;35:425-33. Crossref
4. Albi G, del Campo L, Tagarro D.
Wünderlich’s syndrome: causes, diagnosis and radiological management. Clin
Radiol 2002;57:840-5. Crossref
5. Flum AS, Hamoui N, Said MA, et al.
Update on the diagnosis and management of renal angiomyolipoma. J Urol
2016;195(4 Pt 1):834-46. Crossref