Inadvertent arterial insertion of a central venous catheter
Hong
Kong Med J 2018 Aug;24(4):427.e1-2
DOI: 10.12809/hkmj176866
© Hong Kong Academy of Medicine. CC BY-NC-ND 4.0
PICTORIAL MEDICINE
Inadvertent arterial insertion of a central venous
catheter
Victor WT Chan, MB, BS, FRCR; KW Shek, MB, BS,
FRCR
Department of Radiology and Imaging, Queen
Elizabeth Hospital, Jordan, Hong Kong
Corresponding author: Dr Victor WT Chan (chanwaitat@gmail.com)
Central venous catheterisation is a common
procedure that allows venous access for delivering medications, infusing
fluids or blood products, and monitoring volume status. Traditionally,
anatomical landmarks of the sternocleidomastoid muscle provide a pathway
to catheterise the internal jugular vein (IJV). However, inadvertent
arterial puncture is a risk. Currently, ultrasound guidance by experienced
operators is recommended for reducing the risk of mechanical complications
during central venous catheter (CVC) insertion.1
For venous access via the neck, common carotid and
subclavian artery injuries have been reported.2
The risk of artery injury is about 0.5%, and practice should be reviewed
if the risk exceeds 1%. Other known mechanical complications include
haematoma formation, haemothorax, and pneumothorax.
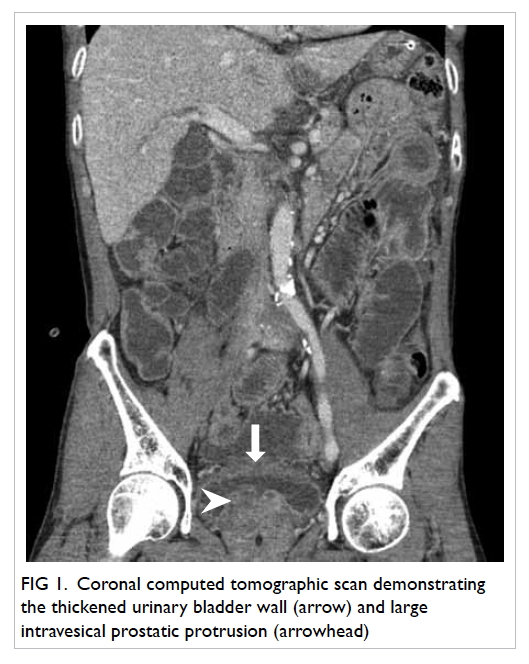
A 55-year-old woman with end-stage renal failure on
continuous ambulatory peritoneal dialysis was admitted to our hospital for
fever and abdominal pain. The clinical diagnosis of continuous ambulatory
peritoneal dialysis peritonitis was made, and the peritoneal dialysis
catheter was removed. Bedside insertion of a CVC was selected for
temporary haemodialysis. The CVC insertion was initially attempted via the
right IJV, unsuccessfully. The CVC was subsequently inserted via the left
IJV. The procedure was performed under ultrasound guidance using the
Seldinger technique; however, inadvertent arterial puncture was not
recognised, and the procedure was continued.
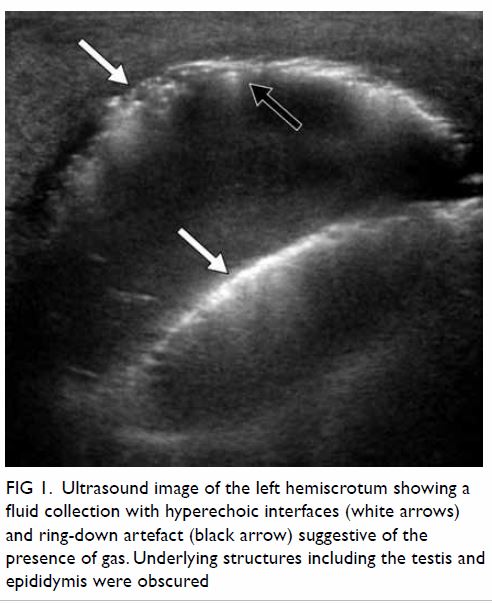
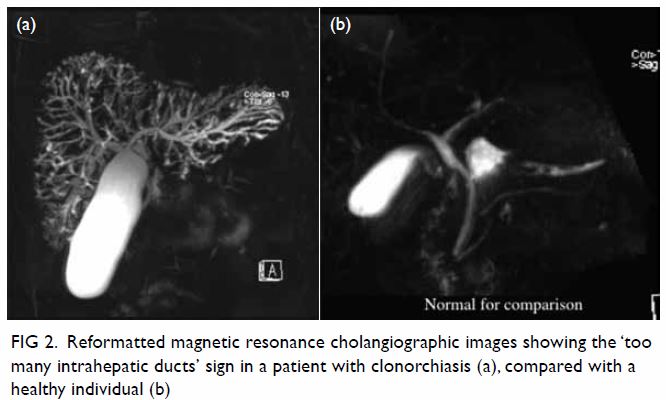
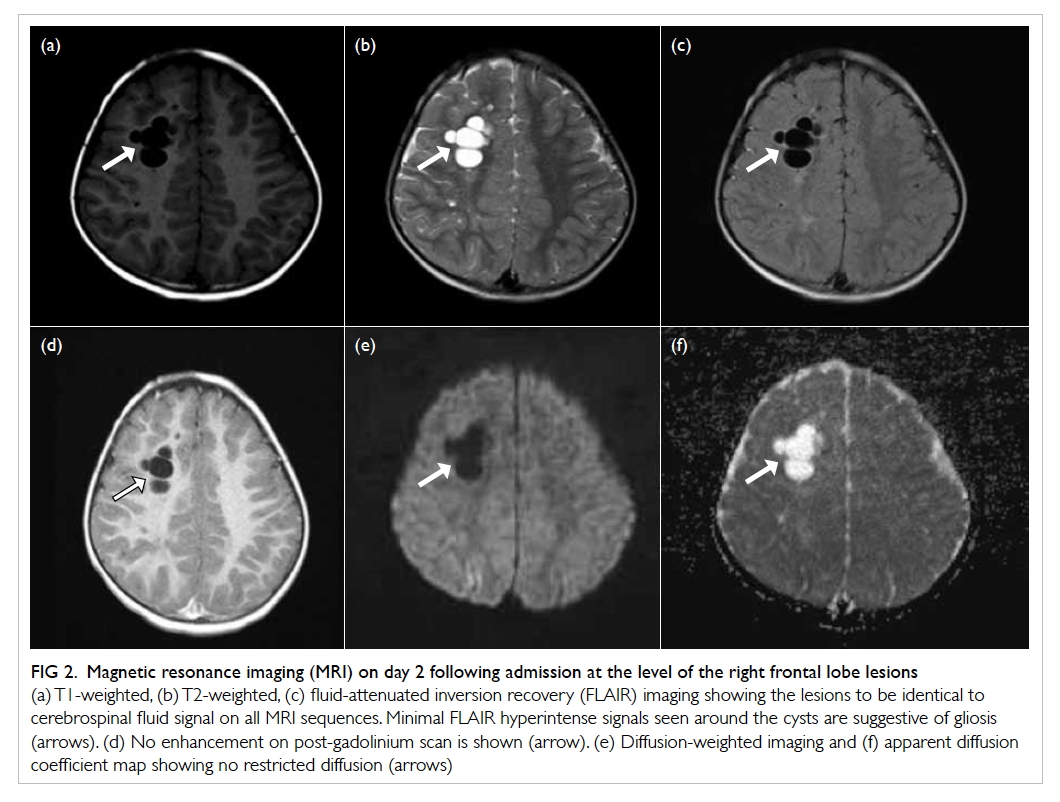
After CVC insertion, a chest radiograph was taken,
showing an abnormal vertical course of the catheter with suspected
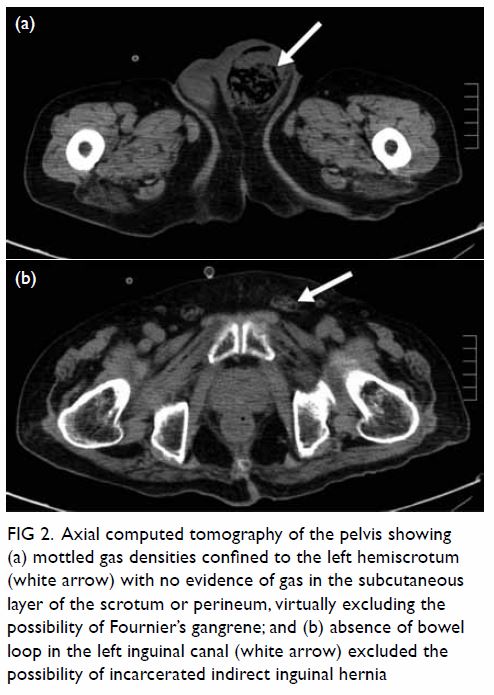
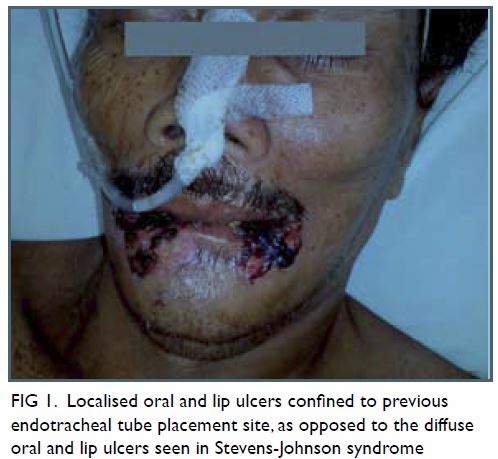
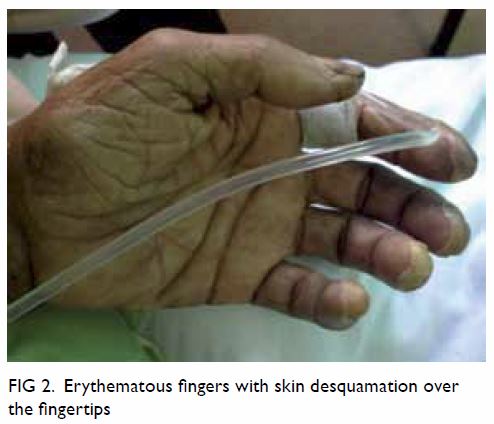
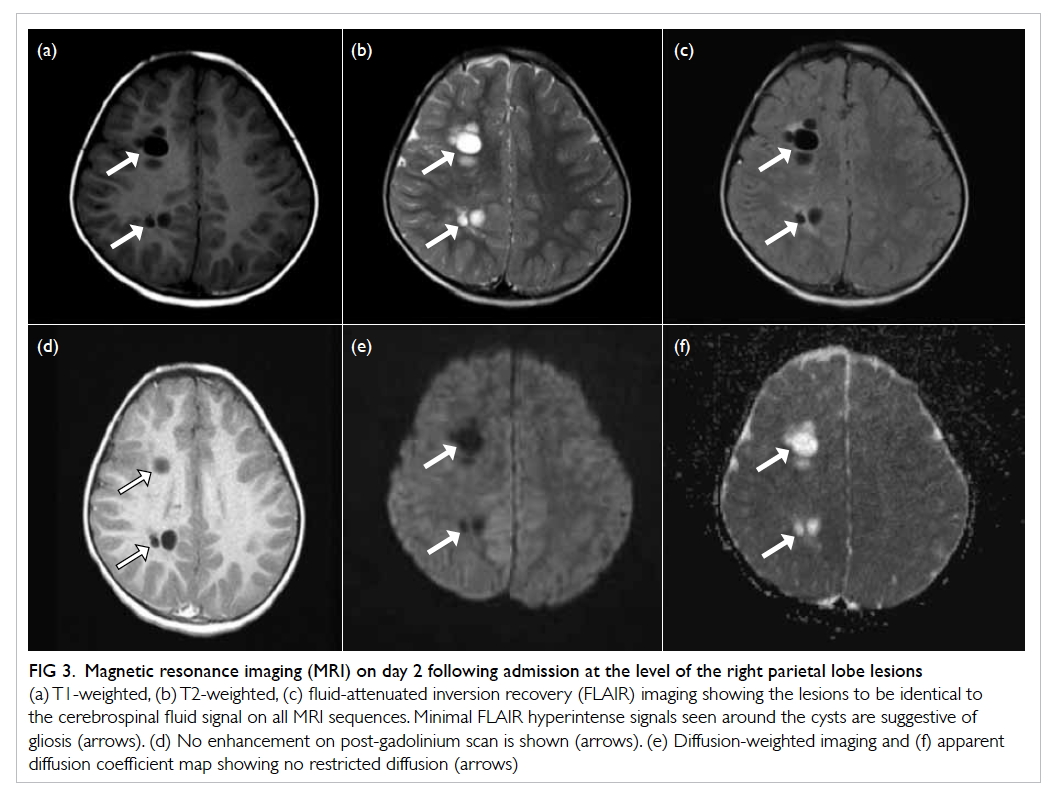
malposition (Fig 1). Urgent contrast computed tomographic
angiogram (Figs 2 and 3) revealed that the catheter had been inserted via
the left IJV, subsequently exiting posteromedially, entering the left
vertebral artery, and harbouring at the origin of the left subclavian
artery. Computed tomographic angiogram also showed abnormal contrast
pooling over the right neck suggestive of a pseudoaneurysm formation from
the right subclavian artery.
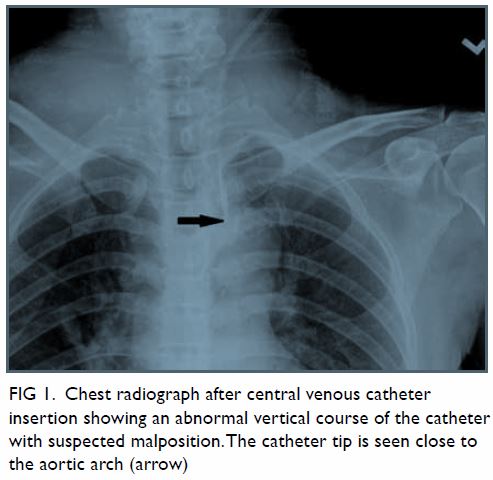
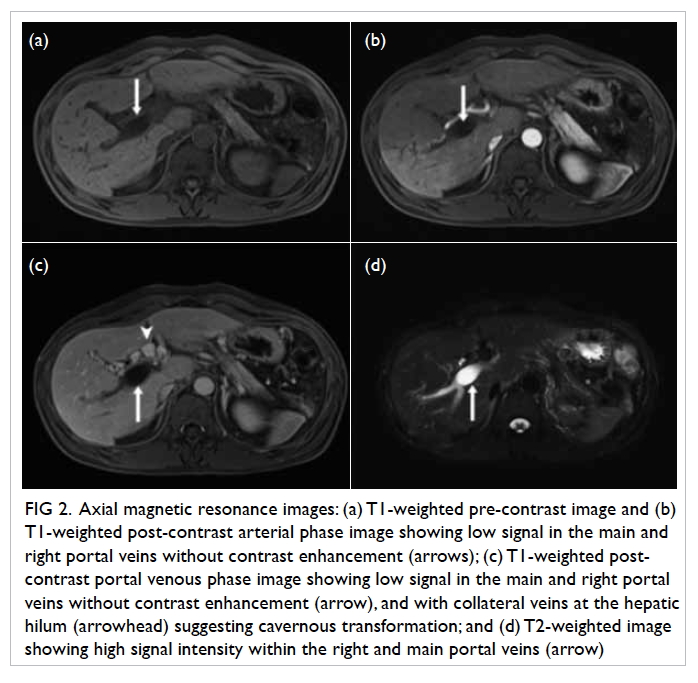
Figure 1. Chest radiograph after central venous catheter insertion showing an abnormal vertical course of the catheter with suspected malposition. The catheter tip is seen close to the aortic arch (arrow)
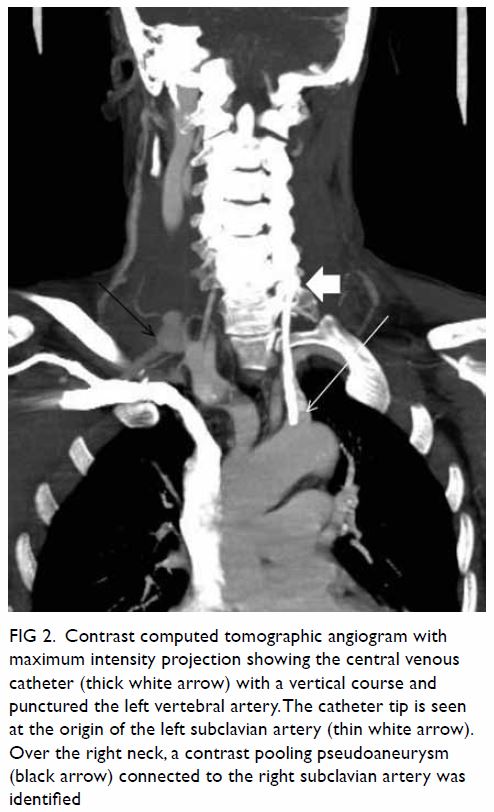
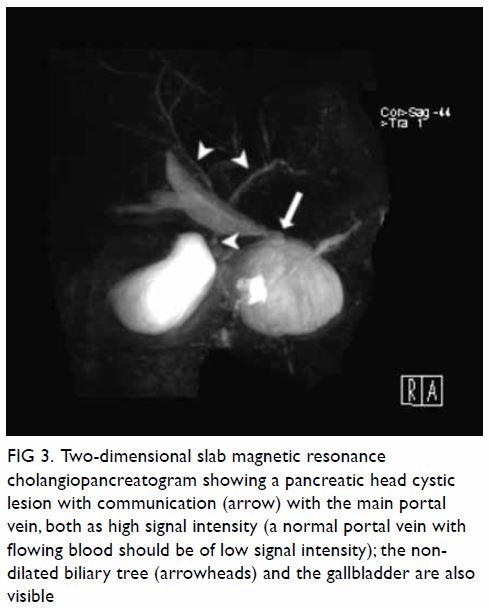
Figure 2. Contrast computed tomographic angiogram with maximum intensity projection showing the central venous catheter (thick white arrow) with a vertical course and punctured the left vertebral artery. The catheter tip is seen at the origin of the left subclavian artery (thin white arrow). Over the right neck, a contrast pooling pseudoaneurysm (black arrow) connected to the right subclavian artery was identified
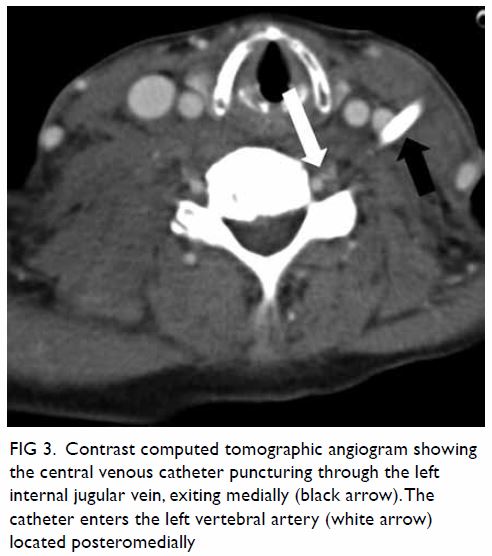
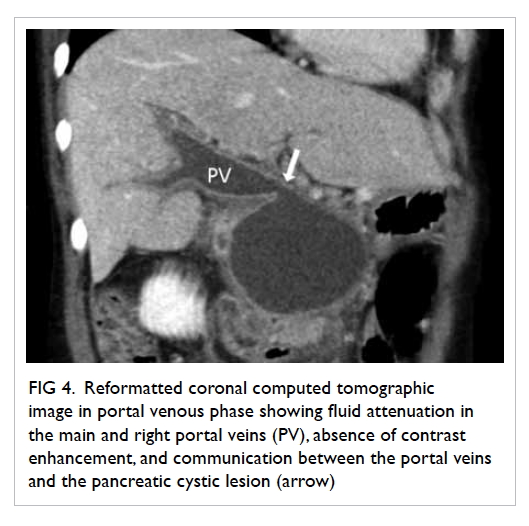
Figure 3. Contrast computed tomographic angiogram showing the central venous catheter puncturing through the left internal jugular vein, exiting medially (black arrow). The catheter enters the left vertebral artery (white arrow) located posteromedially
The opinion of a vascular surgeon was sought and
the catheter was removed under general anaesthesia with repair of the
vertebral artery. Oozing was noted from the left IJV exit site.
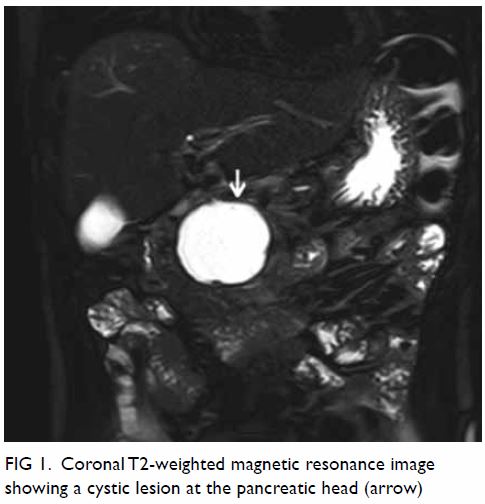
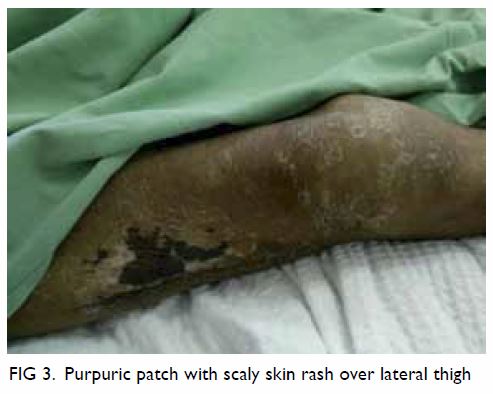
Haemostasis was controlled by direct pressure onto the IJV. Urgent right
subclavian angiogram (Fig 4) was performed by interventional radiologists,
confirming the presence of a pseudoaneurysm, which was successfully
embolised with coils.
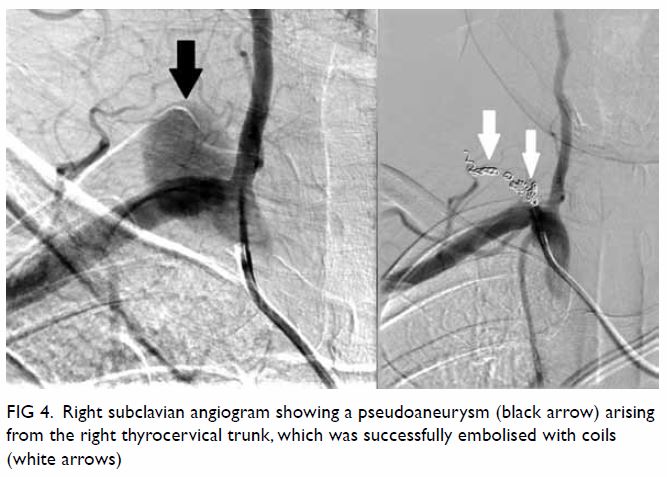
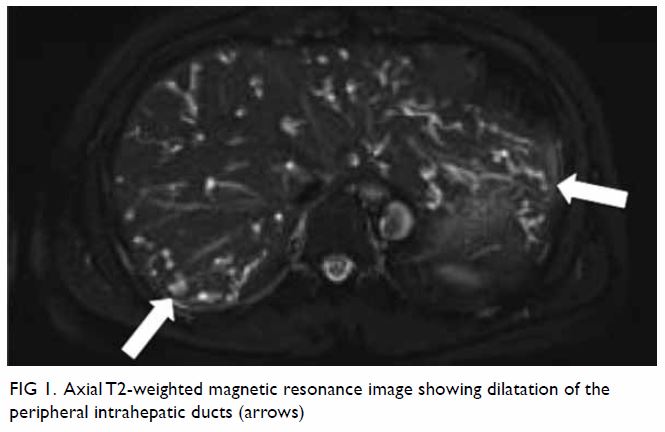
Figure 4. Right subclavian angiogram showing a pseudoaneurysm (black arrow) arising from the right thyrocervical trunk, which was successfully embolised with coils (white arrows)
Postoperatively, the patient progressed well, with
her peritonitis controlled by intravenous antibiotics. A new CVC for
temporary haemodialysis was inserted via her right IJV by interventional
radiologists under fluoroscopic and real-time ultrasound guidance.
This case concurs with a previous report that the
incidence of mechanical complications after multiple attempts is higher
than after one attempt.2 Real-time
ultrasound guided venepuncture of the IJV has a higher first insertion
attempt success rate, and decreased rate of arterial puncture as compared
with the anatomic landmark approach; this technique is currently
recommended by the Association of Anaesthetists of Great Britain and
Ireland for all CVC insertions.3
Credentialing of ultrasound-guided CVC insertion should be advocated in
Hong Kong, with adequate training provided by accredited trainers.
Author contributions
All authors have made substantial contributions to
the concept of this pictorial medicine; acquisition and interpretation of
data, drafting of the article, and critical revision for important
intellectual content.
Funding/support
This research received no specific grant from any
funding agency in the public, commercial, or not-for-profit sectors.
Declaration
All authors have disclosed no conflicts of
interest. All authors had full access to the data, contributed to the
study, approved the final version for publication, and take responsibility
for its accuracy and integrity.
References
1. McGee DC, Gould MK. Preventing
complications of central venous catheterization. N Engl J Med
2003;348:1123-33. Crossref
2. Schummer W, Schummer C, Rose N, Niesen
WD, Sakka SG. Mechanical complications and malpositions of central venous
cannulations by experienced operators. A prospective study of 1794
catheterizations in critically ill patients. Intensive Care Med
2007;33:1055-9. Crossref
3. Bodenham A, Babu S, Bennett J, et al.
Association of Anaesthetists of Great Britain and Ireland: Safe Vascular
Access 2016. Anaesthesia 2016;71:573-85. Crossref