Hong Kong Med J 2014;20:78.e1–2 | Number 1, February 2014
DOI: 10.12809/hkmj133796
© Hong Kong Academy of Medicine. CC BY-NC-ND 4.0
PICTORIAL MEDICINE
Ketamine-induced cholangiopathy
KL Lui, MMedSc, FHKCP1; WK Lee, FRCPath, FHKAM (Pathology)2; Michael KK Li, FRCP, FHKAM (Medicine)
1 Division of Gastroenterology and Hepatology, Department of Medicine
and Geriatrics, Tuen Mun Hospital, Tuen Mun, Hong Kong
2 Department of Pathology, Tuen Mun Hospital, Tuen Mun, Hong Kong
Corresponding author: Dr KL Lui (klluitc@yahoo.com.hk)
A 28-year-old woman presented to us in November
2010 because of deranged liver function test
results; predominantly she had raised ductal
enzyme levels (gamma-glutamyl transferase,
1088; reference range, 12-57 IU/mL); alkaline
phosphatase (ALP) 579 (reference range, 46-127)
IU/mL, alanine transaminase (ALT) 183 (reference
range, 10-57) IU/mL with normal bilirubin
levels. Upon further questioning, she had been a
ketamine abuser for 5 years and was followed up
by psychiatrists. She was completely asymptomatic
and physical examination yielded nil abnormal.
Her ALP level was excessive (154 IU/mL) and her
ALT level was 48 IU/mL. Ultrasound of hepatobiliary
system (HBS) showed a dilated common bile
duct (CBD) of 1.1 cm in diameter with tapering
over lower end. A gallstone was present in the
gallbladder. Therefore, the endoscopic retrograde
cholangiopancreatography (ERCP) was performed
in November 2011, and showed a 5-cm stricture
at the lower end of the CBD together with small
bilateral segmental strictures in the intrahepatic
ducts (Fig 1). Brush cytology of the stricture of
CBD revealed no malignant cells. A plastic stent
bypassing the CBD was inserted for drainage. Liver
function test findings did not improve after stenting
but repeated ultrasonography of the HBS showed
that with the 5.7-mm stent in situ, the CBD was not dilated. A liver biopsy was therefore performed,
and showed mild-to-moderate portal fibrosis with
ductular proliferation (Fig 2) and periportal copper
deposits were noted (Fig 3). These findings were
consistent with chronic cholestasis at both the
extrahepatic and intrahepatic level. There were no
features suggestive of primary biliary cirrhosis, or
primary sclerosing cholangitis. The colonoscopy was
normal and showed no evidence of inflammatory
bowel disease. The patient’s liver function improved after she ceased the recreational use of ketamine.
However, her stricture remained unchanged in the
follow-up ERCP and repeated biopsies over the CBD
stricture only showed reactive changes.
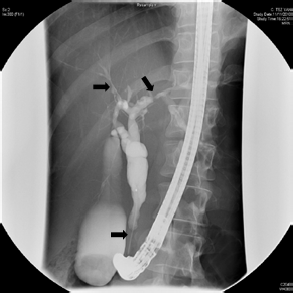
Figure 1. A 5-cm stricture at the lower end of the common bile duct with irregularity over intrahepatic ducts on both sides (arrows)
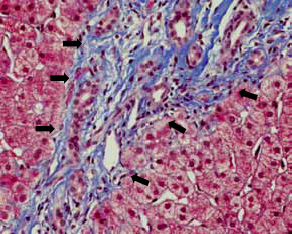
Figure 2. Intermediate power view showing portal fibrosis and ductal proliferation (arrows) [Masson’s trichrome stain, original magnification x 200]
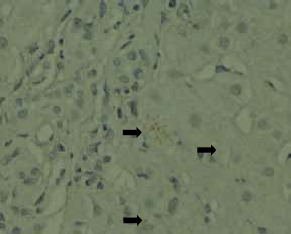
Figure 3. Small periportal copper deposits are evident (arrows) [orange-red granules, rhodanine stain; original magnification x 400]
The first report on the association of liver injury
with ketamine dates back to 1980.1 The exact cause
of the ketamine-induced stricture is not known, but
chronic use is associated with hepatocyte damage
and fibrosis to the liver.2 Ketamine intake also
stimulates the N-methyl-D-aspartic acid receptor in
the smooth muscle cells of the bile duct and chronic
stimulation may induce inflammation and fibrosis
finally resulting in strictures.3 4 Affected patients are
usually asymptomatic initially, and only manifest
abnormal ductal enzyme level after 1 to 2 years of
recreational ketamine use, indicating that chronicity
and repeated use seem to be involved. Both
intrahepatic and extrahepatic stricture might also
develop and complicated with cholangitis, especially
in the presence of gallstones. Definitive management
entails cessation of ketamine intake, whereupon
liver function improves, though the stricture may
be permanent and warrant stenting to relieve any
obstruction.4 5 This case report points that ketamine
abuse also causes liver and biliary damage, quite
apart from urinary and neurological sequelae.
References
1. Dundee JW, Fee JP, Moore J, Mcllroy PD, Wilson DB. Changes in serum enzyme levels following ketamine infusions. Anaesthesia 1980;35:12-6. Crossref
2. Wai MS, Chan WM, Zhang AQ, Wu Y, Yew DT. Longterm ketamine and ketamine plus alcohol treatments produced damages in liver and kidney. Hum Exp Toxicol 2012;31:877-86. Crossref
3. Jankovic SM, Jankovic SV, Stojadinovic D, Jakovljevic M, Milovanovic D. Effect of exogenous glutamate and N-Methyl-Daspartic acid on spontaneous activity of isolated human ureter. Int J Urol 2007;14:833-7. Crossref
4. Lo RS, Krishnamoorthy R, Freeman JG, Austin AS. Cholestasis and biliary dilatation associated with chronic ketamine abuse: a case series. Singapore Med J;52:e52-5.
5. Seto WK, Ng M, Chan P, et al. Ketamine-induced cholangiopathy: a case report. Am J Gastroenterol;106:1004-5. Crossref

