Hong Kong Med J 2016 Aug;22(4):382–92 | Epub 6 Jul 2016
DOI: 10.12809/hkmj154755
© Hong Kong Academy of Medicine. CC BY-NC-ND 4.0
REVIEW ARTICLE CME
Clinical applications of high-intensity focused ultrasound
WH She, MB, BS, FRCS1;
TT Cheung, MS, FRCS1;
Caroline R Jenkins, MB, BS, FRCA2;
Michael G Irwin, MD, FRCA2
1 Department of Surgery, The University of Hong Kong, Queen Mary Hospital, Pokfulam, Hong Kong
2 Department of Anaesthesiology, The University of Hong Kong, Queen Mary Hospital, Pokfulam, Hong Kong
Corresponding author: Dr TT Cheung (tantocheung@hotmail.com)
A video clip showing clinical applications of high-intensity focused ultrasound is available at www.hkmj.org
Abstract
Ultrasound has been developed for therapeutic
use in addition to its diagnostic ability. The use of
focused ultrasound energy can offer a non-invasive
method for tissue ablation, and can therefore be
used to treat various solid tumours. High-intensity
focused ultrasound is being increasingly used
in the treatment of both primary and metastatic
tumours as these can be precisely located for
ablation. It has been shown to be particularly useful
in the treatment of uterine fibroids, and various
solid tumours including those of the pancreas and
liver. High-intensity focused ultrasound is a valid
treatment option for liver tumours in patients with
significant medical co-morbidity who are at high risk
for surgery or who have relatively poor liver function
that may preclude hepatectomy. It has also been used
as a form of bridging therapy while patients awaiting
cadaveric donor liver transplantation. In this article,
we outline the principles of high-intensity focused
ultrasound and its clinical applications, including the
management protocol development in the treatment
of hepatocellular carcinoma in Hong Kong by
performing a search on MEDLINE (OVID), EMBASE,
and PubMed. The search of these databases ranged
from the date of their establishment until December
2015. The search terms used were: high-intensity
focused ultrasound, ultrasound, magnetic resonance
imaging, liver tumour, hepatocellular carcinoma,
pancreas, renal cell carcinoma, prostate cancer,
breast cancer, fibroids, bone tumour, atrial fibrillation,
glaucoma, Parkinson’s disease, essential tremor, and
neuropathic pain.
Introduction
High-intensity focused ultrasound (HIFU) was
initially used in the 1940s to treat brain pathologies
such as Parkinson’s disease.1 2 3 In the 1990s, it was
introduced to ophthalmology to treat raised intra-ocular
pressure, traumatic capsular tears, glaucoma,
retinal detachment, and vitreous haemorrhage.4 5 6 7 8 9
This technique has a unique ability to target deep-seated
soft tissue tumours. Furthermore, as long
as the lesions within solid organs can be clearly
visualised on magnetic resonance imaging (MRI)
or ultrasonography (USG)—that is, the presence of
the acoustic window to allow the transmission of
ultrasound energy—many lesions can be targeted such as
those in the liver, kidney, pancreas and breast; and
uterine fibroids, benign prostatic hypertrophy, and
prostate cancer. In recent years, HIFU has been used
to treat both benign and malignant lesions of various
solid organs. This non-invasive modality allows
treatment of tumours without surgery and offers a
new treatment option for those patients who are not
candidates for surgery, or who do not want surgery.
Methods
A search was performed on the following electronic
databases: MEDLINE (OVID), EMBASE, and
PubMed. The search of these databases ranged
from the date of their establishment until December
2015. The search terms used were: high-intensity
focused ultrasound, ultrasound, magnetic resonance
imaging, liver tumour, hepatocellular carcinoma,
pancreas, renal cell carcinoma, prostate cancer,
breast cancer, fibroids, bone tumour, atrial
fibrillation, glaucoma, Parkinson’s disease, essential
tremor, and neuropathic pain. Only studies reported
in English were included. Full papers were selected
if they contained facts, data, or scientific evidence
related to the treatment of HIFU. The reference lists
of articles selected were screened for full-text review.
Mechanism of high-intensity focused ultrasound
High-intensity focused ultrasound incorporates
multiple ultrasound beams produced by piezoelectric
or piezoceramic transducers directed into a three-dimensional
focal point of typically 1 to 5 mm in
diameter and 10 to 50 mm in length.10 Various
mechanisms have been proposed for the subsequent
tissue destruction with a synergistic effect from
thermal and mechanical means. This technique
induces heat generation due to absorption of the
acoustic energy with the temperature rising rapidly
to 60°C or higher, causing coagulation necrosis in
a short period of time. Focusing is an important
component as only a small volume (eg 1 mm diameter
and 9 mm length) is targeted by the ultrasound beam
and hence HIFU induces minimal thermal damage
to tissue located between the transducer and the
focal point.11
A mechanical effect is produced by acoustic
pulses only at higher intensities. Various phenomena
are observed, including cavitation, microstreaming,
and radiation forces. Cavitation is defined as the
creation or motion of a gas cavity in an acoustic
field due to alternating compression and expansion
of tissue as an ultrasound burst propagates through
it.12 There are two forms of cavitation to consider:
stable and inertial.11 If the tissue expansion or
rarefaction pressure is of sufficient magnitude, gas
can be extracted from the tissue, resulting in bubble
formation. In stable cavitation, the bubble is exposed
to a low-pressure acoustic field, resulting in stable
oscillation of the size of the bubble. In inertial
cavitation, exposure of the bubble to the acoustic
filed results in violent oscillations of the bubble and
rapid growth during the rarefaction phase, eventually
leading to the violent collapse and destruction of
the bubble. It will produce shock waves of very high
pressure (20-30 000 bars) and a high temperature
(2000-5000 K) in the microenvironment.13 14 Micro-streaming is a phenomenon produced by stable
cavitation in which rapid movement of fluid occurs
near the bubble due to its oscillating motion. It can
produce high shear forces that can cause transient
damage to cell membranes and may play a role in
ultrasound-enhanced drug or gene delivery when damage
to the cell membrane is transient.13 15
Radiation forces are developed when a wave
is either absorbed or reflected. If the reflecting or
absorbing medium is tissue or other solid material,
the force presses against the medium, producing a
pressure termed ‘radiation pressure’. If the medium is
liquid and can move under pressure, then streaming
results.16
The intention of HIFU is to raise and maintain
an isolated part of the volume above 60°C for
more than 1 second or longer, in order to cause
coagulative necrosis and immediate cell death.17 18 It aims to deliver the energy required to raise the tissue
temperature to a cytotoxic level sufficiently fast that
the tissue vasculature does not have a significant
effect on the extent of cell killing.19 20 In a study of the application of HIFU in the liver, 2 hours of
exposure resulted in a rim of glycogen-free cells of
about 10 cells wide. These cells were dead 48 hours later,
and showed signs of coagulative necrosis typical of
thermal injury.19
Limitations of high-intensity focused ultrasound
Of all the ablative modalities, HIFU has the advantage
that it does not require the introduction of an
applicator in order to achieve the ablative effect and
is the only non-invasive option. This makes it a very
attractive choice. It has several limitations, however.
This technique is essentially USG and, therefore, any
artefacts, such as acoustic shadowing, reverberation,
and refraction also apply to it. Superficial lesions
are treated most effectively by HIFU due to the
limitations of ultrasound penetrance through many
tissues, but the sound wave reflected carries very
high energy, and can also produce burns in tissues
that lie between the target and the transducer. Many
collateral injuries have been reported due to scattered
and reflected high-intensity ultrasound waves, such as skin
burns, peripheral nerve damage, and bowel injury.21 22
Great care also needs to be taken in areas that are
subject to respiratory movement, because of a lack
of precision, or the presence of sonic shadowing
due to overlying bony substances.21 In such
situations it may be necessary for the anaesthetist
to use controlled ventilation. The amount of energy
absorbed by the tissue may vary, as fibrotic, fatty, and
highly vascularised tissues attenuate sound energy
differently. Excessive energy absorption may result in
an unpredictable distribution of cell death.23 Careful
planning is therefore required to ensure adequate
tumour coverage, as coagulation, desiccation, and
vapour formation are detrimental to ultrasound energy
propagation, as well as precise localisation of the
lesion.24
The high-intensity focused ultrasound system
Both USG and MRI can be used to visualise, target,
and monitor the status of tissue destruction.
Ultrasonography
This is the most common method to target and
monitor the status of HIFU destruction.25 The
therapeutic and diagnostic transducers can be
packaged into one instrument that allows real-time
monitoring of the delivery of HIFU, the outcome
of the lesion, and the outcome of the peripheral
tissues. Although it is cost-effective, it has relatively
low spatial resolution that limits its accuracy for
targeting and it is also hard to visualise the details
of the structures in close proximity to the bowel
because of the gas-containing portions (air conducts
sound very poorly). In our unit, we use an USG-guided
HIFU system.26
Magnetic resonance imaging
This offers excellent resolution and tumour detail.
It locates the tumour boundary very clearly and is
particularly useful in patients in whom tumours
cannot be visualised with USG, for example, in obese
patients.27 Magnetic resonance imaging possesses
real-time thermal resolution with high spatial
resolution, and provides temperature data within
seconds of HIFU exposure. This allows detection of
small temperature elevations before any irreversible
tissue damage occurs.28 Nonetheless, MRI guidance
is expensive, labour intensive, noisy, and bulky.
Equipment such as that used for monitoring and
anaesthesia needs to be non-ferrous and MRI-safe
and treatment time is prolonged.
High-intensity focused ultrasound device
There are several devices available for the treatment
of various diseases, including extracorporeal,
transrectal, and interstitial devices.
Extracorporeal devices
Organs lying externally or those that are readily
accessible—such as breasts, cutaneous tissue,
limbs, abdomen, and brain—are usually treated with
extracorporeal HIFU that is guided by either USG or
MRI. As long as there is a suitable acoustic window
on the skin that allows uninterrupted propagation
of the HIFU energy beam to the target organs, one
can consider the use of extracorporeal HIFU for
treatment.
Ultrasonography-guided transcutaneous high-intensity focused ultrasound
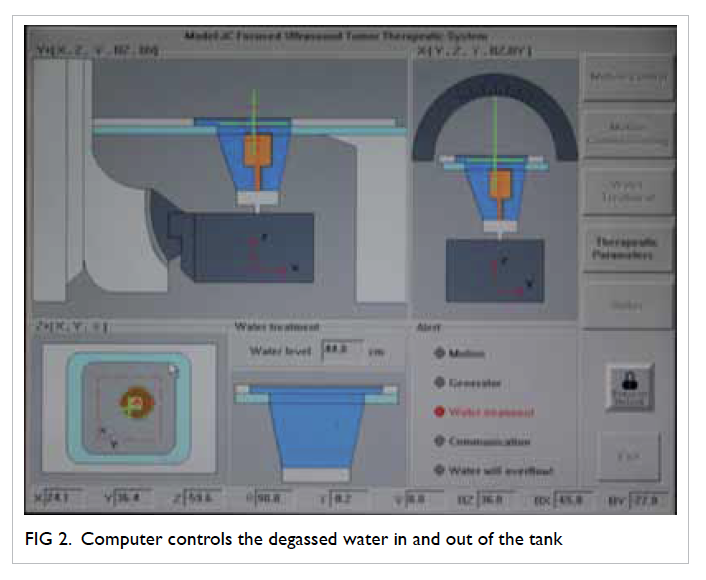
We are currently using a HIFU machine produced
by Haifu Technology Company (Chongqing, China)
[Figs 1, 2, 3]. It has been used and shown to be effective in treatment of a variety of benign and malignant
solid organ tumours, such as liver and pancreatic
cancer, uterine fibroids, soft tissue tumours, breast
cancer, and bladder cancer.29 30 31 This system consists
of three selectable therapeutic transducers and a
real-time imaging transducer. The transducers are
mounted in a water reservoir with the beam axis
directed upward, and the patient is positioned above
the transducers in a prone or decubitus position. The
HIFU exposure level is adjusted until a hyperechoic
region is seen on the USG image.

Figure 1. The usual setup of the high-intensity focused ultrasound (HIFU) system
(a) Control system; (b) HIFU machine and patient lying on the treatment table; and (c) anaesthetic machine
Magnetic resonance imaging–guided high-intensity focused ultrasound
Two major clinical MRI-HIFU systems are available
worldwide: InSightec (Tirat Carmel, Israel) and
Philips Healthcare (Vantaa, Finland).32 33 Their HIFU
transducers are similar in terms of enabling both
mechanical and electronic adjustment of HIFU focus
and MR thermometric temperature monitoring, but
their sonication strategies are different and hence
they differ in energy efficiency.34 These machines are
not available in Hong Kong.
Transrectal devices
Transrectal devices were developed for the treatment
of benign and malignant prostatic diseases. They aim
to ablate the entire prostate. Both USG-guided probes
and MRI-guided systems have been developed. The
USG probes are inserted per rectum and incorporate
both imaging and therapeutic transducers in one
unit,35 36 37 38 39 40 41 such as Ablatherm (Edap Technomed,
France) and the Sonablate (Focus Surgery Inc, US), whereas a prostate-dedicated MRI-HIFU system
makes use of either the transrectal (ExAblate OR;
InSightec) or transurethral (Philips Healthcare)
approach.42 43
Interstitial devices
Ultrasonographic transducers with different shapes
and sizes were developed in order to place the focused
applicators as close as possible to the target area.
Several shapes are available, including cylindrical,
semi-cylindrical, cylindrical with focusing by wave
reflection, plane and cylindrical array. Various
applicators have been developed to facilitate access
and guidance of the device, such as the flexible
applicator in an endoscopically placed HIFU
device for the treatment of cholangiocarcinoma44
or oesophageal tumours45; or rigid applicators for
a linear approach. Probes for percutaneous and
laparoscopic treatment are also being developed
and it is likely that the therapeutic indications will
increase.
Current clinical applications
High-intensity focused ultrasound has been used to
treat various benign and malignant solid tumours. It
is also used in conditions such as ablation for atrial
fibrillation,46 glaucoma,47 and benign obstetric and gynaecological procedures such as fibroids.32
Liver tumours
In general, liver resection is still the mainstay of
treatment of hepatocellular carcinoma (HCC),
provided the patient is surgically fit, has fair liver
function with good liver remnant and resectable
tumour. Liver transplantation is planned for patients
whose tumour is within the transplant criteria, and a
living or deceased donor is available.
Ablative therapy, such as radiofrequency
ablation (RFA), is considered for patients with a
relatively small tumour, preserved liver function,
and favourable location, that is, away from pleural
or gastrointestinal tract. For those patients whose
tumours are relatively small, located at the dome of
the liver, with clinical evidence of ascites or pleural
effusion, HIFU would be an alternative as long as
the lesion can be visualised and located by USG.
For those patients with multifocal tumours that are
not amenable to surgical resection or ablation, and
who have reasonable liver function without evidence
of ascites, transarterial chemoembolisation is the
treatment of choice. Sorafenib is an effective target
therapy for patients undergoing palliative care, but
has significant side-effects.
High-intensity focused ultrasound is now
one of the treatment modalities in our centre for
HCC and has been used as bridging therapy for
patients who are awaiting cadaveric donor liver
transplantation. This technique can be utilised for
patients who are not suitable for percutaneous RFA
but have a satisfactory general condition as assessed
by an anaesthesiologist. They should have intact skin
over the ablative region.
High-intensity focused ultrasound treatment procedure
Before treatment, the patient undergoes USG screening
to ensure that the targeted lesions are visible on
the USG localisation system. An anaesthesiologist
will assess the patient’s co-morbidities and suitability
for general anaesthesia as many patients may be
unfit for open surgery. Standard fasting and drug
administration guidelines apply. Before treatment
starts, the patient’s skin is cleansed with degassed
water and a negative-pressure aspirator is used to
degas the skin and reduce the dampening effect of
ultrasonic waves.
We use the JC HIFU system (Chongqing Haifu
Technology, Chongqing, China); HIFU ablation
is performed under general anaesthesia by a team
of surgeons and radiologists. Total intravenous
anaesthesia is favoured in our centre because of
its titratability, avoidance of nitrous oxide, and no
need for scavenging waste anaesthetic gases that
may be hazardous to the health of attendant staff.48
A dose of antibiotic (Augmentin 1.2 g; Beecham
Pharmaceuticals, Brentford, UK) is given just before
the procedure begins. Artificial pleural effusion
of 500 mL of normal saline is introduced if the liver
tumour is located at the dome, in order to facilitate
better ultrasound access to this region and protect
the lung. In addition, intermittent cessation of
respiratory movement by the anaesthesiologist
facilitates better localisation of the lesions during
energy transfer. For right-sided lesions, the patient
is placed in the right lateral position after tracheal
intubation. For left-sided lesions, the patient is
placed in the prone position. The JC HIFU system
consists of a treatment unit that delivers focused
ultrasound energy with a focal length of 12 cm deep. The
body is immersed in a degassed water circulation
unit that provides a medium for ultrasound transmission.
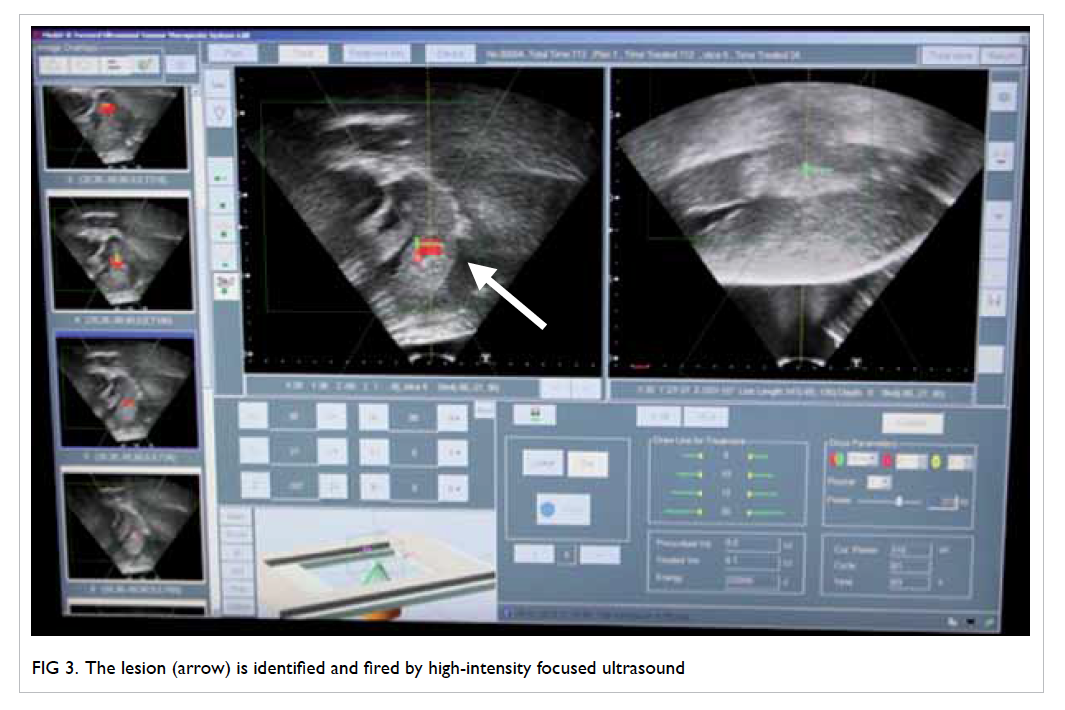
Grey-scale changes at the ablation site are observed
during the procedure, indicating the temperature
change inside the targeted lesion. Oral antibiotics
are given for 5 days after treatment.49
When ablating a large tumour, the ultrasound energy
is focused on the deep margin of the lesion first so
as to avoid prohibition of effective penetration of
energy by the cavitation effect and the presence
of coagulation necrosis. Meticulous planning of
the focus point before the procedure, in which
the ablation sequence is from the deepest layer to
the most superficial layer, is required for maximal
destruction of the targeted lesion. Intermittent
cessations of the procedure allow recovery of the
cavitation effect shown under USG monitoring,
giving additional allowance for ablation of the
residual lesion in the periphery.50
Treatment results
Although a minimally invasive approach can
be employed in patients with HCC and liver
cirrhosis, hepatectomy is contra-indicated in
patients with decompensated cirrhosis.51 Our pilot
studies suggested that HIFU is relatively safe and
effective.52 53 Patients who have poor liver function can still be offered HIFU.54 Its effectiveness in
treating small HCC of size <3 cm was proven to be
comparable with percutaneous RFA.55 Furthermore,
its application in recurrent HCC allows patients to
undergo ablation, especially when the abdomen is
hostile due to previous surgery, or there is inadequate
liver remnant due to previous major hepatectomy.56
In the treatment of HCC in non-surgical candidates,
1- and 3-year overall survival rates of 87.7% and
62.4%, respectively were achieved in 49 patients with
a median tumour size of 2.2 cm.49 In addition, our
prospective study suggested that the response rate
for those patients with HCC who underwent HIFU
as the bridging treatment while awaiting cadaveric
liver transplantation was higher than in those who
underwent transarterial chemoembolisation.52 It is
particularly useful in patients who also have poor
liver function with clinical ascites, as ascites itself is
a good acoustic media for HIFU.57
High-intensity focused ultrasound does carry
certain risks in the treatment for HCC. Minor
complications such as skin and subcutaneous
tissue injury occur in most patients.54 At our centre, there has been a case of post-HIFU bile duct stricture requiring endoscopic retrograde cholangiography.
Pancreatic cancer
Inoperable locally advanced pancreatic cancer
remains difficult to treat. Local ablative therapy with
HIFU has been used in patients with unresectable
pancreatic cancer and proven to be safe in both
clinical trials and retrospective studies,31 58 59 60 with
no damage to the exocrine or endocrine function.61 62 63
It has been used as a form of palliative treatment in
some pilot studies with a median survival ranging
from 10 to 12.6 months, either alone or combined
with chemotherapy.62 63 64 65 Pain relief was also found
to be effective.31 58 59 63 65 66 Unfortunately, treatment
after HIFU usually lacked histomorphological
examination. The survival benefit needs to be tested
in further studies, and preferably confirmed by
randomised controlled trials.
Urological applications
Prostate cancer
Transrectal HIFU is advocated as a form of minimally
invasive treatment for localised prostate cancer.
It is suggested primarily for patients with low- to
intermediate-risk prostate cancer, according to
D’Amico Risk stratification.67 68 69 70 71 72 It has also been used
to treat locally recurrent prostate cancer. Patients
with unifocal and multifocal prostate cancer were
subjected to HIFU and had no evidence of disease on
MRI at 12 months.73 Good functional outcome was
achieved after the treatment, such as continence and
good erectile function. Nonetheless, complications
such as acute retention of urine or more severe rectal
wall injury can occur. More sophisticated MRI-guided
HIFU will allow more precise localisation
of such lesions.74 To date, most studies have been in
the form of retrospective studies or case series only
with no randomised controlled trial of HIFU for the
treatment of prostate cancer.
Renal tumours
International consensus panels recommend ablative
techniques in patients who are unfit for surgery,
who are not considered candidates for or elect
against active surveillance, and who have a small
renal mass.75 76 European Association of Urology
guidelines recommend the use of an ablative
method only in tumours of less than 4 cm.76 In fact, HIFU
has been investigated in the treatment of both
primary and metastatic renal tumours.77 78 79 Results
suggest that there were discrete zones of ablation
in 67% of patients in the final histology and HIFU
achieved stable lesions in two thirds of patients
with minimal morbidity; 90% of patients had good
pain control immediately after HIFU.79 There were
several limitations, however, such as the degree of
subcutaneous and perinephric fat and the position
of the tumour in relation to the ribs.80 Higher
acoustic output is needed to compensate for the
energy loss due to the thickness of the perinephric
fat that might in turn increase the risk of prefocal
and surrounding tissue damage.81 Currently, there
is no controlled study to suggest the superiority of
HIFU over various ablative techniques, such as RFA
or cryoablation.
Brain diseases
High-intensity focused ultrasound was first used in
the 1950s to treat Parkinson’s disease.82 83 84 It required
access through the skull to the brain and, therefore,
craniotomy was necessary. It subsequently became
unpopular due to the concurrent development of
the drug levodopa. With the advancement of MRI
guidance, there has been a resurgence of interest
as a non-invasive treatment for essential tremor,
neuropathic pain, and Parkinson’s disease. It is
safe, without major risk of infection or bleeding,
but may result in transient oedema. In Parkinson’s
disease, as the disease progresses, patients will
eventually require levodopa that is associated with
tolerance and, eventually, development of levodopa-resistant
symptoms with movement fluctuations and
dyskinesias. At this time surgical intervention may
be considered. High-intensity focused ultrasound
can allow ablation of the fibres that join the thalamus
with the globus pallidus. Results of a pilot study
suggested that there was improvement in terms of the
functional score as rated by the Unified Parkinson’s
Disease Rating Scale.85
Essential tremor is a common neurological
condition usually managed conservatively, or with
propranolol and primidone. For those treatment-resistant
patients, surgical intervention may be
considered. Usually RFA, stereotactic radiosurgery,
gamma knife thalamotomy, or deep brain stimulation
are used to either cause tissue destruction or to block
abnormal nerve signals. High-intensity focused
ultrasound has been used in a clinical trial context
with promising results and marked reduction (>80%)
of tremor.86 87
Neuropathic pain is a complex condition
often associated with damage to or dysfunction of
the nerve fibres that then send incorrect signals to
the pain centres with minimal stimulation. High-intensity
focused ultrasound can be directed to the
part of the central lateral thalamic nucleus of those
patients suffering from chronic therapy-resistant
neuropathic pain. Significant pain relief has been
observed with long-term follow-up in a pilot study.88
Breast tumour
High-intensity focused ultrasound is an ideal breast-conserving
therapy because it does not significantly
change the patients’ breast shape and does not cause
bleeding or scarring. It does not require general
anaesthesia, and hence has a reduced recovery
time. Both USG- and MRI-guided HIFU ablations
have been used. The aim is to achieve complete
tumour necrosis but results have been inconsistent
with some showing complete necrosis,89 and others
residual tumour of less than 10% and residual tumour
between 10% and 90%.90 91 92 93 94 A negative margin is the
most important basis and factor for local control
of the breast cancer.95 96 Nonetheless, it is difficult
to ensure a negative margin after HIFU therapy
with the aid of imaging alone, hence adjuvant
radiotherapy has been suggested. Currently, there is
limited prospective study or randomised trial in this
area. Most work has been pilot studies or feasibility
studies only.
Complications include pain, skin burns,
oedema, pectoralis major muscle injury,97 and rib
pain.98 These are relatively minor compared with
those following traditional breast surgery with its
attendant potential complications of wound pain,
infection, bleeding, and impaired wound healing.
Fibroids
Uterine fibroid is a common benign gynaecological
condition in women of childbearing age. Patients
usually suffer symptoms such as heavy, painful,
and prolonged menstrual bleeding, mass effect
with urinary urgency, and constipation.99
Conservative medical therapy with non-steroidal
anti-inflammatory drugs, contraceptive steroids,
and gonadotropin-releasing hormone agonists are
the first-line treatment. Ablative treatment as
well as surgery will be necessary for those in whom
conservative management fails or in those with
progressive symptoms. Treatment is by means of
myomectomy or hysterectomy. In cases where the
patient does not want surgery, or where the patient
is planning a future pregnancy, HIFU is a good
option.100 101 Extracorporeal HIFU enables ablation of various sizes and shape of fibroid. Symptoms are
reduced by more than 50% in terms of pain,102 103 104
bulk-related and menstrual symptoms, comparable
with the results of conventional surgery.105 After
HIFU, the ablated fibroid volume is decreased, and
is related to the non-perfused volume of the tumour
immediately after treatment.44 106 107 Nonetheless,
HIFU cannot propagate through air-filled viscera
such as bowel. There is a potential risk of bowel
perforation if it lies close to the fibroids.108 Most of
the articles were from China, as HIFU has been used
in China for treatment of fibroids. In our hospital,
the following criteria are used for patient selection:
(1) premenopausal women with no plans for further
childbearing; (2) severe fibroid symptoms (as defined
by a transformed symptom severity score of >41 on
the Uterine Fibroid Symptom and Quality of Life
questionnaire); (3) a clinical uterine size of less than
20 weeks’ gestation, a dominant fibroid of less than
10 cm in diameter without areas of necrosis as judged
by contrast-enhanced MRI, a non-pedunculated
fibroid, and a fibroid not suspicious of malignancy;
(4) no evidence of known or suspected extensive
pelvic adhesions such as history of acute pelvic
inflammatory disease, severe pelvic endometriosis,
or lower abdominal surgery; and (5) an abdominal
wall thickness of less than 5 cm.109
Bone tumour
Bone metastases
In palliative treatment of bone tumours, therapeutic
goals include pain palliation, tumour reduction,
prevention of impending pathological fractures, and/or tumour decompression. Opioid analgesics and
radiation therapy are widely used for pain control in
patients suffering from bone metastases but this does
not always provide desired relief in many patients
and is associated with undesirable side-effects.110 111 112 113 114 115
Research reveals that MRI-guided HIFU is safe
and effective in the treatment of painful bone
tumours.116 117 118 Periosteal denervation and tumour
debulking may play a significant role in symptom
relief.74 116 117 Response to HIFU is rapid and good
pain control has been seen within days of treatment.
This greatly improves the quality of life for many
patients with disseminated cancer.119 There is also
evidence of a reduction in lesion viability after HIFU
and a remineralisation of spongious bone.120
Tumour ablation in curative treatment aims for
complete coagulation necrosis of the primary lesion.
Primary bone malignancy such as osteosarcoma
has been treated with HIFU. A combination of
chemotherapy with HIFU seems to be as effective
as limb-sparing surgery and chemotherapy for
malignant bone tumours.121 This is potentially useful
for patients who are not fit for surgery. Treatment
complications include skin burns, procedure-related
pain, and post-treatment fractures.121 122 Most of the
studies were clinical trials. More studies should focus
on the treatment outcome in terms of the function,
quality of life, and survival.
Glaucoma
The specific advantage of HIFU is that the energy
can be focused through non-optically transparent
media without uncontrolled energy absorption, thus
reducing the effects on adjacent tissues. It allows a
defined and adjustable tissue volume to be heated
and treated at any depth or location within the eye.
Intra-ocular pressure is decreased both by reducing
aqueous humour production (aqueous inflow) and
by facilitating the evacuation of aqueous humour
from the eye (aqueous outflow).123 Prospective case
series suggest there is a significant reduction in
intra-ocular pressure without significant peri- or
post-treatment side-effects.124
Atrial fibrillation
High-intensity focused ultrasound has been used
to treat atrial fibrillation in cardiac surgery. This is
designed to deliver pulmonary vein and posterior
left atrial wall isolation on the beating heart using
an encircling ‘cinch’ and create left atrial lines using
a handheld wand device. It then ablates areas around
the ganglionic plexi where dense collections of
complex fractionated atrial electrograms are found.
It has been proven to be safe and effective.125 Patients
can be reverted to sinus rhythm and the results
are more pronounced in patients with paroxysmal
atrial fibrillation.46 126 Selected use of this technique
has been suggested.127 If symptoms persist, other
modalities should be considered.125
Conclusions
High-intensity focused ultrasound has many
applications in both benign and malignant diseases.
It offers an alternative to those patients for whom
surgery is contra-indicated or inappropriate. The
results of HIFU in the management of HCC patients
in our centre are particularly promising. Further
studies of the application of HIFU in various organs
should be conducted for both clinical trials as well
as comparative studies with other ablative modalities
in the form of randomised controlled trials.
Declaration
No funding was received for the study or its
publication. None of the authors has any conflict of
interest with regard to the study or its publication.
References
1. Lynn JG, Zwemer RL, Chick AJ, Miller AE. A new method
for the generation and use of focused ultrasound in
experimental biology. J Gen Physiol 1942;26:179-93. Crossref
2. Fry WJ, Fry FJ. Fundamental neurological research and
human neurosurgery using intense ultrasound. IRE Trans
Med Electron 1960;ME-7:166-81. Crossref
3. Lynn JG, Putnam TJ. Histology of cerebral lesions produced
by focused ultrasound. Am J Pathol 1944;20:637-49.
4. Coleman DJ, Lizzi FL, el-Mofty AA, Driller J, Franzen
LA. Ultrasonically accelerated resorption of vitreous
membranes. Am J Ophthalmol 1980;89:490-9. Crossref
5. Coleman DJ, Lizzi FL, Torpey JH, et al. Treatment of
experimental lens capsular tears with intense focused
ultrasound. Br J Ophthalmol 1985;69:645-9. Crossref
6. Coleman DJ, Lizzi FL, Driller J, et al. Therapeutic
ultrasound in the treatment of glaucoma. II. Clinical
applications. Ophthalmology 1985;92:347-53. Crossref
7. Rosenberg RS, Purnell EW. Effects of ultrasonic radiation
to the ciliary body. Am J Ophthalmol 1967;63:403-9. Crossref
8. Silverman RH, Vogelsang B, Rondeau MJ, Coleman DJ.
Therapeutic ultrasound for the treatment of glaucoma. Am
J Ophthalmol 1991;111:327-37. Crossref
9. Rosecan LR, Iwamoto T, Rosado A, Lizzi FL, Coleman
DJ. Therapeutic ultrasound in the treatment of retinal
detachment: clinical observations and light and electron
microscopy. Retina 1985;5:115-22. Crossref
10. Mearini L. High intensity focused ultrasound, liver disease
and bridging therapy. World J Gastroenterol 2013;19:7494-9. Crossref
11. Dubinsky TJ, Cuevas C, Dighe MK, Kolokythas O, Hwang
JH. High-intensity focused ultrasound: current potential
and oncologic applications. AJR Am J Roentgenol
2008;190:191-9. Crossref
12. Yang R, Reilly CR, Rescorla FJ, et al. High-intensity focused
ultrasound in the treatment of experimental liver cancer.
Arch Surg 1991;126:1002-9; discussion 1009-10. Crossref
13. Holland CK, Apfel RE. Thresholds for transient cavitation
produced by pulsed ultrasound in a controlled nuclei
environment. J Acoust Soc Am 1990;88:2059-69. Crossref
14. Marmottant P, Hilgenfeldt S. Controlled vesicle
deformation and lysis by single oscillating bubbles. Nature
2003;423:153-6. Crossref
15. Pitt WG, Husseini GA, Staples BJ. Ultrasonic drug
delivery—a general review. Expert Opin Drug Deliv
2004;1:37-56. Crossref
16. Vaezy S, Shi X, Martin RW, et al. Real-time visualization
of high-intensity focused ultrasound treatment using
ultrasound imaging. Ultrasound Med Biol 2001;27:33-42. Crossref
17. Sapareto SA, Dewey WC. Thermal dose determination in
cancer therapy. Int J Radiat Oncol Biol Phys 1984;10:787-800. Crossref
18. Dewhirst MW, Viglianti BL, Lora-Michiels M, Hanson
M, Hoopes PJ. Basic principles of thermal dosimetry and
thermal thresholds for tissue damage from hyperthermia.
Int J Hyperthermia 2003;19:267-94. Crossref
19. ter Haar GR, Robertson D. Tissue destruction with focused
ultrasound in vivo. Eur Urol 1993;23 Suppl 1:8-11.
20. Wu F, Chen WZ, Bai J, et al. Pathological changes in human
malignant carcinoma treated with high-intensity focused
ultrasound. Ultrasound Med Biol 2001;27:1099-106. Crossref
21. Kim YS, Rhim H, Choi MJ, Lim HK, Choi D. High-intensity
focused ultrasound therapy: an overview for radiologists.
Korean J Radiol 2008;9:291-302. Crossref
22. Li JJ, Xu GL, Gu MF, et al. Complications of high intensity
focused ultrasound in patients with recurrent and
metastatic abdominal tumors. World J Gastroenterol
2007;13:2747-51. Crossref
23. Sibille A, Prat F, Chapelon JY, et al. Extracorporeal ablation
of liver tissue by high-intensity focused ultrasound.
Oncology 1993;50:375-9. Crossref
24. Roberts WW, Hall TL, Ives K, Wolf JS Jr, Fowlkes JB,
Cain CA. Pulsed cavitational ultrasound: a noninvasive
technology for controlled tissue ablation (histotripsy) in
the rabbit kidney. J Urol 2006;175:734-8. Crossref
25. ter Haar G. Ultrasound focal beam surgery. Ultrasound
Med Biol 1995;21:1089-100. Crossref
26. Hynynen K, Freund WR, Cline HE, et al. A clinical,
noninvasive, MR imaging–monitored ultrasound surgery
method. Radiographics 1996;16:185-95. Crossref
27. Yagel S. High-intensity focused ultrasound: a revolution
in non-invasive ultrasound treatment? Ultrasound Obstet
Gynecol 2004;23:216-7. Crossref
28. Hynynen K, Pomeroy O, Smith DN, et al. MR imaging–guided
focused ultrasound surgery of fibroadenomas in the
breast: a feasibility study. Radiology 2001;219:176-85. Crossref
29. Dong X, Yang Z. High-intensity focused ultrasound
ablation of uterine localized adenomyosis. Curr Opin
Obstet Gynecol 2010;22:326-30. Crossref
30. Jung SE, Cho SH, Jang JH, Han JY. High-intensity focused
ultrasound ablation in hepatic and pancreatic cancer:
complications. Abdom Imaging 2011;36:185-95. Crossref
31. Orsi F, Zhang L, Arnone P, et al. High-intensity focused
ultrasound ablation: effective and safe therapy for solid
tumors in difficult locations. AJR Am J Roentgenol
2010;195:W245-52. Crossref
32. Tempany CM, Stewart EA, McDannold N, Quade BJ,
Jolesz FA, Hynynen K. MR imaging–guided focused
ultrasound surgery of uterine leiomyomas: a feasibility
study. Radiology 2003;226:897-905. Crossref
33. Kim YS, Keserci B, Partanen A, et al. Volumetric MR-HIFU
ablation of uterine fibroids: role of treatment cell size
in the improvement of energy efficiency. Eur J Radiol
2012;81:3652-9. Crossref
34. Kim YS. Advances in MR image–guided high-intensity
focused ultrasound therapy. Int J Hyperthermia
2015;31:225-32. Crossref
35. Sanghvi NT, Foster RS, Bihrle R, et al. Noninvasive surgery
of prostate tissue by high intensity focused ultrasound: an
updated report. Eur J Ultrasound 1999;9:19-29. Crossref
36. Beerlage HP, Thüroff S, Debruyne FM, Chaussy C, de la
Rosette JJ. Transrectal high-intensity focused ultrasound
using the Ablatherm device in the treatment of localized
prostate carcinoma. Urology 1999;54:273-7. Crossref
37. Andreou C, Blana A, Orovan W, Hassouna M, Warner
J, Woods E. Technical review: high-intensity focused
ultrasound for prostate cancer. Can J Urol 2005;12:2684-5;
discussion 2686.
38. Christopher T. HIFU focusing efficiency and a twin annular
array source for prostate treatment. IEEE Trans Ultrason
Ferroelectr Freq Control 2005;52:1523-33. Crossref
39. Curiel L, Chavrier F, Souchon R, Birer A, Chapelon JY.
1.5-D high intensity focused ultrasound array for non-invasive
prostate cancer surgery. IEEE Trans Ultrason
Ferroelectr Freq Control 2002;49:231-42. Crossref
40. Rebillard X, Gelet A, Davin JL, et al. Transrectal high-intensity
focused ultrasound in the treatment of localized
prostate cancer. J Endourol 2005;19:693-701. Crossref
41. Saleh KY, Smith NB. A 63 element 1.75 dimensional
ultrasound phased array for the treatment of benign
prostatic hyperplasia. Biomed Eng Online 2005;4:39. Crossref
42. Lindner U, Ghai S, Spensieri P, et al. Focal magnetic
resonance guided focused ultrasound for prostate cancer:
Initial North American experience. Can Urol Assoc J
2012;6:E283-6.
43. Siddiqui K, Chopra R, Vedula S, et al. MRI-guided
transurethral ultrasound therapy of the prostate gland
using real-time thermal mapping: initial studies. Urology
2010;76:1506-11. Crossref
44. Lafon C, Chapelon JY, Prat F, et al. Design and preliminary
results of an ultrasound applicator for interstitial thermal
coagulation. Ultrasound Med Biol 1998;24:113-22. Crossref
45. Melodelima D, Salomir R, Chapelon JY, Theillère Y,
Moonen C, Cathignol D. Intraluminal high intensity
ultrasound treatment in the esophagus under fast MR
temperature mapping: in vivo studies. Magn Reson Med
2005;54:975-82. Crossref
46. Reyes G, Ruyra X, Valderrama F, et al. High intensity
focused ultrasound ablation for atrial fibrillation: results
from the National Spanish Registry. Minerva Cardioangiol
2015 May 26. Epub ahead of print.
47. Melamed S, Goldenfeld M, Cotlear D, Skaat A, Moroz I.
High-intensity focused ultrasound treatment in refractory
glaucoma patients: results at 1 year of prospective clinical
study. Eur J Ophthalmol 2015;25:483-9. Crossref
48. Yao CL, Trinh T, Wong GT, Irwin MG. Anaesthesia for high
intensity focused ultrasound (HIFU) therapy. Anaesthesia
2008;63:865-72. Crossref
49. Ng KK, Poon RT, Chan SC, et al. High-intensity focused
ultrasound for hepatocellular carcinoma: a single-center
experience. Ann Surg 2011;253:981-7. Crossref
50. Cheung TT, Poon RT, Jenkins CR, et al. Survival analysis of
high-intensity focused ultrasound therapy vs. transarterial
chemoembolization for unresectable hepatocellular
carcinomas. Liver Int 2014;34:e136-43. Crossref
51. Cheung TT, Poon RT, Yuen WK, et al. Long-term survival
analysis of pure laparoscopic versus open hepatectomy
for hepatocellular carcinoma in patients with cirrhosis: a
single-center experience. Ann Surg 2013;257:506-11. Crossref
52. Cheung TT, Fan ST, Chan SC, et al. High-intensity focused
ultrasound ablation: an effective bridging therapy for
hepatocellular carcinoma patients. World J Gastroenterol
2013;19:3083-9. Crossref
53. Wu F, Wang ZB, Chen WZ, et al. Advanced hepatocellular
carcinoma: treatment with high-intensity focused
ultrasound ablation combined with transcatheter arterial
embolization. Radiology 2005;235:659-67. Crossref
54. Cheung TT, Chu FS, Jenkins CR, et al. Tolerance of high-intensity
focused ultrasound ablation in patients with
hepatocellular carcinoma. World J Surg 2012;36:2420-7. Crossref
55. Cheung TT, Fan ST, Chu FS, et al. Survival analysis of high-intensity focused ultrasound ablation in patients with small
hepatocellular carcinoma. HPB (Oxford) 2013;15:567-73. Crossref
56. Chan AC, Cheung TT, Fan ST, et al. Survival analysis
of high-intensity focused ultrasound therapy versus
radiofrequency ablation in the treatment of recurrent
hepatocellular carcinoma. Ann Surg 2013;257:686-92. Crossref
57. Chok KS, Cheung TT, Lo RC, et al. Pilot study of high-intensity
focused ultrasound ablation as a bridging therapy
for hepatocellular carcinoma patients wait-listed for liver
transplantation. Liver Transpl 2014;20:912-21. Crossref
58. Orgera G, Krokidis M, Monfardini L, et al. High intensity
focused ultrasound ablation of pancreatic neuroendocrine
tumours: report of two cases. Cardiovasc Intervent Radiol
2011;34:419-23. Crossref
59. Wang K, Zhu H, Meng Z, et al. Safety evaluation of high-intensity
focused ultrasound in patients with pancreatic
cancer. Onkologie 2013;36:88-92. Crossref
60. Sung HY, Jung SE, Cho SH, et al. Long-term outcome of
high-intensity focused ultrasound in advanced pancreatic
cancer. Pancreas 2011;40:1080-6. Crossref
61. Shi Y, Ying X, Hu X, et al. Influence of high intensity
focused ultrasound (HIFU) treatment to the pancreatic
function in pancreatic cancer patients. Pak J Pharm Sci
2015;28 Suppl:1097-100.
62. Zhao H, Yang G, Wang D, et al. Concurrent gemcitabine
and high-intensity focused ultrasound therapy in patients
with locally advanced pancreatic cancer. Anticancer Drugs
2010;21:447-52. Crossref
63. Gao HF, Wang K, Meng ZQ, et al. High intensity focused
ultrasound treatment for patients with local advanced
pancreatic cancer. Hepatogastroenterology 2013;60:1906-10.
64. Xiong LL, Hwang JH, Huang XB, et al. Early clinical
experience using high intensity focused ultrasound
for palliation of inoperable pancreatic cancer. JOP
2009;10:123-9.
65. Wang K, Chen Z, Meng Z, et al. Analgesic effect of high
intensity focused ultrasound therapy for unresectable
pancreatic cancer. Int J Hyperthermia 2011;27:101-7. Crossref
66. Wu F, Wang ZB, Zhu H, et al. Feasibility of US-guided high-intensity
focused ultrasound treatment in patients with
advanced pancreatic cancer: initial experience. Radiology
2005;236:1034-40. Crossref
67. Uchida T, Shoji S, Nakano M, et al. Transrectal high-intensity
focused ultrasound for the treatment of
localized prostate cancer: eight-year experience. Int J Urol
2009;16:881-6. Crossref
68. Blana A, Walter B, Rogenhofer S, Wieland WF. High-intensity
focused ultrasound for the treatment of localized
prostate cancer: 5-year experience. Urology 2004;63:297-300. Crossref
69. Ripert T, Azémar MD, Ménard J, et al. Six years’ experience with high-intensity focused ultrasonography for prostate
cancer: oncological outcomes using the new ‘Stuttgart’
definition for biochemical failure. BJU Int 2011;107:1899-905. Crossref
70. Crouzet S, Chapelon JY, Rouvière O, et al. Whole-gland
ablation of localized prostate cancer with high-intensity
focused ultrasound: oncologic outcomes and morbidity in
1002 patients. Eur Urol 2014;65:907-14. Crossref
71. Mearini L, D’Urso L, Collura D, Nunzi E, Muto G, Porena
M. High-intensity focused ultrasound for the treatment of
prostate cancer: A prospective trial with long-term follow-up.
Scand J Urol 2015;49:267-74. Crossref
72. Heidenreich A, Bastian PJ, Bellmunt J, et al. EAU guidelines
on prostate cancer. part 1: screening, diagnosis, and local
treatment with curative intent—update 2013. Eur Urol
2014;65:124-37. Crossref
73. Ahmed HU, Hindley RG, Dickinson L, et al. Focal therapy
for localised unifocal and multifocal prostate cancer: a
prospective development study. Lancet Oncol 2012;13:622-32. Crossref
74. Napoli A, Anzidei M, De Nunzio C, et al. Real-time
magnetic resonance–guided high-intensity focused
ultrasound focal therapy for localised prostate cancer:
preliminary experience. Eur Urol 2013;63:395-8. Crossref
75. Van Poppel H, Becker F, Cadeddu JA, et al. Treatment of
localised renal cell carcinoma. Eur Urol 2011;60:662-72. Crossref
76. Ljungberg B, Bensalah K, Canfield S, et al. EAU guidelines
on renal cell carcinoma: 2014 update. Eur Urol 2015;67:913-24. Crossref
77. Illing RO, Kennedy JE, Wu F, et al. The safety and feasibility
of extracorporeal high-intensity focused ultrasound
(HIFU) for the treatment of liver and kidney tumours in a
Western population. Br J Cancer 2005;93:890-5. Crossref
78. Ritchie RW, Leslie T, Phillips R, et al. Extracorporeal high
intensity focused ultrasound for renal tumours: a 3-year
follow-up. BJU Int 2010;106:1004-9. Crossref
79. Wu F, Wang ZB, Chen WZ, Bai J, Zhu H, Qiao TY.
Preliminary experience using high intensity focused
ultrasound for the treatment of patients with advanced
stage renal malignancy. J Urol 2003;170(6 Pt 1):2237-40. Crossref
80. Kohrmann KU, Michel MS, Gaa J, Marlinghaus E, Alken P.
High intensity focused ultrasound as noninvasive therapy
for multilocal renal cell carcinoma: case study and review
of the literature. J Urol 2002;167:2397-403. Crossref
81. Ritchie R, Collin J, Coussios C, Leslie T. Attenuation and
de-focusing during high-intensity focused ultrasound
therapy through peri-nephric fat. Ultrasound Med Biol
2013;39:1785-93. Crossref
82. Fry WJ, Mosberg WH Jr, Barnard JW, Fry FJ. Production of
focal destructive lesions in the central nervous system with
ultrasound. J Neurosurg 1954;11:471-8. Crossref
83. Fry WJ, Barnard JW, Fry EJ, Krumins RF, Brennan JF.
Ultrasonic lesions in the mammalian central nervous
system. Science 1955;122:517-8. Crossref
84. Fry FJ. Precision high intensity focusing ultrasonic
machines for surgery. Am J Phys Med 1958;37:152-6. Crossref
85. Jeanmonod D, Moser D, Magara A, et al. Study of
incisionless transcranial magnetic resonance–guided
focused ultrasound treatment of Parkinson’s disease:
Safety, accuracy and initial clinical outcomes. Proceedings
of the Current and Future Applications of Focused
Ultrasound 2012, 3rd International Symposium; 2012 Oct
14-17; Washington, DC Metro Area, United States.
86. Elias WJ, Huss D, Voss T, et al. A pilot study of focused
ultrasound thalamotomy for essential tremor. N Engl J
Med 2013;369:640-8. Crossref
87. Lipsman N, Schwartz ML, Huang Y, et al. MR-guided
focused ultrasound thalamotomy for essential tremor: a
proof-of-concept study. Lancet Neurol 2013;12:462-8. Crossref
88. Jeanmonod D, Werner B, Morel A, et al. Transcranial
magnetic resonance imaging–guided focused ultrasound:
noninvasive central lateral thalamotomy for chronic
neuropathic pain. Neurosurg Focus 2012;32:E1. Crossref
89. Wu F, Wang ZB, Cao YD, et al. Changes in biologic
characteristics of breast cancer treated with high-intensity
focused ultrasound. Ultrasound Med Biol 2003;29:1487-92. Crossref
90. Furusawa H, Namba K, Thomsen S, et al. Magnetic
resonance–guided focused ultrasound surgery of breast
cancer: reliability and effectiveness. J Am Coll Surg
2006;203:54-63. Crossref
91. Gianfelice D, Khiat A, Amara M, Belblidia A, Boulanger Y.
MR imaging–guided focused US ablation of breast cancer:
histopathologic assessment of effectiveness—initial
experience. Radiology 2003;227:849-55. Crossref
92. Gianfelice D, Khiat A, Amara M, Belblidia A, Boulanger Y.
MR imaging–guided focused ultrasound surgery of breast
cancer: correlation of dynamic contrast-enhanced MRI
with histopathologic findings. Breast Cancer Res Treat
2003;82:93-101. Crossref
93. Zippel DB, Papa MZ. The use of MR imaging guided
focused ultrasound in breast cancer patients; a preliminary
phase one study and review. Breast Cancer 2005;12:32-8. Crossref
94. Khiat A, Gianfelice D, Amara M, Boulanger Y. Influence
of post-treatment delay on the evaluation of the response
to focused ultrasound surgery of breast cancer by dynamic
contrast enhanced MRI. Br J Radiol 2006;79:308-14. Crossref
95. Poggi MM, Danforth DN, Sciuto LC, et al. Eighteen-year
results in the treatment of early breast carcinoma
with mastectomy versus breast conservation therapy:
the National Cancer Institute randomized trial. Cancer
2003;98:697-702. Crossref
96. van Dongen JA, Bartelink H, Fentiman IS, et al. Factors
influencing local relapse and survival and results of salvage
treatment after breast-conserving therapy in operable
breast cancer: EORTC trial 10801, breast conservation
compared with mastectomy in TNM stage I and II breast
cancer. Eur J Cancer 1992;28A(4-5):801-5. Crossref
97. Myers MR. Transient temperature rise due to ultrasound
absorption at a bone/soft-tissue interface. J Acoust Soc Am
2004;115:2887-91. Crossref
98. Zderic V, Foley J, Luo W, Vaezy S. Prevention of post-focal
thermal damage by formation of bubbles at the focus
during high intensity focused ultrasound therapy. Med
Phys 2008;35:4292-9. Crossref
99. Vollenhoven BJ, Lawrence AS, Healy DL. Uterine fibroids:
a clinical review. Br J Obstet Gynaecol 1990;97:285-98. Crossref
100. Wu F, Wang ZB, Chen WZ, et al. Extracorporeal focused
ultrasound surgery for treatment of human solid
carcinomas: early Chinese clinical experience. Ultrasound
Med Biol 2004;30:245-60. Crossref
101. Smart OC, Hindley JT, Regan L, Gedroyc WM. Magnetic
resonance guided focused ultrasound surgery of uterine
fibroids—the tissue effects of GnRH agonist pre-treatment.
Eur J Radiol 2006;59:163-7. Crossref
102. Yoon SW, Kim KA, Cha SH, et al. Successful use of
magnetic resonance–guided focused ultrasound surgery
to relieve symptoms in a patient with symptomatic focal
adenomyosis. Fertil Steril 2008;90:2018.e13-5. Crossref
103. Fennessy FM, Tempany CM, McDannold NJ, et al. Uterine
leiomyomas: MR imaging–guided focused ultrasound
surgery—results of different treatment protocols.
Radiology 2007;243:885-93. Crossref
104. Lénárd ZM, McDannold NJ, Fennessy FM, et al. Uterine leiomyomas: MR imaging–guided focused ultrasound
surgery—imaging predictors of success. Radiology
2008;249:187-94. Crossref
105. Mikami K, Murakami T, Okada A, Osuga K, Tomoda
K, Nakamura H. Magnetic resonance imaging–guided
focused ultrasound ablation of uterine fibroids: early
clinical experience. Radiat Med 2008;26:198-205. Crossref
106. LeBlang SD, Hoctor K, Steinberg FL. Leiomyoma shrinkage
after MRI-guided focused ultrasound treatment: report of
80 patients. AJR Am J Roentgenol 2010;194:274-80. Crossref
107. Zhang L, Chen WZ, Liu YJ, et al. Feasibility of magnetic
resonance imaging–guided high intensity focused
ultrasound therapy for ablating uterine fibroids in
patients with bowel lies anterior to uterus. Eur J Radiol
2010;73:396-403. Crossref
108. Kennedy JE, Ter Haar GR, Cranston D. High intensity
focused ultrasound: surgery of the future? Br J Radiol
2003;76:590-9. Crossref
109. Cheung VY. Sonographically guided high-intensity
focused ultrasound for the management of uterine
fibroids. J Ultrasound Med 2013;32:1353-8. Crossref
110. Chow E, Harris K, Fan G, Tsao M, Sze WM. Palliative
radiotherapy trials for bone metastases: a systematic
review. J Clin Oncol 2007;25:1423-36. Crossref
111. Hartsell WF, Scott CB, Bruner DW, et al. Randomized trial
of short- versus long-course radiotherapy for palliation of
painful bone metastases. J Natl Cancer Inst 2005;97:798-804. Crossref
112. Cleeland CS. The measurement of pain from metastatic
bone disease: capturing the patient’s experience. Clin
Cancer Res 2006;12(20 Pt 2):6236s-42s. Crossref
113. van der Linden YM, Lok JJ, Steenland E, et al. Single
fraction radiotherapy is efficacious: a further analysis
of the Dutch Bone Metastasis Study controlling for the
influence of retreatment. Int J Radiat Oncol Biol Phys
2004;59:528-37. Crossref
114. Mundy GR. Metastasis to bone: causes, consequences and
therapeutic opportunities. Nat Rev Cancer 2002;2:584-93. Crossref
115. Saarto T, Janes R, Tenhunen M, Kouri M. Palliative
radiotherapy in the treatment of skeletal metastases. Eur J
Pain 2002;6:323-30. Crossref
116. Catane R, Beck A, Inbar Y, et al. MR-guided focused
ultrasound surgery (MRgFUS) for the palliation of pain
in patients with bone metastases—preliminary clinical
experience. Ann Oncol 2007;18:163-7. Crossref
117. Gianfelice D, Gupta C, Kucharczyk W, Bret P, Havill
D, Clemons M. Palliative treatment of painful bone
metastases with MR imaging–guided focused ultrasound.
Radiology 2008;249:355-63. Crossref
118. Liberman B, Gianfelice D, Inbar Y, et al. Pain palliation in
patients with bone metastases using MR-guided focused
ultrasound surgery: a multicenter study. Ann Surg Oncol
2009;16:140-6. Crossref
119. Zeng L, Chow E, Bedard G, et al. Quality of life after
palliative radiation therapy for patients with painful bone
metastases: results of an international study validating
the EORTC QLQ-BM22. Int J Radiat Oncol Biol Phys
2012;84:e337-42. Crossref
120. Napoli A, Anzidei M, Marincola BC, et al. Primary pain
palliation and local tumor control in bone metastases
treated with magnetic resonance–guided focused
ultrasound. Invest Radiol 2013;48:351-8. Crossref
121. Chen W, Zhu H, Zhang L, et al. Primary bone malignancy:
effective treatment with high-intensity focused ultrasound
ablation. Radiology 2010;255:967-78. Crossref
122. Hurwitz MD, Ghanouni P, Kanaev SV, et al. Magnetic
resonance–guided focused ultrasound for patients with
painful bone metastases: phase III trial results. J Natl
Cancer Inst 2014;106:dju082. Crossref
123. Aptel F, Begle A, Razavi A, et al. Short- and long-term
effects on the ciliary body and the aqueous
outflow pathways of high-intensity focused ultrasound
cyclocoagulation. Ultrasound Med Biol 2014;40:2096-106. Crossref
124. Aptel F, Dupuy C, Rouland JF. Treatment of refractory
open-angle glaucoma using ultrasonic circular
cyclocoagulation: a prospective case series. Curr Med Res
Opin 2014;30:1599-605. Crossref
125. Davies EJ, Bazerbashi S, Asopa S, Haywood G, Dalrymple-Hay M. Long-term outcomes following high intensity
focused ultrasound ablation for atrial fibrillation. J Card
Surg 2014;29:101-7. Crossref
126. Garcia R, Sacher F, Oses P, et al. Electrophysiological study
6 months after Epicor high-intensity focused ultrasound
atrial fibrillation ablation. J Interv Card Electrophysiol
2014;41:245-51. Crossref
127. Colli A, Romero-Ferrer B. It is time to revisit the theory of
acute conduction block: efficacy of high-intensity focused
ultrasound epicardial ablation. Eur J Cardiothorac Surg
2013;43:451-2. Crossref