Hong Kong Med J 2018 Jun;24(3):270–6 | Epub 25 May 2018
DOI: 10.12809/hkmj176990
© Hong Kong Academy of Medicine. CC BY-NC-ND 4.0
ORIGINAL ARTICLE
Clinical and biochemical characteristics of infants
with prolonged neonatal jaundice
Sylvia LY Siu, MB, ChB, FHKAM (Paediatrics)1;
Lilian WM Chan, MB, BS, FHKAM (Paediatrics)2; Albert NS Kwong,
MB, BS, FHKAM (Paediatrics)1
1 Department of Paediatrics and
Adolescent Medicine, Tuen Mun Hospital, Tuen Mun, Hong Kong
2 Ha Kwai Chung Child Assessment Centre,
Kwai Chung, Hong Kong
Corresponding author: Dr Sylvia LY Siu (siulys@ha.org.hk)
Abstract
Introduction: Protocols for
investigating neonatal prolonged jaundice vary and the yield from
screening has not been assessed. International guidelines recommend
establishing cholestasis before proceeding to investigate the underlying
pathology. However, in most hospitals administered by the Hospital
Authority, full liver function is checked at the first neonatal jaundice
clinic visit. To study the diagnostic yield of this approach, we carried
out a retrospective study of all infants referred for prolonged
jaundice.
Methods: Attendance records from
the neonatal jaundice clinic at the Tuen Mun Hospital, Hong Kong, the
clinical management system, and electronic patient records were used to
retrieve epidemiological, clinical, and laboratory data, and patients’
clinical progress.
Results: During the 8-month
study period from 8 July 2015 to 8 March 2016, 1164 infants were
referred to the neonatal jaundice clinic for prolonged jaundice. Among
them, 16 (1.4%) had conjugated hyperbilirubinaemia. Diagnoses included
biliary atresia (n=1), cytomegalovirus (CMV) infection (n=3), neonatal
hepatitis syndrome (n=2), and transient cholestasis (n=10). In total, 98
(8.4%) infants had elevated alanine transaminase levels. Diagnoses
included biliary atresia (n=1), hepatic congestion related to congestive
heart failure (n=1), CMV infection (n=5), neonatal hepatitis syndrome
(n=16), and non-specific elevated alanine transaminase (n=75). In total,
59 infants had elevated alkaline phosphatase levels.
Conclusions: A stepwise approach
is recommended, in which full liver function is checked and the
underlying cause of jaundice is investigated only after confirming
cholestasis.
New knowledge added by this study
- Among healthy infants with physiological or breastmilk jaundice, a transitional stage of cholestasis may occur when the jaundice is resolving. The lower conjugated bilirubin level and the downtrend of total bilirubin help to differentiate these infants from those with pathological cholestasis.
- Breastfed infants usually have mild elevation of alanine transaminase which may reflect higher metabolism instead of pathology. Therefore, checking alanine transaminase levels at the first neonatal jaundice clinic visit is not recommended because of the potential detrimental effect on breastfeeding rates.
- Late preterm infants with prolonged jaundice are at risk for osteopenia of prematurity.
- A stepwise approach is recommended, checking full liver function and investigating the underlying cause only after confirming cholestasis.
- Bone profile blood test (alkaline phosphatase, albumin, calcium, and phosphate) is recommended for late preterm or low birth weight infants to screen for osteopenia of prematurity at the first neonatal jaundice clinic visit.
Introduction
All newborns have elevated unconjugated bilirubin
concentrations relative to normal adult values. Two thirds or more of
breastfed infants have unconjugated hyperbilirubinaemia that extends into
the second and third weeks of life, and often up to age 8 to 12 weeks.1 Guidelines on the evaluation of cholestasis in infants
recommend establishing cholestasis before proceeding to investigate the
underlying pathology.2 3 However, of the hospitals administered by the Hong Kong
Hospital Authority that care for newborns, most check full liver function
at the first Neonatal Jaundice Clinic (NNJC) visit.
The Food and Health Bureau established the
Committee on Promotion of Breastfeeding in Hong Kong in 2014. Since then,
there has been a substantial improvement in exclusive breastfeeding rates
and a continuous upward trend in ever-breastfeeding rates.4
The Tuen Mun Hospital is a regional hospital in the
New Territories West region of Hong Kong. An outpatient Neonatal Jaundice
Clinic is available every Monday to Friday from 14:00 to 17:00 to treat
infants with jaundice. The increasing breastfeeding rates have led to
increasing numbers of infants with prolonged jaundice being referred to
the NNJC.
The present study aimed to learn about the clinical
and biochemical characteristics of prolonged neonatal jaundice, and to
study the diagnostic yield of a full liver function check at first NNJC
visit. We reviewed the clinical and laboratory records of infants visiting
the NNJC between 8 July 2015 and 8 March 2016. On the basis of the
findings, we propose a more effective evaluation procedure for prolonged
neonatal jaundice.
Methods
Attendance records maintained by the NNJC included
the registration number, sex, and attendance date of the patients. Using
the recorded patient registration numbers, gestational age, birth weight,
glucose-6-phosphate dehydrogenase (G6PD) deficiency status, mode of
feeding, phototherapy history, liver function test results, and clinical
progress were retrieved from the clinical management system and electronic
patient record. The study period was chosen for convenience. All infants
who visited the NNJC during the study period were included to prevent
possible selection bias. All suspicious data were verified by revisiting
the electronic patient record to ensure data accuracy.
For gestational age, completed weeks of gestation
were recorded. Preterm births were categorised as very preterm (before 34
weeks of gestation) or late preterm (between 34 weeks and 36 weeks 6 days
of gestation). Full-term births were those at 37 weeks of gestation or
later. Birth weights were categorised as very low (<1500 g), low
(1500-2499 g), normal (2500-3999 g), or high (>3999 g). Modes of
feeding were categorised as exclusive breastfeeding, mixed feeding, or
exclusive formula feeding.
Data analysis
The SPSS for Windows version 15.0 (SPSS Inc,
Chicago [IL], United States) was used for all statistical analyses.
Student’s t test and univariate analysis of variance (ANOVA) were
used in comparing the outcomes between groups. Tukey’s test was used for
pairwise comparisons in ANOVA. Cross-tabulation was used to measure
associations between binary outcome variables and binary predictor
variables.
Results
Demographic characteristics
In total, 1164 infants (663 males, 501 females;
male-to-female ratio=1.3:1) with prolonged jaundice were referred to the
NNJC during the 8-month study period. The gestational ages of the infants
ranged from 29 to 41 weeks, and there were eight (0.69%) very preterm
infants and 94 (8.08%) late preterm infants. The birth weights of the
infants ranged from 1425 g to 4670 g. The sample included only one (0.09%)
very low birth weight infant, in addition to 80 (6.87%) low birth weight
infants and 13 (1.12%) high birth weight infants. In total, 34 (5.13%)
male infants and two (0.40%) female infants had G6PD deficiency. The mode
of feeding was exclusive breastfeeding in 648 (55.70%) infants, mixed
feeding in 400 (34.36%) infants, and exclusive formula feeding in 114
(9.79%) infants; the mode of feeding was not recorded in two infants.
Among the 114 exclusively formula fed infants, 24 (21.10%) had a history
of breastfeeding. At the first NNJC visit, 70 (6.01%) infants were 2 weeks
old, 156 (13.40%) 3 weeks old, 758 (65.12%) 4 weeks old, 165 (14.18%) 5
weeks old, 10 (0.86%) 6 weeks old, and three (0.26%) 7 weeks old. One
(0.09%) infant first visited at 8.7 weeks old and another (0.09%) first
visited at 11.4 weeks old.
Full liver function test
At the first NNJC visit, 1139 (97.90%) infants
received a full liver function test, which included taking measurements of
alkaline phosphatase (ALP), calcium, and phosphate. In seven infants, low
transcutaneous bilirubinometer readings were observed (peak reading, 42-86
μmol/L). Therefore, blood tests were not performed in these infants. At
the first NNJC visit, 17 infants were seen by a doctor who adopted the
stepwise approach and who screened only for cholestasis; one infant
visiting on day 14 had only total bilirubin (TB) checked.
Influence of breastfeeding on liver biochemistry
There was a significant effect of mode of feeding
on TB (F2,1155=18.058; P<0.01). The mean (M) TB
levels in the exclusive breastfeeding group (M=146.73) were significantly
higher than those in the mixed feeding group (M=131.05) and in the
exclusive formula feeding group (M=121.21). Tukey’s test showed
significant differences in TB levels between the exclusive breastfeeding
and the mixed feeding groups (P<0.01) and between the exclusive
breastfeeding and the exclusive formula feeding groups (P<0.01). There
was no significant difference in TB level between the mixed feeding and
exclusive formula feeding groups.
There was a significant effect of feeding on
alanine transaminase (ALT) levels (F2,1133=15.015;
P<0.01). The ALT levels in the exclusive breastfeeding group (M=21.79)
were significantly higher than those in the mixed feeding group (M=18.97)
and in the exclusive formula feeding group (M=16.55). Tukey’s test showed
significant differences in ALT levels between the exclusive breastfeeding
and the mixed feeding groups (P<0.01) and between the exclusive
breastfeeding and the exclusive formula feeding groups (P<0.01). There
was no significant difference in ALT levels between the mixed feeding and
exclusive formula feeding groups.
There was a significant effect of feeding on ALP (F2,1135=6.276;
P<0.01). The ALP levels in the exclusive breastfeeding group (M=348.83)
were significantly higher than those in the mixed feeding group (M=329.08)
and in the exclusive formula feeding group (M=327.76). Tukey’s test showed
a significant difference in ALP level between the exclusive breastfeeding
and the mixed feeding groups (P<0.01). There was no significant
difference in ALP levels between the exclusive breastfeeding and the
exclusive formula feeding groups (P=0.07) or between the mixed feeding and
the exclusive formula feeding groups (P=0.99).
There was a significant effect for infants who had
received phototherapy (t(1141)=3.57; P<0.01). Infants who had
received phototherapy had higher TB levels (M=147.12) than those who had
not (M=135.06).
There was no significant difference in TB levels
between male and female infants. There was also no statistically
significant difference in TB levels between G6PD-deficient and
G6PD-sufficient infants.
Cholestasis
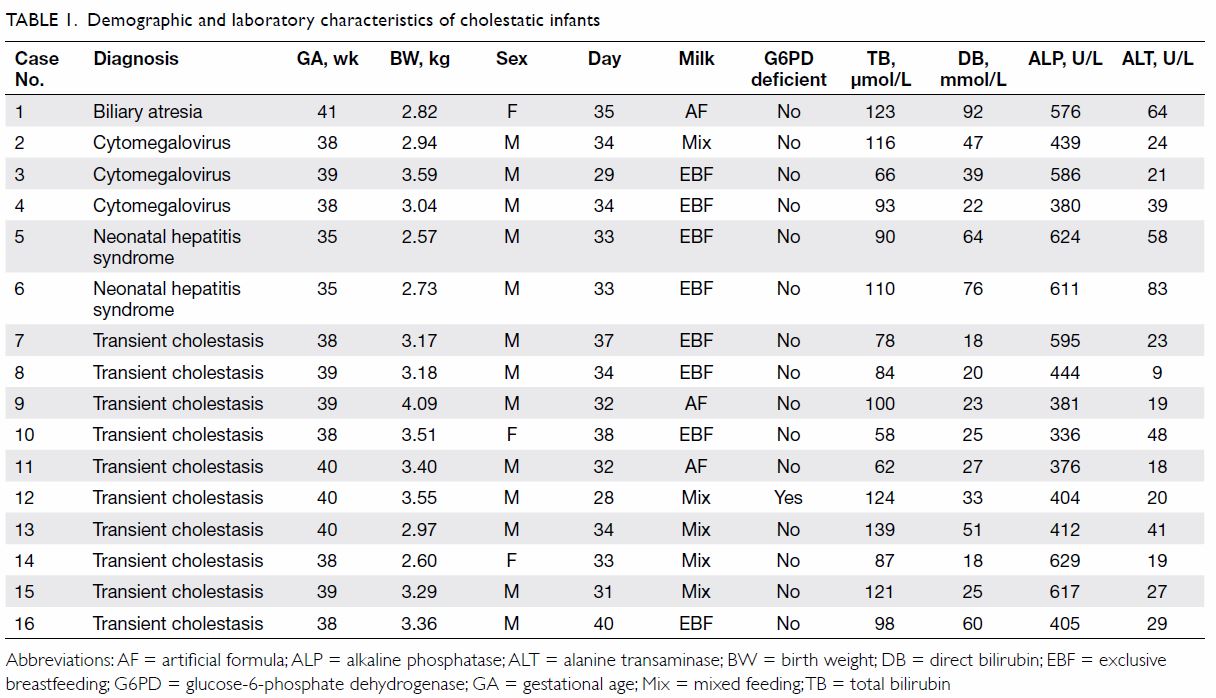
At the first NNJC visit, 16 (1.4%) infants had
conjugated hyperbilirubinaemia. Diagnoses included biliary atresia in one,
cytomegalovirus (CMV) infection in three, neonatal hepatitis syndrome in
two, and transient cholestasis in 10 infants (Table 1). All infants were thriving well and did not
have dark urine or pale stool at their first NNJC visit.
Elevated alanine transaminase level
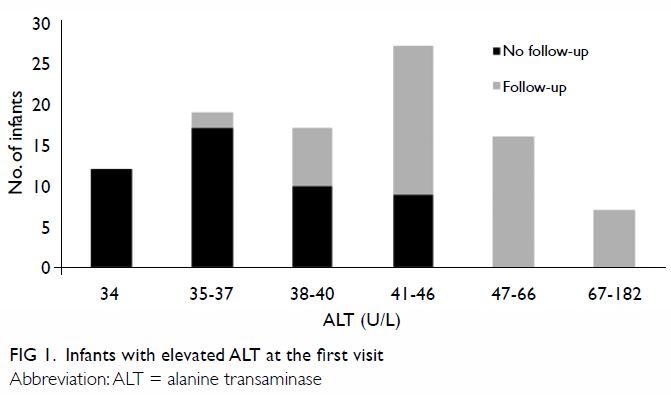
At the first NNJC visit, 98 (8.4%) infants had
elevated ALT levels. The reference range used for ALT level is 5 U/L to 33
U/L. The proportion of infants that were followed up increased with
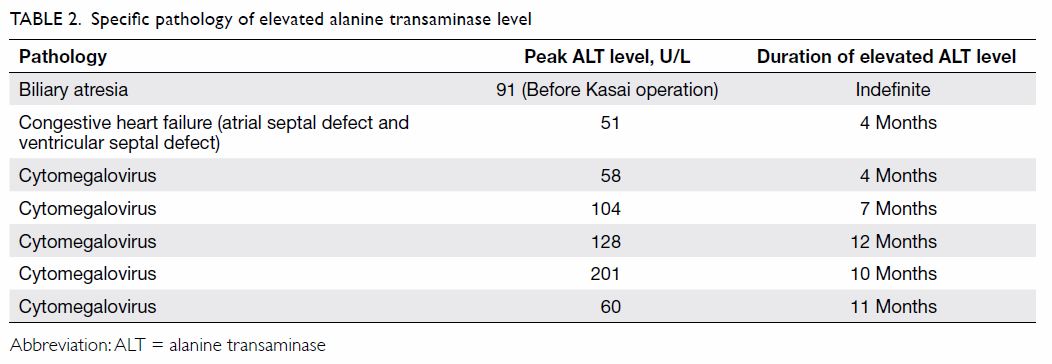
increasing ALT level, as shown in Figure 1. Specific causes for elevated ALT level
included biliary atresia in one infant, hepatic congestion related to
congestive heart failure in one infant, and CMV in five infants (Table
2). Of the remaining 91 infants with elevated ALT levels, 16 had
neonatal hepatitis syndrome and 75 had non-specific elevated ALT levels.
Urine tests revealed CMV infection in five infants
with elevated ALT levels; CMV infection was believed to be acquired
postnatally. These infants were all full-term births of normal birth
weight and were asymptomatic. Of these five infants, four were exclusively
breastfed and one was fed a mix of breastmilk and formula. After their
elevated ALT levels resolved, three infants were discharged from the NNJC
and the other two were followed up for coincidental findings
(developmental concern in one and familial small head in the other) and
not for concern over CMV infection. These five infants and the 75 infants
with non-specific elevated ALT were all healthy and asymptomatic and had
good weight gain; their elevated ALT levels resolved without treatment.
In this study, inflammation of the liver occurring
in early infancy that could not be attributed to a specific cause of liver
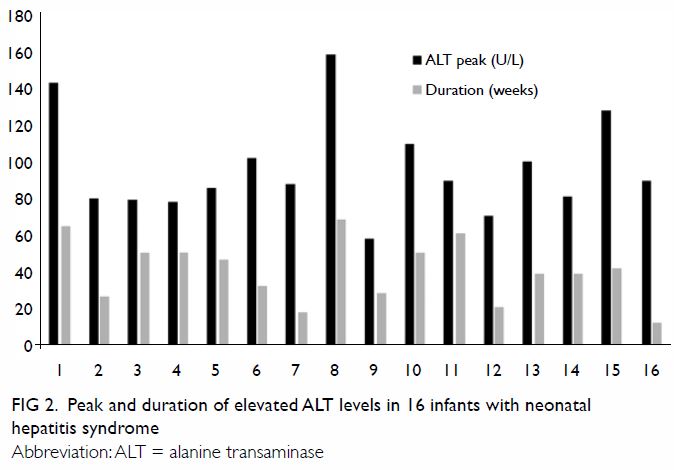
disease was termed neonatal hepatitis syndrome. The peak ALT levels and
the duration of elevated ALT levels in 16 infants with neonatal hepatitis
syndrome are shown in Figure 2. The duration of elevated ALT level ranged
from 13 to 69 weeks and was shorter than 28 weeks in only four infants.
Infants with neonatal hepatitis syndrome were followed up until ALT levels
returned to normal and then for an average of 1 month longer.
In 75 infants who were otherwise healthy,
non-specific elevated ALT levels were within double the usual upper limit.
These infants were either not followed up or their liver function was
monitored periodically (time intervals in months determined on
case-by-case basis) with limited diagnostic testing.
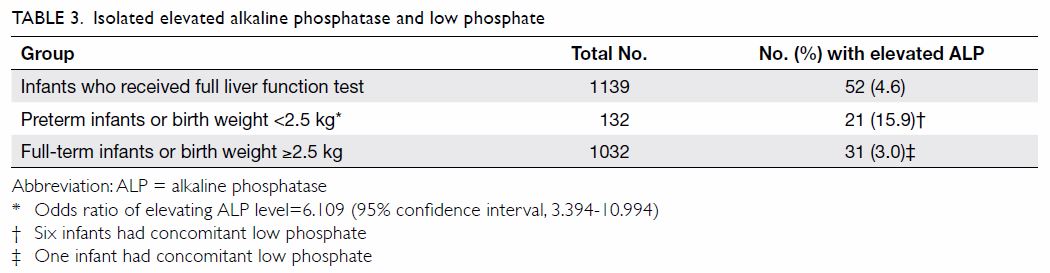
Elevated alkaline phosphatase level and low phosphate
level
The mode of feeding, gestational age, and birth
weight were all found to affect ALP levels. Among 132 preterm or low birth
weight infants, 21 (15.9%) had elevated ALP, compared with 31 (3.0%) among
1032 full-term and normal birth weight infants. For preterm or low birth
weight infants versus term infants with birth weight >2499 g, the odds
ratio for elevated ALP was 6.109 (95% confidence interval=3.394-10.994) [Table 3]. Among 1139 infants who underwent full
liver function tests, 10 had low phosphate levels: seven had concomitant
high ALP and low phosphate levels, and the remaining two late preterm
infants and one full-term infant had isolated low phosphate levels.
Discussion
We found that TB levels were significantly higher
in the exclusive breastfeeding and mixed feeding groups than in the
exclusive formula feeding group. Eight infants that were exposed to
breastmilk and two exclusively formula-fed infants had transient
cholestasis. Our findings also revealed that ALT levels were significantly
higher in the exclusive breastfeeding and mixed feeding groups than in the
exclusive formula feeding group. Besides, preterm or low birth weight
infants had increased odds of high ALP levels compared with term infants
with birth weight >2499 g.
Cholestasis
Breast milk jaundice (BMJ) was first described by
Newman and Gross in 1963.5
Subsequently, BMJ has been reported to be associated with increased
conjugated and unconjugated bilirubin levels.6
In 1991, investigators in Japan studied 58 breastfed infants with indirect
hyperbilirubinaemia and found that 18 (31%) with BMJ had elevated ALP,
gamma-glutamyltranspeptidase or serum bile acid, reflecting alterations in
the hepatobiliary system.7 The
serum bile acid levels in patients with BMJ are similar to those with
cholestatic jaundice caused by diseases such as extrahepatic biliary
atresia.7 This finding suggests
that BMJ may be caused by hepatic dysfunction related to cholestasis.
Moreover, for infants with BMJ and increased fasting serum bile acid
levels, discontinuation of breastfeeding did not cause a rapid
normalisation of the serum TB levels.7
This observation suggests that hyperbilirubinaemia in infants with
increased serum bile acid levels is not directly related to breastfeeding.
Our finding of transient cholestasis in 1.75% (2/114) of exclusive formula
feeding infants versus 0.76% (8/1048) exclusive breast milk feeding and
mixed feeding infants supported the hypothesis that cholestasis is not
related to breastfeeding.
In this study, we used the term ‘transient
cholestasis’ to describe the delay in the decline of conjugated bilirubin
levels, as observed in 10 infants. Because TB levels declined first
without a concomitant decline or even with an increase in conjugated
bilirubin levels, there seemed to be a transitional stage of cholestasis.
When TB levels decreased further, the conjugated bilirubin levels then
decreased. Moreover, the conjugated bilirubin levels in these 10 infants
were lower than those with biliary atresia or hepatitis. This finding
supports the use of direct bilirubin as a surrogate marker in assessing
the severity of cholestasis to ensure optimal timing of hepatobiliary
scanning.8
Elevated alanine transaminase level
In 1981, Landaas et al9
first reported a significant difference in ALT levels between breastfed
and formula-fed infants. They proposed a normal range for ALT level of 14
to 84 IU/L until 4.5 months of age. In 1984, Gómez et al10 examined 2099 out-patient children and found that ALT
levels (40-97 IU/L, 3rd to 97th percentile) were higher in children <1
year than in older children. In 2003, investigators in Denmark found
higher mean serum bilirubin, albumin, and aspartate transaminase (AST)
levels in healthy exclusively breastfed full-term infants; they also found
a strong positive association between AST and insulin-like growth factor-1
levels at 2 months (r=0.47; P=0.004).11
Protein levels in breast milk are lower than those in infant formulas.
Serum albumin levels have been used to evaluate the adequacy of protein
levels in infant formula. Thus, the finding of higher serum albumin levels
in breastfed infants than formula feeding infants suggests that there were
no protein deficiencies or abnormalities affecting albumin production in
breastfed infants.11 Insulin-like
growth factor-1 is an anabolic hormone in infants; thus, those authors
believed that the most likely explanation for the elevated AST values
among breastfed infants is a stimulation of liver metabolism through one
of several growth factors in human milk.11
Therefore, the higher AST levels in breastfed infants were believed to be
a reflection of a higher liver metabolism, rather than a reflection of
liver cell damage. Alanine transaminase is present primarily in the liver
and thus is a more specific marker of hepatocellular cell injury.
Aspartate transaminase is present in the liver and other organs, a less
specific marker of hepatocellular function. The aforementioned study used
AST in a restrictive sense to reflect liver biochemistry. Therefore, the
higher ALT levels among breastfed infants should logically be interpreted
by the same token as reflection of higher liver metabolism rather than
liver cell injury.
In the present study, other than those in which CMV
was identified, findings were negative. Elevated ALT concentrations
resolved in all infants other than the infant with biliary atresia. These
findings support the hypothesis that elevated ALT in breastfed infants is
a reflection of higher metabolism rather than of any pathology.
Isolated elevated alkaline phosphatase level and low
phosphate level
Serum ALP is derived predominately from the liver
and bones. In the present study, elevated ALP in seven infants was of
hepatic origin. Tests of ALP’s heat stability index revealed that 52
infants with isolated ALP elevation were of bone origin.
In this study, 21 (15.9%) preterm or low birth
weight infants had isolated elevated ALP (Table 3). Among the eight very preterm infants, only
one had isolated elevated ALP and none had low phosphate. Among 94 late
preterm infants, 20 (21.3%) had isolated elevated ALP and among these, six
(30%) also had low phosphate.
The biochemical characteristics of high ALP and low
phosphate are compatible with osteopenia of prematurity. During pregnancy,
calcium and phosphorus are actively transferred from the mother to the
fetus, reaching a peak accretion rate at 32 to 36 weeks of gestation. The
third trimester is the period of most active growth and the increased
accretion rate is in response to the higher fetal needs for the developing
skeleton. As a result, in preterm infants, calcium and phosphate
requirements increase with decreasing gestational age, to compensate for
the loss of accretion of these minerals. In the present study, very
preterm infants were managed in neonatal wards for long durations;
therefore, this need for increased mineral supplements was recognised and
addressed. However, late preterm infants may not be clearly distinguished
from full-term infants, and their need for additional minerals may not be
apparent or addressed during their short hospital stay.
Diagnostic yield of full liver function test at first
neonatal jaundice clinic visit
Breastfed infants have been reported to acquire CMV
via breastmilk.12 13 Cytomegalovirus excreted in breastmilk is likely
caused by reactivated infection in the presence of maternal antibody
transferred transplacentally.12
This type of milk-borne CMV infection apparently protects children from
CMV diseases and the seropositivity for CMV may protect the next
generation from CMV inclusion disease.
Poddighe et al14
reported a full-term breastfed infant with prolonged jaundice who had
undergone extensive tests but with negative findings. Liver function test
results returned to normal by age 7 months when breastmilk intake was
significantly reduced. The authors14
then proposed that, in otherwise healthy infants and in the absence of
risk factors, elevated ALT levels should be monitored for 7 months before
performing further sophisticated tests. If this proposal were applied to
the present study, only four infants had elevated ALT for less than 7
months and would have avoided further tests.
Proposed prolonged neonatal jaundice evaluation
The above discussion suggests that measuring ALT
levels at the first NNJC visit is of limited benefit. For jaundiced
infants, repeated follow-up examinations for elevated ALT levels may
increase the risk of premature cessation of breastfeeding and of
development of vulnerable child syndrome.15
Therefore, we propose measuring ALT levels only after noting cholestasis.
In the present study, late preterm infants were
identified to be at high risk of having ALP elevation. Preterm or low
birth weight infants accounted for <10% of those visiting the NNJC. In
addition to cholestasis screening at the first NNJC visit, ALP, albumin,
calcium, and phosphate should be checked for late preterm or low birth
weight infants.
Vitamin D deficiency has been reported in 18% of
Hong Kong women.16 In this study,
the prevalence of elevated ALP was 4.6%. The recently published global
consensus on prevention and management of nutritional rickets recommends
vitamin D supplementation at 400 IU daily in all infants, independent of
their mode of feeding in the first year of life.17
Therefore, the best way to protect infants may be to educate pregnant
mothers to take vitamin D supplements during pregnancy and to give 400 IU
daily vitamin D supplements to their infants.17
Limitations
Information bias and selection bias are two
potential limitations of our study. First, for simplicity, all infants
taking just one mouthful of breastmilk and those taking just one mouthful
of formula were classified in the mixed feeding group. In the mixed
feeding group, this information bias may mask the effect of breastfeeding
on liver biochemistry. Second, the study period was chosen for convenience
and not at random. This may create selection bias because outbreaks of
diseases in infants tend to create clusters of clinic visits within a
certain timeframe.
Conclusions
For full-term and normal or high birth weight
infants, the most effective way to manage prolonged neonatal jaundice is
to screen for cholestasis before full liver function examination. For late
preterm or low birth weight infants, the most effective way to manage
prolonged neonatal jaundice is to screen for cholestasis and to check bone
profile (ALP, albumin, calcium, and phosphate) at the first NNJC visit.
Author contributions
All authors have made substantial contributions to
the concept or design of this study; acquisition of data; analysis or
interpretation of data; drafting of the article; and critical revision for
important intellectual content.
Acknowledgement
The authors thank Ms CK Ho and Mr WF Wu for
maintaining the neonatal jaundice clinic attendance records that made this
retrospective study possible.
Funding/support
This research received no specific grant from any
funding agency in the public, commercial, or not-for-profit sectors.
Declaration
All authors have disclosed no conflicts of
interest. All authors had full access to the data, contributed to the
study, approved the final version for publication, and take responsibility
for its accuracy and integrity. This paper was presented orally in Joint
Annual Research & Scientific Meeting 2017, 19 August 2017, Hong Kong.
Ethical approval
Ethical approval for the study was obtained and
patient/parental consent was waived by the New Territories West Cluster
Clinical and Research Ethics Committee.
References
1. Academy of Breastfeeding Medicine
Protocol Committee. ABM clinical protocol #22: guidelines for management
of jaundice in the breastfeeding infant equal to or greater than 35 weeks’
gestation. Breastfeed Med 2010;5:87-93. Crossref
2. Fawaz R, Baumann U, Ekong U, et al.
Guideline for the evaluation of cholestatic jaundice in infants: joint
recommendations of the North American Society for Pediatric
Gastroenterology, Hepatology, and Nutrition and the European Society for
Pediatric Gastroenterology, Hepatology, and Nutrition. J Pediatr
Gastroenterol Nutr 2017;64:154-68. Crossref
3. Moyer V, Freese DK, Whitington PF, et
al. Guideline for the evaluation of cholestatic jaundice in infants:
recommendations of the North American Society for Pediatric
Gastroenterology, Hepatology and Nutrition. J Pediatr Gastroenterol Nutr
2004;39:115-28. Crossref
4. Family Health Service, Department of
Health, HKSAR Government. Breastfeeding Survey 2017.
5. Newman AJ, Gross S. Hyperbilirubinemia
in breast-fed infants. Pediatrics 1963;32:995-1001.
6. Winfield CR, Macfaul R. Clinical study
of prolonged jaundice in breast- and bottle-fed babies. Arch Dis Child
1978;53:506-7. Crossref
7. Tazawa Y, Abukawa D, Watabe M, et al.
Abnormal results of biochemical liver function tests in breast-fed infants
with prolonged indirect hyperbilirubinaemia. Eur J Pediatr 1991;150:310-3.
Crossref
8. Siu LY, Wong KN, Li KW, et al. Outcome
of hepatobiliary scanning: preterm versus full-term cholestatic infants. J
Paediatr Child Health 2013;49:E46-51. Crossref
9. Landaas S, Skrede S, Steen JA. The
levels of serum enzymes, plasma proteins and lipids in normal infants and
small children. J Clin Chem Clin Biochem 1981;19:1075-80. Crossref
10. Gómez P, Coca C, Vargas C, et al.
Normal reference-intervals for 20 biochemical variables in healthy
infants, children and adolescents. Clin Chem 1984;30:407-12.
11. Jørgensen MH, Ott P, Juul A, et al.
Does breast feeding influence liver biochemistry? J Pediatr Gastroenterol
Nutr 2003;37:559-65. Crossref
12. Minamishima I, Ueda K, Minematsu T, et
al. Role of breast milk in acquisition of cytomegalovirus infection.
Microbiol Immunol 1994;38:549-52. Crossref
13. Hamprecht K, Maschmann J, Vochem M, et
al. Epidemiology of transmission of cytomegalovirus from mother to preterm
infants by breastfeeding. Lancet 2001;357:513-8. Crossref
14. Poddighe D, Castelli L, Marseglia GL,
et al. Prolonged, but transient, elevation of liver and biliary functions
tests in a healthy infant affected with breast milk jaundice. BMJ Case Rep
2014;pii:bcr2014204124. Crossref
15. Kemper K, Forsyth B, McCarthy P.
Jaundice, terminating breast-feeding and the vulnerable child. Pediatrics
1989;84:773-8.
16. Woo J, Lam CW, Leung J, et al. Very
high rates of vitamin D insufficiency in women of child-bearing age living
in Beijing and Hong Kong. Br J Nutr 2008;99:1330-4. Crossref
17. Munns CF, Shaw N, Kiely M, et al.
Global consensus recommendations on prevention and management of
nutritional rickets. J Clin Endocrinol Metab 2016;10:394-415. Crossref