DOI: 10.12809/hkmj144260
© Hong Kong Academy of Medicine. CC BY-NC-ND 4.0
CASE REPORT
Technical considerations for ligation of ruptured hepatic artery aneurysm: is arterial
reconstruction necessary?
S Lam, MB, BS, MRes (Med)1;
Albert CY Chan, FRCS (Edin), FCSHK2;
Ronnie TP Poon, FRCS (Edin), FCSHK2
1 Department of Surgery, Queen Mary Hospital, Pokfulam, Hong Kong
2 Department of Surgery, The University of Hong Kong, Pokfulam, Hong
Kong
Corresponding author: Dr S Lam (lamshi07@gmail.com)
Abstract
Ruptured hepatic artery aneurysm is a rare life-threatening
condition. Open surgery with ligation
of the aneurysm is the treatment of choice if the
patient presents with haemodynamic instability.
Controversies exist on whether hepatic artery
reconstruction is needed after exclusion of the
aneurysm. Involvement of the gastroduodenal
artery origin was proposed as an indication for
reconstruction, but this might be difficult to ascertain
upon laparotomy. Recent studies showed that arterial
ligation distal to the gastroduodenal artery origin
does not necessarily result in ischaemic liver injury,
implying that reconstruction in such cases may
not be required, especially in a haemodynamically
unstable patient. A patient with common hepatic
artery aneurysm involving the gastroduodenal
artery origin presented with rupture and underwent
aneurysm ligation. Adequacy of intrahepatic arterial
flow was determined by intra-operative Doppler
ultrasonography and arterial reconstruction was not
performed. The technical considerations during the
operative management of ruptured hepatic artery
aneurysms are discussed.
Introduction
Hepatic artery aneurysm (HAA) is an uncommon
condition that can present with life-threatening
haemorrhage. In case of catastrophic intraperitoneal
rupture, laparotomy with ligation of the aneurysm is
often the treatment of choice due to its rapid access
and ability to secure control of the extravasation site.
Controversy exists whether hepatic artery should
be reconstructed after ligation of the aneurysm,
especially when distal ligation is performed on
the hepatic artery proper. We herein describe
the technical considerations during the operative
management of a patient with ruptured HAA.
Case report
A 69-year-old male presented to Queen Mary
Hospital, Hong Kong, with a 3-week history of
epigastric pain in December 2010. A contrast-enhanced
computed tomography (CT) of the
abdomen showed a common HAA with involvement
of the root of gastroduodenal artery (GDA; Fig 1a) but there was no evidence of contrast leakage or
haematoma formation at the time of diagnosis.
Complete blood profile showed evidence of
inflammation that included mild leukocytosis with
neutrophil predominance, thrombocytosis, and
mildly elevated erythrocyte sedimentation rate.
Serum biochemistry showed elevated γ-glutamyl
transpeptidase and C-reactive protein. Screening for
vasculitis and infection was unremarkable.
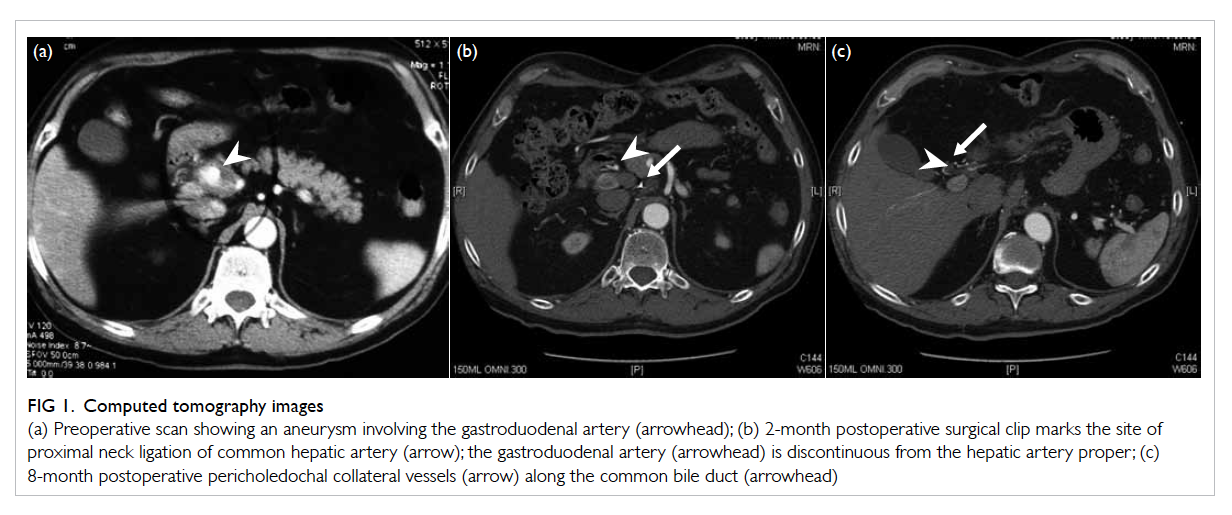
Figure 1. Computed tomography images
(a) Preoperative scan showing an aneurysm involving the gastroduodenal artery (arrowhead); (b) 2-month postoperative surgical clip marks the site of proximal neck ligation of common hepatic artery (arrow); the gastroduodenal artery (arrowhead) is discontinuous from the hepatic artery proper; (c) 8-month postoperative pericholedochal collateral vessels (arrow) along the common bile duct (arrowhead)
Three days later, however, the patient developed
generalised peritonitis complicated by hypovolaemic
shock during the workup for this condition in the
hospital. There was an acute drop in haemoglobin
level from 150 g/L to 125 g/L. A diagnosis of rupture
of the common HAA was made clinically and an
emergency laparotomy was performed.
On entering the peritoneal cavity, 5 L of
hemoperitoneum was recovered, and after four-quadrant
packing of the peritoneal cavity, supracoeliac
aortic clamping was performed to secure
temporary haemostasis. The lesser sac was entered
and a ruptured 4 x 3 cm fusiform aneurysm of the
common hepatic artery surrounded by inflamed
tissues cranial to the pancreatic head region was
identified. The proximal and distal neck of the
aneurysm was subsequently isolated and encircled by
tape without further dissection at the hepatoduodenal
ligament. After release of the aortic clamp, the distal
neck of the aneurysm at the hepatic artery proper
was test clamped and Doppler ultrasonography was
performed, which confirmed good biphasic flow in
both the right and left hepatic arteries. The aneurysm
was then ligated and suture-transfixed at its proximal
and distal neck region (Fig 2).
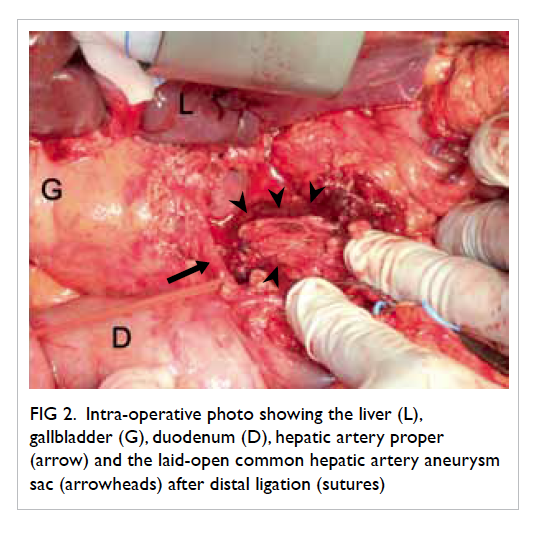
Figure 2. Intra-operative photo showing the liver (L), gallbladder (G), duodenum (D), hepatic artery proper (arrow) and the laid-open common hepatic artery aneurysm sac (arrowheads) after distal ligation (sutures)
In the postoperative period, the alanine and
aspartate transaminase levels were elevated to 1607
U/L and >3000 U/L, respectively. Both the liver
parenchymal enzymes returned to normal levels 3
weeks after the procedure. A short course of renal
replacement therapy was required for the acute renal
injury sustained during the shock. Otherwise, the
patient made an uneventful recovery. Pathological
examination of the aneurysm wall showed infiltration
of lymphocytes and neutrophils but there was no
evidence of infection.
Follow-up CT performed at 2 and 8 months
postoperatively showed arterial collateral formation
along the porta hepatis, and there was no evidence of
previous liver infarction (Figs 1b and 1c).
Discussion
Hepatic artery aneurysm is a rare condition with an
estimated prevalence of 0.1% according to autopsy
series,1 and incidence of 0.002%.2 The majority
(75%) of cases are incidentally diagnosed, and pain
in the right upper quadrant or epigastrium is the
most common presenting symptom.1 Rupture is the
second most common mode of presentation and
accounts for 14% of the newly diagnosed HAAs.2
The reported mortality rate for ruptured
HAA is 21% to 43%,2 3 and the key to successful
resuscitation is timely control of the ruptured site.
Although endovascular exclusion of HAAs has a
reported success rate of up to 100%,4 open surgery
is often the treatment of choice if the exsanguinating
haemorrhage does not permit a failed trial of
endoluminal haemostasis.
The objectives of open surgery are to exclude
the diseased artery segment and preserve the
hepatic arterial inflow. It is generally accepted that
aneurysms of the common hepatic artery which do
not involve the GDA can be simply ligated without
reconstruction of inflow to the hepatic artery proper
because the latter is supplied by the reverse flow
from the GDA.5 For aneurysms involving GDA or
distal to its origin, the conventional advocate is to
reconstruct hepatic arterial inflow after ligation
or resection of the aneurysm to avoid the risk of
ischaemic injury to the liver.6 Depending on the
morphology and size of the aneurysm, inflow
restoration may take the form of aneurysmectomy
with patch graft repair or exclusion of aneurysm
with coeliac or aorto-hepatic bypass grafting.
Autogenous saphenous vein is the preferred graft,
although a dacron prosthetic graft can also be used.6
In our patient, because of the clinical suspicion of
an infective aetiology for the ruptured aneurysm and
due to the satisfactory hepatic arterial flow during
intra-operative ultrasound, arterial reconstruction
was not considered.
Graham and Cannel7 had reviewed that
accidental hepatic artery ligation would result in
greater than 50% mortality when the site of ligation
is distal to the GDA. In later studies, however,
such a high mortality rate was not observed with
hepatic artery ligation during iatrogenic injuries or
therapeutic procedures to ‘de-arterialise’ tumour-laden
livers.5 8 9 It is believed that the liver is protected
from ischaemic insults after hepatic artery ligation
by a buffer response of the portal venous system. The
portal vein maintains the supply of 70% of hepatic
inflow and 50% of hepatic oxygen consumption
during acute hepatic artery interruption, before
compensatory arterial supply develops.6 With time, a
multitude of arterial collateral channels can develop
to compensate for the interruption of original arterial
inflow. In fact, postoperative CT scan at 8 months in
our patient clearly showed collateral formation via
the pericholedochal route (Fig 1c). Besides, Michels10
has described 26 potential sites of collateral arterial
supplies to the liver originating from the celiac
trunk, superior mesenteric artery and intrathoracic
arteries, routing via the hepatoduodenal ligament,
hepatogastric ligament, omentum, retroperitoneum,
falciform ligament, and diaphragmatic connections.
Collateral formation is observed after ligation of
hepatic arteries in angiographic studies as early as 10
hours after ligation.5 Moreover, replaced or accessory
arterial supplies to the left or right hepatic lobes are
present in about 30% of the general population,5 11 rendering feasible supply to the contralateral lobe
via the interlobar anastomosis at the liver hilum.
The clinical relevance of such vascular recovery
potential was demonstrated by Chirica et al,12
wherein the technique of simple aneurysm ligation
with minimal dissection in the hepatoduodenal
ligament was adopted so as to preserve the potential
arterial collaterals and hence avoiding the need for
reconstruction of the extirpated hepatic artery in
three out of four patients in their series of HAAs
with GDA involvement.
Establishment of arterial collaterals, however,
is a latent process and the anatomical information
on the presence of aberrant or replaced hepatic
arterial supply may not be readily available at the
time of laparotomy for a ruptured aneurysm. In this
situation, intra-operative Doppler ultrasonography
is a convenient tool for evaluation of intrahepatic
arterial flow. In transplanted livers, a normal
Doppler waveform of the hepatic artery includes a
rapid upstroke with an acceleration time of <80 ms,
a continuous diastolic flow giving a resistive index
between 0.5 and 0.7, and a peak systolic velocity
of <2 m/s; any deviation from normality from any
of these parameters is suggestive of hepatic artery
stenosis and predictive of subsequent ischaemic
bile duct injury.13 Perhaps, the same criteria can be
adopted to assess the arterial sufficiency in a native
liver. Back bleed from distal cut end of hepatic artery
and cyanosis of hepatic lobe should be counted with
caution as these have been reported to be inaccurate
in predicting adequate perfusion or subsequent
ischaemic injury, respectively.14
Conclusion
Simple ligation of the hepatic artery is considered
safe and effective in the treatment of ruptured HAA.
The establishment of arterial collaterals should be
expected given that the hepatoduodenal ligament
is not severely disrupted. The use of intra-operative
ultrasound facilitates the decision-making in
devising the operative strategy for this condition.
Declaration
No conflict of interest was declared by the authors.
References
1. Arneson MA, Smith RS. Ruptured hepatic artery
aneurysm: case report and review of literature. Ann Vasc
Surg 2005;19:540-5. Crossref
2. Abbas MA, Fowl RJ, Stone WM, et al. Hepatic artery
aneurysm: factors that predict complications. J Vasc Surg
2003;38:41-5. Crossref
3. Shanley CJ, Shah NL, Messina LM. Common splanchnic
artery aneurysms: splenic, hepatic, and celiac. Ann Vasc
Surg 1996;10:315-22. Crossref
4. Tulsyan N, Kashyap VS, Greenberg RK, et al. The
endovascular management of visceral artery aneurysms
and pseudoaneurysms. J Vasc Surg 2007;45:276-83. Crossref
5. Mays ET, Wheeler CS. Demonstration of collateral arterial
flow after interruption of hepatic arteries in man. N Engl J
Med 1974;290:993-6. Crossref
6. Tygstrup N, Winkler K, Mellemgaard K, Andreassen
M. Determination of the hepatic arterial blood flow and
oxygen supply in man by clamping the hepatic artery
during surgery. J Clin Invest 1962;41:447-54. Crossref
7. Graham RR, Cannel D. Accidental ligation of the hepatic
artery. Br J Surg 1933;20:566-79. Crossref
8. Ariyan S, Cahow CE, Greene FL, Stansel HC Jr. Successful
treatment of hepatic artery aneurysm with erosion into the
common duct. Ann Surg 1975;182:169-72. Crossref
9. Brittain RS, Marchioro TL, Hermann G, Waddell WR,
Starzl TE. Accidental hepatic artery ligation in humans.
Am J Surg 1964;107:822-32. Crossref
10. Michels NA. Collateral arterial pathways to the liver after
ligation of the hepatic artery and removal of the celiac axis.
Cancer 1953;6:708-24. Crossref
11. Covey AM, Brody LA, Maluccio MA, Getrajdman GI,
Brown KT. Variant hepatic arterial anatomy revisited:
digital subtraction angiography performed in 600 patients.
Radiology 2002;224:542-7. Crossref
12. Chirica M, Alkofer B, Sauvanet A, Vullierme MP, Levy
Y, Belghiti J. Hepatic artery ligation: a simple and safe
technique to treat extrahepatic aneurysms of the hepatic
artery. Am J Surg 2008;196:333-8. Crossref
13. Crossin JD, Muradali D, Wilson SR. US of liver transplants:
normal and abnormal. Radiographics 2003;23:1093-114. Crossref
14. Mathisen DJ, Athanasoulis CA, Malt RA. Preservation of
arterial flow to the liver: goal in treatment of extrahepatic
and post-traumatic intrahepatic aneurysms of the hepatic
artery. Ann Surg 1982;196:400-11. Crossref

