Hong Kong Med J 2015 Jun;21(3):217–23 | Epub 26 Mar 2015
DOI: 10.12809/hkmj144325
© Hong Kong Academy of Medicine. CC BY-NC-ND 4.0
ORIGINAL ARTICLE
Local review of treatment of hand enchondroma (artificial bone substitute versus autologous bone graft) in a tertiary referral centre: 13 years’ experience
YW Hung, FHKCOS, FHKAM (Orthopaedic Surgery)1,2;
WS Ko, MB, ChB2;
WH Liu, MB, BS1;
CS Chow, FHKCOS, FHKAM (Orthopaedic Surgery)1,2;
YY Kwok, BNurs, RN1;
Clara WY Wong, FHKCOS, FHKAM (Orthopaedic Surgery)1,2;
WL Tse, FHKCOS, FHKAM (Orthopaedic Surgery)1,2;
PC Ho, FHKCOS, FHKAM (Orthopaedic Surgery)1,2
1 Department of Orthopaedics and Traumatology, Prince of Wales Hospital, Shatin, Hong Kong
2 Department of Orthopaedics and Traumatology, Alice Ho Miu Ling Nethersole Hospital, Tai Po, Hong Kong
Corresponding author: Dr YW Hung (hungyukwah@yahoo.com.hk)
Abstract
Objective: To evaluate the treatment outcomes
of enchondroma of the hand with artificial bone
substitute versus autologous (iliac) bone graft.
Design: Historical cohort study.
Setting: Tertiary referral centre, Hong Kong.
Patients: A total of 24 patients with hand
enchondroma from January 2001 to December 2013
who underwent operation at the Prince of Wales
Hospital and Alice Ho Miu Ling Nethersole Hospital
in Hong Kong were reviewed. Thorough curettage of
the tumour was performed in all patients, followed
by either autologous bone graft impaction under
general anaesthesia in 13 patients, or artificial
bone substitute in 11 patients (10 procedures were
performed under local or regional anaesthesia
and 1 was done under general anaesthesia). The
functional outcomes and bone incorporation were
measured by QuickDASH (shortened version of the Disabilities of the
Arm, Shoulder and Hand questionnaire) scores and radiological
appearance, respectively. The mean follow-up period
was 59 months.
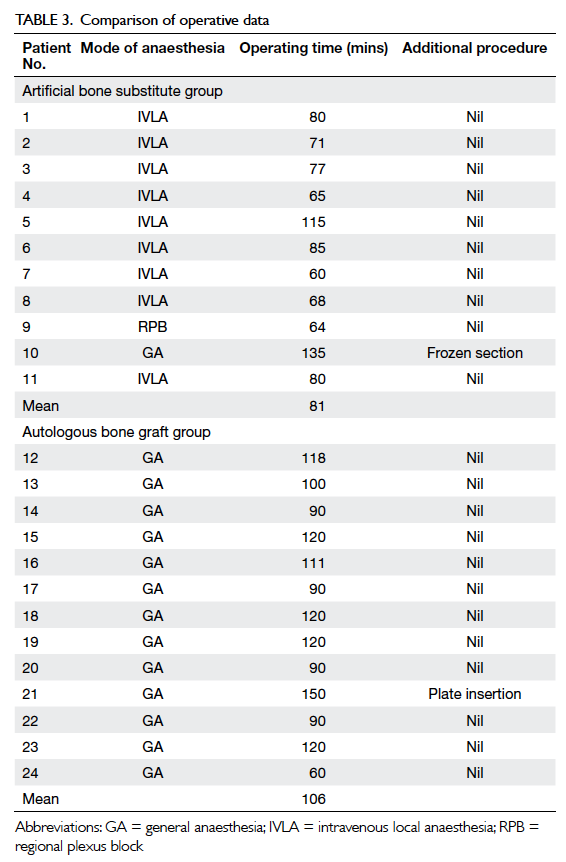
Results: There were eight men and 16 women, with
a mean age of 40 years. Overall, 17 cases involved
phalangeal bones and seven involved metacarpal bones.
Among both groups of patients, most of the affected
digits had good range of motion and function
after surgery. One patient in each study group had
complications of local soft tissue inflammation. One
patient in the artificial bone substitute group was
suspected to have recurrence 8 years after operation.
Among the autologous bone graft group, four
patients had persistent donor site morbidity at the
last follow-up. In all patients, radiographs showed
satisfactory bone incorporation.
Conclusions: Artificial bone substitute is a safe and
effective treatment option for hand enchondroma,
with satisfactory functional and radiographic
outcomes. Artificial bone substitute offers the
additional benefits of enabling the procedure to be
done under local anaesthesia on a day-case basis
with minimal complications.
New knowledge added by this
study
- Curettage followed by artificial bone substitute is a safe and effective way to manage enchondroma in the hand.
- Using artificial bone substitute to replace classic autologous bone graft for managing enchondroma in the hand has several advantages: (a) reduced donor site morbidity; (b) significantly reduced surgical time; (c) comparable results to autologous bone graft in terms of clinical and radiological outcomes; and (d) enables the surgery to be performed under local or regional anaesthesia, thus, patients can be discharged on the same day as the surgery.
Introduction
Enchondroma is one of the most common benign
bone tumours of the hand. It originates from cartilage
and is commonly located in the proximal metaphysis
of the proximal phalanx.1 The tumour usually
presents as an incidental finding or pathological
fracture.
Despite being the most common bone tumour
in the hand, standardised treatment protocols are
lacking.1 Options vary from observation alone,
curettage alone, and curettage with bone grafting
(recently with artificial bone substitute). At the
Prince of Wales Hospital (PWH), we used to treat
enchondroma of the hand by complete curettage
and filling the defect with autologous bone graft.
Although autologous bone graft provides both
biological and mechanical advantages in managing
the bone void, this procedure is not without risk.
Patients must undergo general anaesthesia to obtain
the bone graft from the iliac crest, and most patients
have considerable postoperative pain, which limits
their walking ability for a variable period.
Recently, studies evaluating the clinical
application of artificial bone substitute have shown
promising results.2 3 However, this is a relatively new technique in local practice. In a cohort study, we
retrospectively analysed the treatment outcomes of
patients with hand enchondroma and compared the
results for autologous bone graft and artificial bone
substitute.
Methods
From January 2001 to December 2013, all patients
with symptomatic monostotic enchondroma of the
phalanges or metacarpals treated at the PWH or the
Alice Ho Miu Ling Nethersole Hospital (AHNH)
underwent thorough curettage according to the
standard protocol. The bone defects were filled
by either autologous bone graft or artificial bone
substitute, depending on the surgeon’s and patient’s
preferences. All operations were done by the
same team of orthopaedic specialists. For patients
presenting with pathological fracture, the fracture
was first managed conservatively until healed. The
surgery for the tumour was performed 3 months
after initial presentation.
Surgical technique
In the artificial bone substitute group, an incision
was centred on the lesion, and the extensor tendon
was retracted, with no subperiosteal dissection. A
small oval cortical window was made by connecting
multiple drill hole perforations prepared by a
0.9-mm Kirschner wire. The tumour was removed
by small-angle curettage and clearance was checked
under fluoroscopic control (Figs a and b). The cavity
was then filled with artificial bone substitute (Fig c).
A custom-made paper funnel was used for
precise insertion of bone substitute to avoid spillage
to the surrounding soft tissue, which could be
difficult to remove (Figs d and e). Bone substitute
granules were impacted tightly by using a punch
(Fig f). The piece of oval cortical bone was placed
back in position, and the periosteum was repaired
where possible; alternatively, the window was sealed
with fibrin glue (Tisseel; Baxter Healthcare Corp,
Deerfield [IL], US) to contain the bone substitute (Fig
g). The wound was closed with fine nylon suture. A
radiograph was taken to confirm filling of the defect
and absence of fracture (Fig h). Free mobilisation
was allowed postoperatively.
In the autologous bone graft group, the
operation was done under general anaesthesia. The
surgical approach and procedures to the affected
bone were the same as for the artificial bone substitute
group, except that autologous cancellous bone grafts
harvested from iliac crest were used instead of
artificial bone substitute. We do not usually obtain
the bone graft from the ipsilateral distal radius as the
quantity is insufficient for packing the wound. Free
mobilisation was allowed postoperatively.
Statistical analysis
The operative details and postoperative clinical
and radiological outcomes were reviewed by an
independent reviewer. Fisher’s exact test was used for
sex, tumour site, and pain score. Mann-Whitney U
test was used for the Disabilities of the Arm, Shoulder
and Hand questionnaire (DASH) score, and t test was used for
the other parameters. Clinically, the active range of
motion, symptoms, and function measured by the
Chinese and shortened version of the DASH (QuickDASH)4 were evaluated.
Plain radiographs were taken at standard intervals (1
week, 4 weeks, 3 months, 6 months, and annually)
postoperatively to determine bone incorporation.
Bone incorporation was defined as a seamless
appearance with no gap between the cancellous
bone and the bone substitute. For any suspicious
symptoms or radiographic appearance, computed
tomography or magnetic resonance imaging (MRI)
was performed to look for any recurrence.
Results
There were 24 patients (eight men and 16 women),
with a mean age of 40 years. Overall, 17 cases
involved the phalangeal bones and seven involved
the metacarpal bones; 13 patients underwent
autologous bone graft and 11 had artificial bone
substitute. Five patients in each group presented
with pathological fracture, among whom nine were
managed conservatively until the fracture was healed.
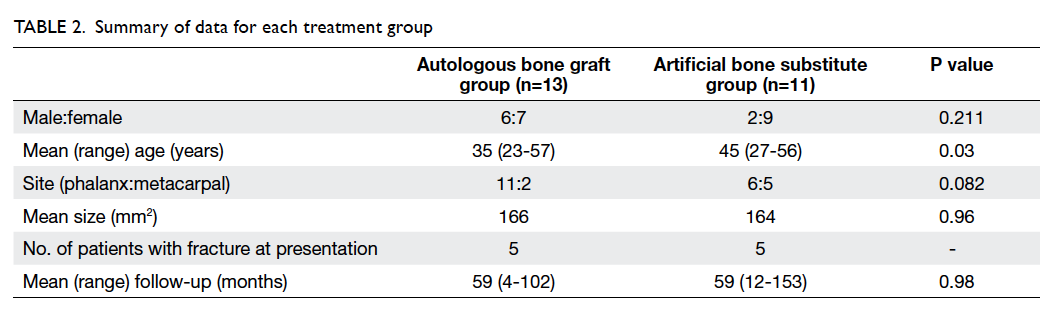
Patients’ demographics, including site and size of
the tumour, presentation, and time to operation
are shown in Tables 15 and 2. In all patients, the
histology confirmed the diagnosis of enchondroma.
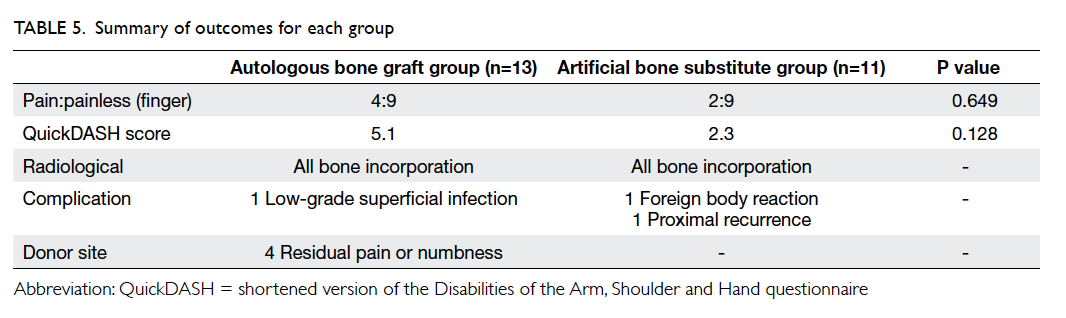
The operative details and postoperative outcomes
are shown in Tables 3, 4, and 5.
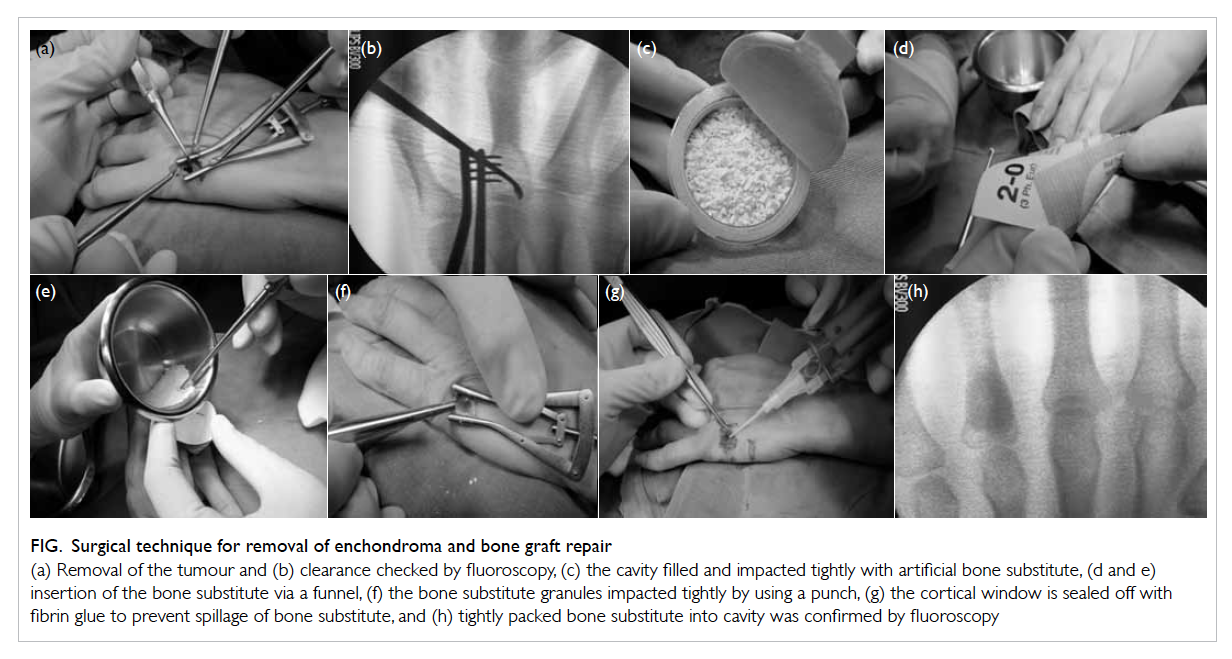
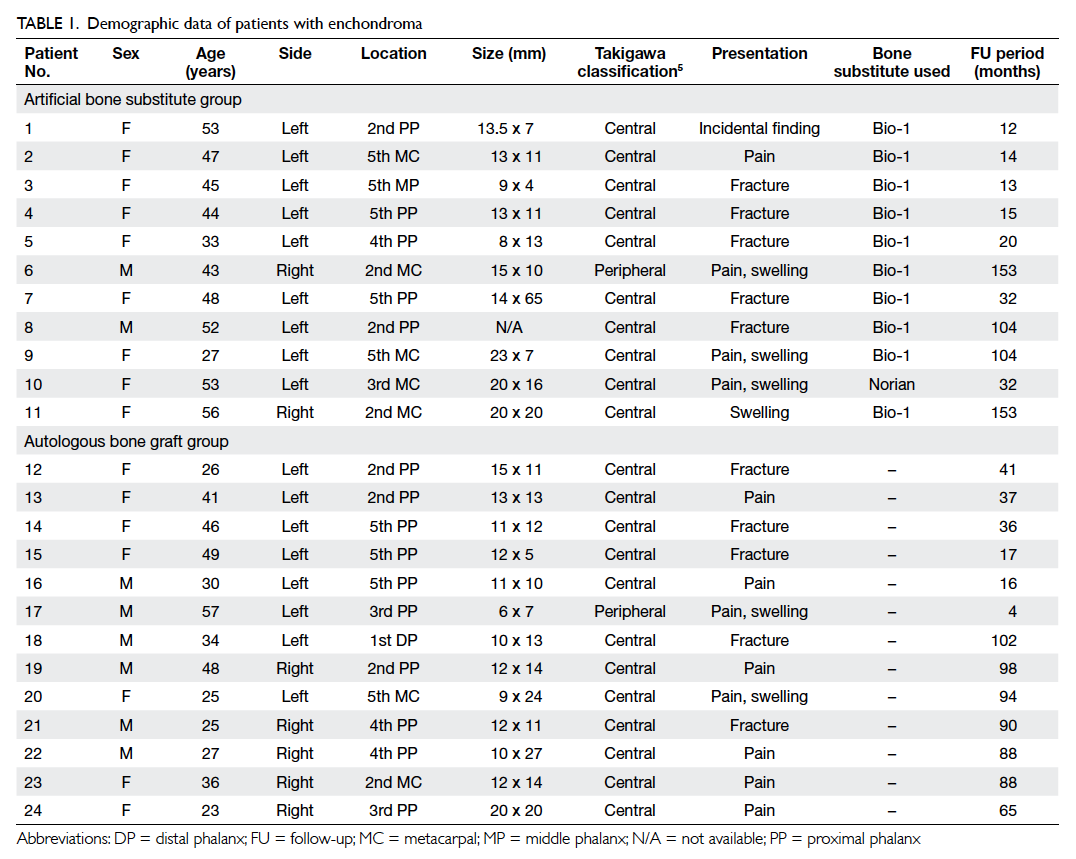
Figure. Surgical technique for removal of enchondroma and bone graft repair
(a) Removal of the tumour and (b) clearance checked by fluoroscopy, (c) the cavity filled and impacted tightly with artificial bone substitute, (d and e) insertion of the bone substitute via a funnel, (f) the bone substitute granules impacted tightly by using a punch, (g) the cortical window is sealed off with fibrin glue to prevent spillage of bone substitute, and (h) tightly packed bone substitute into cavity was confirmed by fluoroscopy
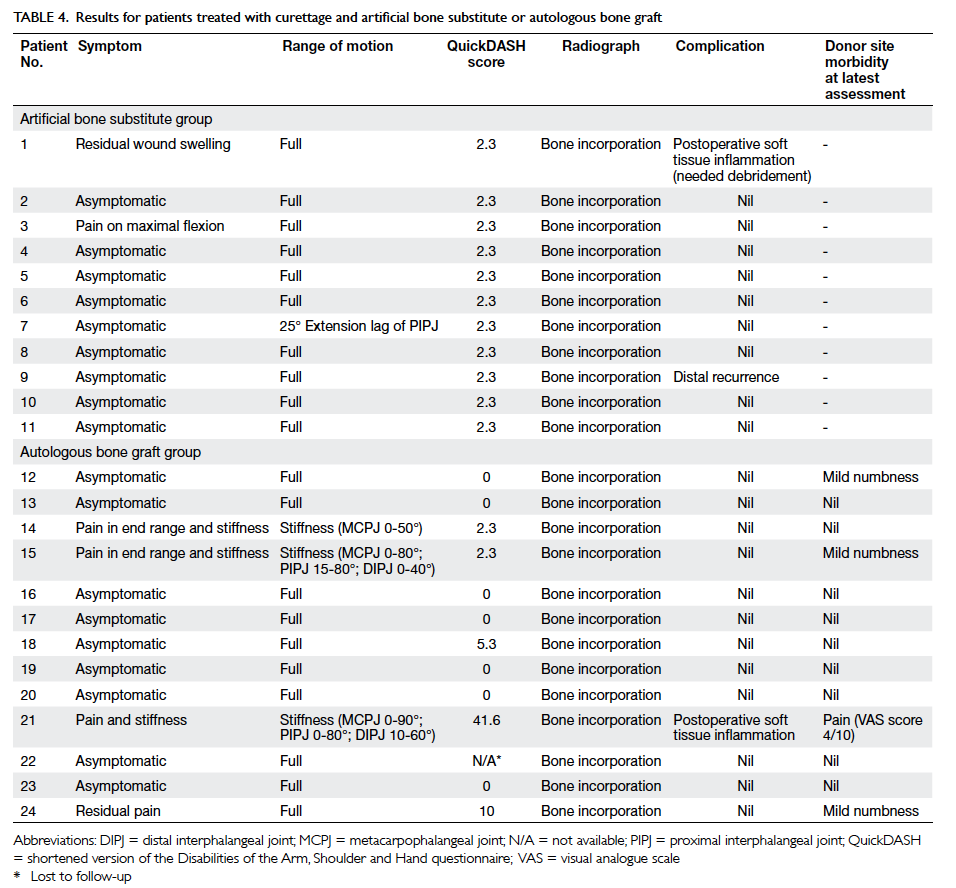
Table 4. Results for patients treated with curettage and artificial bone substitute or autologous bone graft
For the artificial bone substitute group, 10 of
11 patients were operated on using intravenous local
anaesthesia or regional plexus block. One patient
was operated on under general anaesthesia as the
MRI showed suspicion for malignancy, and frozen
section was performed during the operation. The
mean surgical time was 81 minutes (range, 60-135
minutes). All surgeons used Bio-1 granules (SBM
France, Lourdes, France) except for one patient
for whom injectable bone substitute (Norian SRS;
Synthes USA, Paoli [PA], US) was used because of
the surgeon’s preference.
For the autologous bone graft group, all 13
patients were operated on under general anaesthesia.
The mean surgical time was 106 minutes (range,
60-150 minutes), which was 25 minutes longer than
for the artificial bone substitute group (P=0.008).
The mean follow-up period was 59 months
(mean 59 months, range 4-102 months in the
autologous bone graft group and mean 59 months,
range 12-153 months in the artificial bone substitute
group). All patients demonstrated satisfactory bone
incorporation. There were no significant observable
radiological differences between the groups 1 year
after operation. Functional recovery was similar in
both groups. There were no significant differences
in QuickDASH scores (mean 2.3 for artificial bone
substitute group and 5.1 for autologous bone graft
group; P=0.128).
Complications
One patient in each group developed soft tissue
complications 3 weeks after the operation. The
patient in autologous bone graft group presented
with erythema over the surgical site. The condition
improved after intravenous antibiotic treatment
was given and the patient was diagnosed to have
a low-grade superficial infection. The patient in
the artificial bone substitute group presented with
discharge from the wound and radiograph showed a
trace amount of tiny calcifications in the soft tissue
adjacent to the affected digit. The culture swab of
the discharge fluid showed negative growth. The
patient was treated with empirical antibiotics and
surgical debridement showed a small amount of
bone substitute in the subcutaneous plane of the
wound. The diagnosis was probable inflammation
secondary to foreign body reaction, rather than a
genuine infection. Both patients had satisfactory
wound healing and bone healing.
Recurrence of enchondroma was suspected in
one patient in the artificial bone substitute group.
The patient had a radiolucent lesion at the proximal
part of the affected metacarpal on plain radiograph
during routine follow-up 8 years after the index
operation. The patient subsequently underwent a
second operation to remove the lesion.
Discussion
Joosten et al2 first reported treatment of enchondroma with artificial bone substitute in 2000. Eight patients
were treated with hydroxyapatite cement to fill the
bone cavity. All of the patients gained full function
of the hand and no complications were observed
during 1-year follow-up. Subsequently, studies
from Japan3 and South Korea6 have also shown satisfactory outcomes using calcium bone cement
and calcium-based pellets, respectively.
At the PWH and AHNH, we treat all hand
enchondromas surgically because the tumour
will usually grow, weaken the bone, and result in
pathological fracture. Since the bone will be further
weakened by curettage alone, we believe that
replacement with an osteogenic or osteoconductive
substance will facilitate bone healing and remodelling
so that this fracture-prone period can be shortened.
We traditionally treated hand enchondroma with
curettage and filled the defect with autologous bone
graft. However, we started treating with artificial bone
substitute in 2001. In 2010, we changed to routine use
of autologous bone graft because there are several
advantages of reduced donor site morbidity, use of
local anaesthesia, reduced operating time (mean, 25
minutes less), and the surgery can be performed on a
day-case basis.
There are different types of bone substitute
available in the market. In this study, either Bio-1
granules or injectable Norian7 was used. These
bone substitutes are synthetic materials made with
resorbable calcium phosphate. The composition
comprises calcium and phosphate ions, which are
biocompatible with natural bone minerals.8 An in-vitro
study shows that calcium phosphate allows
osteoblast fixation and proliferation,8 followed by
osteointegration and bone resorption mimicking
normal bone healing. Calcium phosphate is available
in granules or cubes and in an injectable form.
In this study, complete curettage of the tumour
was achieved, with histological confirmation of the
diagnosis. There were no significant differences in
QuickDASH scores between the two groups.
Autologous bone graft takes around 4 to 6
months to incorporate while, for artificial bone
substitute, the time to incorporation depends
on the type of bone substitute used. Bio-1 takes
approximately 9 to 12 months to incorporate. There
were no significant radiological differences between
the groups at 1 year postoperatively. Norian stays in
the bone for longer than Bio-1 and is not completely
resorbed up to 3 years postoperatively.
The mean follow-up period of this study was
59 months, which is longer than in most studies.
The numbers of patients in each treatment group
were comparable with other studies. We observed
suspected recurrence in the affected metacarpal in
one patient, who had undergone operation 8 years
previously. A radiolucent lesion was noted beneath
the bone substitute. We postulated that there might
have been residual enchondroma cells seeding at
the base of the lesion after curettage, which were
displaced proximally during impaction of the bone
substitute.
There were several limitations to this study.
First, there was a difference in patient age between
the two groups, which is a confounding variable.
This might be accounted for by the relatively low
incidence of enchondroma despite it being the most
common upper limb tumour. Second, the choice
of artificial bone substitute was not standardised,
as two different substitutes were used. Norian SRS
injectable bone substitute was used in one patient
and Bio-1 was used in the other patients. Third,
radiological assessment postoperatively might not be
accurate. Despite all radiographs being reviewed by
experienced orthopaedic specialists, the diagnosis of
bone incorporation was subjective, with the chance
for inter- and intra-observer bias. Finally, this study
was retrospective and non-randomised.
Conclusions
Overall, most patients gained full range of motion
and satisfactory function, with radiological evidence
of bone incorporation and, later, bone growth.
The application of artificial bone substitute gives
comparable functional and radiological results in
treating enchondroma of the hand. The procedure
allows reduction in operating time, elimination of
donor site morbidity, and day-case surgery under
local or regional anaesthesia. Meticulous curettage
and bone substitute impaction without spillage to
the surrounding soft tissues are key to achieving
good outcomes and avoiding complications.
References
1. Sassoon AA, Fitz-Gibbon PD, Harmsen WS, Moran SL.
Enchondromas of hand: factors affecting recurrence,
healing, motion, and malignant transformation. J Hand
Surg Am 2012;37:1229-34. Crossref
2. Joosten U, Joist A, Frebel T, Walter M, Langer M. The use
of in situ curing hydroxyapatite cement as an alternative of
bone graft following removal of enchondroma of the hand.
J Hand Surg Br 2000;25:288-91. Crossref
3. Yasuda M, Masada K, Takeuchi E. Treatment of
enchondroma of the hand with injectable calcium
phosphate bone cement. J Hand Surg Am 2006;31:98-102. Crossref
4. Lee EW. Chinese QuickDASH (PWH, HK version).
Physiotherapy Department, Prince of Wales Hospital.
Toronto: Institute for Work and Health; 2006.
5. Takigawa K. Chondroma of the bones of the hand. A review
of 110 cases. J Bone Joint Surg Am 1971;53:1591-600.
6. Choy WS, Kim KJ, Lee SK, Yang DS, Park HJ. Treatment
for hand enchondroma with curettage and calcium sulfate
pellet (OsteoSet®) grafting. Eur J Orthop Surg Traumatol
2012;22:295-9. Crossref
7. Hak DJ. The use of osteoconductive bone graft substitutes in
orthopaedic trauma. J Am Acad Orthop Surg 2007;15:525-36.
8. Le Huec JC, Clément D, Lesprit E, Faber J. The use of
calcium phosphate, their biological properties. Eur J
Orthop Surg Traumatol 2000;10:223-9. Crossref