Hong Kong Med J 2026;32:Epub 16 Apr 2026
© Hong Kong Academy of Medicine. CC BY-NC-ND 4.0
PERSPECTIVE
New frontiers in rectal cancer treatment:
implementing Watch and Wait in Hong Kong
Trevor MY Yeung, DPhil, FRCS; Kaori Futaba, MB, ChB, FRCS; Simon SM Ng, MD, FRCS
Division of Colorectal Surgery, Department of Surgery, The Chinese University of Hong Kong, Hong Kong SAR, China
Corresponding author: Dr Trevor MY Yeung (trevoryeung@surgery.cuhk.edu.hk)
Introduction
Conventional treatment for patients with locally
advanced rectal cancer involves neoadjuvant therapy
(NAT) to downstage the tumour, followed by total
mesorectal excision (TME) and adjuvant therapy if
required. However, surgery is associated with risks,
including anastomotic leakage and low anterior
resection syndrome, which can substantially affect
overall quality of life. Furthermore, surgery for
low rectal cancer may result in the formation of a
permanent colostomy.
A subset of patients with stage II/III rectal
cancer develop a good clinical response to NAT,
termed either complete clinical response (cCR)
or near-complete response (nCR), depending on
endoscopic and radiological assessment. In 2004,
Habr-Gama et al1 first proposed the concept of
organ preservation, otherwise known as Watch and
Wait (WW), as an option for patients who respond
well to NAT, thereby avoiding the morbidity and
mortality associated with surgery, as well as the
potential need for a stoma. In their seminal paper,
they demonstrated that patients who achieved
cCR and were managed non-operatively had better
5-year overall survival and similar 5-year disease-free
survival compared with those who underwent
surgery and had a pathological complete response
(pCR) in the resection specimen.1
In recent years, the use of total neoadjuvant
therapy (TNT) has become increasingly popular, as
it is associated with a reduced risk of distant failure,2
greater compliance with chemotherapy, shorter
time to ileostomy closure, and increased tumour
response.3 Randomised controlled trials have
confirmed that TNT can enhance tumour response;
both PRODIGE 234 and RAPIDO5 reported a
doubling of pCR rates to 28% with TNT. Given
the increasing proportion of patients offered TNT,
more patients will develop cCR or nCR and would
therefore be eligible for WW.
Clinical assessment of patients
after neoadjuvant therapy
All patients with stage II/III biopsy-proven rectal
cancer who lack evidence of metastatic disease and
develop a good tumour response after NAT can be considered candidates for WW. Patients should be
restaged within 8 (±4) weeks of completing NAT; this
should include digital rectal examination, endoscopic
assessment, and magnetic resonance imaging (MRI).
Endoscopic response should be graded as cCR, nCR,
or incomplete clinical response (iCR) according to
the MSKCC (Memorial Sloan Kettering Cancer
Center) tumour regression criteria.6 Magnetic
resonance imaging response should be graded
according to tumour regression grade.
Patients with an iCR should be offered standard
TME surgery within 12 weeks of completing NAT.
They will then undergo standard postoperative
surveillance in accordance with institutional
guidelines. Patients who develop a good tumour
response (cCR or nCR) should be offered the option
of WW. As established in the Organ Preservation for
Rectal Adenocarcinoma (OPRA) trial, if there is a
discrepancy between endoscopic and MRI findings
regarding tumour response, the endoscopic findings
take precedence.7 Patients who agree to undergo
WW should follow the intensive surveillance
protocol detailed below.
Patient selection
It is important to establish which patients should
be offered WW surveillance. Earlier studies focused
only on patients who had achieved cCR and did
not include those with nCR, owing to uncertainty
regarding its underlying biology. However, there is
evidence that nCR comprises two subpopulations:
one group that will eventually achieve cCR, and
another that will subsequently demonstrate tumour
enlargement and develop iCR.
Several studies have demonstrated that
increasing the interval between NAT completion and
surgery increases the proportion of patients suitable
for WW. Probst et al8 reported that, in an analysis
of 17 255 patients with stage II/III locally advanced
rectal cancer who underwent NAT followed by
surgical resection, waiting longer than 8 weeks after
NAT was associated with higher odds of pCR and
tumour downstaging. Furthermore, Hupkens et al9
reported that 90% of patients with nCR at 8 to 10
weeks after NAT subsequently developed cCR (6
to 12 weeks later). Even when trimodal assessment indicates residual tumour, 15% of patients eventually
achieve cCR.10 In a multicentre phase 2 trial, Garcia-Aguilar et al11 demonstrated that extending the
interval between neoadjuvant chemoradiotherapy
and surgery increased the proportion of patients
who eventually developed pCR.
The OPRA trial7 reported promising outcomes
with a WW strategy in patients who developed
either cCR or nCR. In this study, the 3-year TME-free
survival rate was 53% among patients who
underwent TNT with consolidation chemotherapy.
Provided that patients with cCR show no signs of
local regrowth, and those with nCR continue to
demonstrate tumour regression after NAT, they
remain suitable for ongoing surveillance within a
WW programme.
Appropriate candidates for WW are often
patients with mid- to distal rectal cancer. This trend
arises because the alternatives—abdominoperineal
resection with a permanent colostomy, or an ultralow
or coloanal anastomosis—are both associated with
poor functional outcomes. Age does not appear to
affect clinical outcomes. The International Watch
and Wait Database (IWWD) reported no differences
in disease-specific survival, local regrowth, or risk
of distant metastases between younger patients and
those over 50 years of age.12
Certain pathological features are associated
with lower cCR rates, including larger tumour
size, a circumferential resection margin of smaller
than 1 mm, extramural venous invasion, extensive
mesorectal or pelvic lymph nodes, and mutations
in TP53 and SMAD4.13 Patients with ulcerated or circumferential tumours carry a risk of severe
scarring and narrowing of the rectal lumen, which
may preclude endoscopic follow-up; such tumours
represent a relative contraindication to WW.14
Surveillance
Intensive surveillance is required to detect early
tumour regrowth in any patient choosing WW
(Table). Surveillance is most frequent during the
first 2 years, given that 88% of local regrowth
occurs within this period.15 International guidelines
for WW surveillance recommend 4-monthly
flexible sigmoidoscopy and measurement of
carcinoembryonic antigen levels during the first 2
years, followed by 6-monthly assessments for the
subsequent 3 years.7 Magnetic resonance imaging is
recommended every 6 months for the first 2 years,
then annually for the following 3 years.7 Computed
tomography of the thorax, abdomen, and pelvis
should be performed annually, and colonoscopy in
Years 1 and 5. However, patients may be reviewed
more frequently if concerns are raised by either the
patient or the clinician.
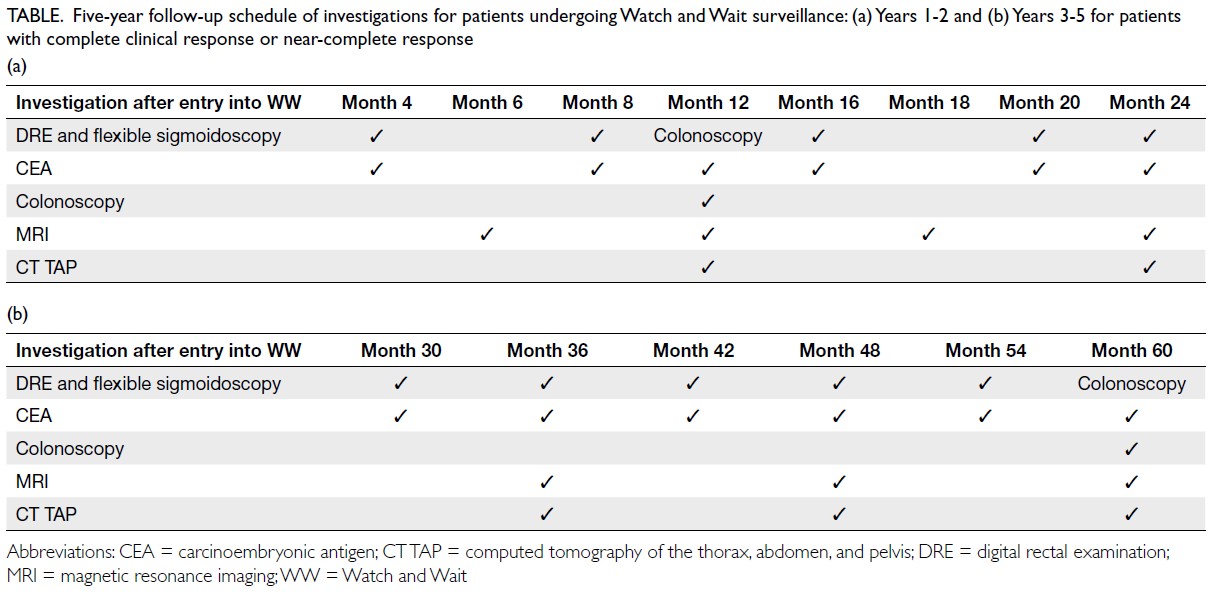
Table. Five-year follow-up schedule of investigations for patients undergoing Watch and Wait surveillance: (a) Years 1-2 and (b) Years 3-5 for patients with complete clinical response or near-complete response
During WW surveillance, patients with
sustained cCR, as well as those with nCR who
subsequently achieve cCR or continue to demonstrate
tumour regression, should remain on the WW
follow-up protocol. If there are signs of tumour
regrowth in a patient who previously achieved cCR,
or if a patient with nCR shows an increase in tumour
size compared with the previous clinical evaluation,
TME should be offered within 4 to 6 weeks.
Resource requirements
Watch and Wait has not previously been offered in
the Hong Kong public healthcare system. Several
key challenges must be addressed before WW
can be widely implemented locally. Watch and
Wait surveillance involves regular endoscopic
and MRI examinations, which require additional
infrastructure and funding. Although it may be
feasible for Hong Kong colorectal surgeons to
arrange additional flexible sigmoidoscopies within
their own departments, scheduling extra MRI scans
remains challenging because of limited appointment
availability. There is increasing evidence that
luminal assessment is more informative than MRI
for detecting tumour regrowth during WW. In
the OPRA trial, when there was a discrepancy
between endoscopic and MRI findings, endoscopic
findings took precedence, given that rectal cancer is
primarily a mucosal disease.7 Furthermore, analysis
of data from the IWWD revealed that 97% of local
regrowth occurred in the bowel wall.15 Considering
the limited availability of MRI appointments within
the Hong Kong public healthcare system, it may be
necessary to develop a WW surveillance programme
tailored to local resources, for example by arranging
MRI examinations more selectively, particularly in
response to an elevated carcinoembryonic antigen
level.
Implementation of WW also relies on
consistent and robust endoscopic assessment of
tumour response.6 Training is essential to ensure
high-quality and consistent reporting. Increasing
awareness of WW in Hong Kong, and educating
local clinicians on the endoscopic classification of
tumour response, will help ensure that the most
appropriate patients are offered WW within a robust
surveillance programme.
Oncological outcomes
Because WW is not yet widely adopted, there may be
hesitancy among local clinicians regarding the long-term
oncological outcomes of patients undergoing
WW. However, these concerns are not supported by
recent data. The IWWD examined 880 patients with
cCR who underwent WW and reported a 2-year
local regrowth rate of 25%, a 5-year overall survival
rate of 85%, and a 5-year disease-free survival rate
of 94%.15 For patients who develop local regrowth,
salvage surgery was feasible in more than 95% of
cases16; pelvic control was maintained in 91% of those
who underwent salvage surgery.17 Regarding distant
metastasis, data from MSKCC indicate an increased
risk in patients with local regrowth compared
with those without local failure,17 and the IWWD
reported an incidence of distant metastasis of 18%
in patients with local regrowth compared with 5% in
those without local failure.15 However, these findings
may be explained by the fact that regrowing tumours are more likely to have aggressive underlying biology,
rather than reflecting an inherent risk associated
with delayed surgery.
Further evidence supporting the importance
of tumour biology—rather than timing of surgery—in determining oncological outcomes was provided
by the OPRA trial, which demonstrated an overall
distant metastasis rate of 20%, with no significant
difference between patients who underwent TME
at restaging and those who underwent TME after
regrowth.7 Habr-Gama et al18 also reported no
survival disadvantage in patients who underwent
delayed surgery for suspected cCR compared with
those who had delayed surgery for other reasons. A
recent study by Thompson et al6 demonstrated that
clinical tumour response grade was associated with
disease-free survival, local recurrence-free survival,
distant metastasis-free survival, and overall survival;
the authors reported 3-year TME-free survival (ie,
organ preservation) rates of 77% for patients with
cCR and 40% for those with nCR.6 Taken together,
these studies support the conclusion that underlying
tumour biology, rather than timing of surgery, is the
key determinant of oncological outcomes.
Patient perspectives
Patient education is essential to ensure widespread
enrolment and adherence to the WW surveillance
schedule. Patients’ perspectives and their willingness
to accept the risk of regrowth as part of organ
preservation should be explored before WW is
offered. Gani et al19 showed that 83% of patients
would choose WW if they achieved cCR despite
higher rates of local regrowth, and 94% accepted
a local regrowth rate of 25%. In another study
examining patient and physician preferences for
non-operative management of low rectal cancer
compared with abdominoperineal resection, patients
were willing to accept a higher absolute risk of local
regrowth than physicians (20% vs 5%); they were
also willing to accept a greater absolute decrease in
overall survival (20% vs 5%).20 These findings suggest
that patients may be more willing than physicians to
accept the higher risks associated with WW.
Conclusion
In Hong Kong, WW has the potential to reduce the
number of unnecessary operations, and improve
patient quality of life. Successful adoption depends
on overcoming several challenges, including resource
constraints, robust implementation of a surveillance
programme, and comprehensive education of
both clinicians and patients. By addressing these
barriers, Hong Kong will have the opportunity to
offer more personalised treatment options for rectal
cancer, ultimately benefiting patients and the public
healthcare system.
Author contributions
All authors contributed to the concept or design of the study,
acquisition of the data, analysis or interpretation of the
data, drafting of the manuscript, and critical revision of the
manuscript for important intellectual content. All authors
had full access to the data, contributed to the study, approved
the final version for publication, and take responsibility for its
accuracy and integrity.
Conflicts of interest
All authors have disclosed no conflicts of interest.
Funding/support
This study received no specific grant from any funding agency
in the public, commercial, or not-for-profit sectors.
References
1. Habr-Gama A, Perez RO, Nadalin W, et al. Operative versus nonoperative treatment for stage 0 distal rectal cancer following chemoradiation therapy: long-term results. Ann Surg 2004;240:711-7.
Crossref
2. Banwell VC, Phillips HA, Duff MJ, et al. Five-year oncological outcomes after selective neoadjuvant radiotherapy for resectable rectal cancer. Acta Oncol 2019;58:1267-72.
Crossref
3. Cercek A, Roxburgh CS, Strombom P, et al. Adoption of total neoadjuvant therapy for locally advanced rectal cancer. JAMA Oncol 2018;4:e180071.
Crossref
4. Conroy T, Bosset JF, Etienne PL, et al. Neoadjuvant chemotherapy with FOLFIRINOX and preoperative chemoradiotherapy for patients with locally advanced rectal cancer (UNICANCER-PRODIGE 23): a multicentre, randomised, open-label, phase 3 trial. Lancet Oncol 2021;22:702-15.
Crossref
5. Bahadoer RR, Dijkstra EA, van Etten B, et al. Short-course radiotherapy followed by chemotherapy before total mesorectal excision (TME) versus preoperative chemoradiotherapy, TME, and optional adjuvant chemotherapy in locally advanced rectal cancer (RAPIDO): a randomised, open-label, phase 3 trial. Lancet Oncol 2021;22:29-42.
Crossref
6. Thompson HM, Omer DM, Lin S, et al. Organ preservation and survival by clinical response grade in patients with rectal cancer treated with total neoadjuvant therapy: a secondary analysis of the OPRA randomized clinical trial. JAMA Netw Open 2024;7:e2350903.
Crossref
7. Garcia-Aguilar J, Patil S, Gollub MJ, et al. Organ preservation in patients with rectal adenocarcinoma treated with total neoadjuvant therapy. J Clin Oncol 2022;40:2546-56.
Crossref
8. Probst CP, Becerra AZ, Aquina CT, et al. Extended intervals after neoadjuvant therapy in locally advanced rectal cancer: the key to improved tumor response and potential organ preservation. J Am Coll Surg 2015;221:430-40.
Crossref
9. Hupkens BJ, Maas M, Martens MH, et al. Organ preservation in rectal cancer after chemoradiation: should we extend the observation period in patients with a clinical near-complete response? Ann Surg Oncol 2018;25:197-203.
Crossref
10. Maas M, Lambregts DM, Nelemans PJ, et al. Assessment of clinical complete response after chemoradiation for rectal cancer with digital rectal examination, endoscopy, and MRI: selection for organ-saving treatment. Ann Surg Oncol 2015;22:3873-80.
Crossref
11. Garcia-Aguilar J, Chow OS, Smith DD, et al. Effect of adding mFOLFOX6 after neoadjuvant chemoradiation in locally advanced rectal cancer: a multicentre, phase 2 trial. Lancet Oncol 2015;16:957-66.
Crossref
12. Bahadoer RR, Peeters KC, Beets GL, et al. Watch and wait after a clinical complete response in rectal cancer patients younger than 50 years. Br J Surg 2021;109:114-20.
Crossref
13. Chapman BC, Lai SH, Friedrich T, et al. Rectal cancer: clinical and molecular predictors of a complete response to total neoadjuvant therapy. Dis Colon Rectum 2023;66:521-30.
Crossref
14. Habr-Gama A, Perez RO, Wynn G, Marks J, Kessler H, Gama-Rodrigues J. Complete clinical response after neoadjuvant chemoradiation therapy for distal rectal cancer: characterization of clinical and endoscopic findings for standardization. Dis Colon Rectum 2010;53:1692-8.
Crossref
15. van der Valk MJ, Hilling DE, Bastiaannet E, et al. Long-term outcomes of clinical complete responders after neoadjuvant treatment for rectal cancer in the International Watch & Wait Database (IWWD): an international multicentre registry study. Lancet 2018;391:2537-45.
Crossref
16. Dossa F, Chesney TR, Acuna SA, Baxter NN. A watch-and-wait approach for locally advanced rectal cancer after a clinical complete response following neoadjuvant chemoradiation: a systematic review and meta-analysis. Lancet Gastroenterol Hepatol 2017;2:501-13.
Crossref
17. Smith JJ, Strombom P, Chow OS, et al. Assessment of a watch-and-wait strategy for rectal cancer in patients with a complete response after neoadjuvant therapy. JAMA Oncol 2019;5:e185896.
Crossref
18. Habr-Gama A, Perez RO, Proscurshim I, et al. Interval between surgery and neoadjuvant chemoradiation therapy for distal rectal cancer: does delayed surgery have an impact on outcome? Int J Radiat Oncol Biol Phys 2008;71:1181-8.
Crossref
19. Gani C, Gani N, Zschaeck S, et al. Organ preservation in rectal cancer: the patients’ perspective. Front Oncol 2019;9:318.
Crossref
20. Kennedy ED, Borowiec AM, Schmocker S, et al. Patient and physician preferences for nonoperative management for low rectal cancer: is it a reasonable treatment option? Dis Colon Rectum 2018;61:1281-9.
Crossref

