Hong Kong Med J 2026;32:Epub 26 Mar 2026
© Hong Kong Academy of Medicine. CC BY-NC-ND 4.0
CASE REPORT
Pericardial effusion with right atrial angiosarcoma differentiated from aortic dissection: a case report
Haoyuan Yang, MMed1 #; Fusheng Zhang, MD2 #; Xusheng Zhang, BMed1 #; Zexu Chen, BMed3 #; Zhenqiang Xu, MMed1; Gang Zhang, MD1
1 Department of Cardiovascular Surgery, Shandong Provincial Hospital Affiliated to Shandong First Medical University, Jinan, China
2 Department of Neurology, Shandong Provincial Hospital Affiliated to Shandong First Medical University, Jinan, China
3 Department of Cardiovascular Surgery, Qilu Hospital of Shandong University, Jinan, China
Corresponding authors: Dr Zhenqiang Xu (imxzq@163.com); Dr Gang Zhang (surgeonzg@outlook.com)
Case presentation
A 62-year-old male presented to Shandong Provincial
Hospital in China in October 2024 with chest
pain. Physical examination was unremarkable, and
electrocardiography showed normal sinus rhythm
with no ST-segment depression or elevation. The
patient had a 5-year history of hypertension, with
a maximum systolic blood pressure of 170 mm Hg.
Transthoracic echocardiography revealed a small
pericardial effusion but no significant structural
cardiac abnormalities (Fig a). The patient was
transferred to the cardiac intensive care unit for
pain management and symptomatic treatment. On
admission, his systolic blood pressure gradually
decreased to around 50 mm Hg, and his heart rate
increased, with clear signs of pericardial tamponade.
Following treatment with vasopressors and fluid
resuscitation, his systolic blood pressure rose to
90 to 100 mm Hg and gradually stabilised. Follow-up
transthoracic echocardiography revealed a
significant increase in pericardial effusion, prompting
pericardiocentesis that drained approximately
700 mL of haemorrhagic fluid. Microbial cultures and
cytological examination were negative. A subsequent
echocardiogram confirmed no recurrence of the
pericardial effusion.
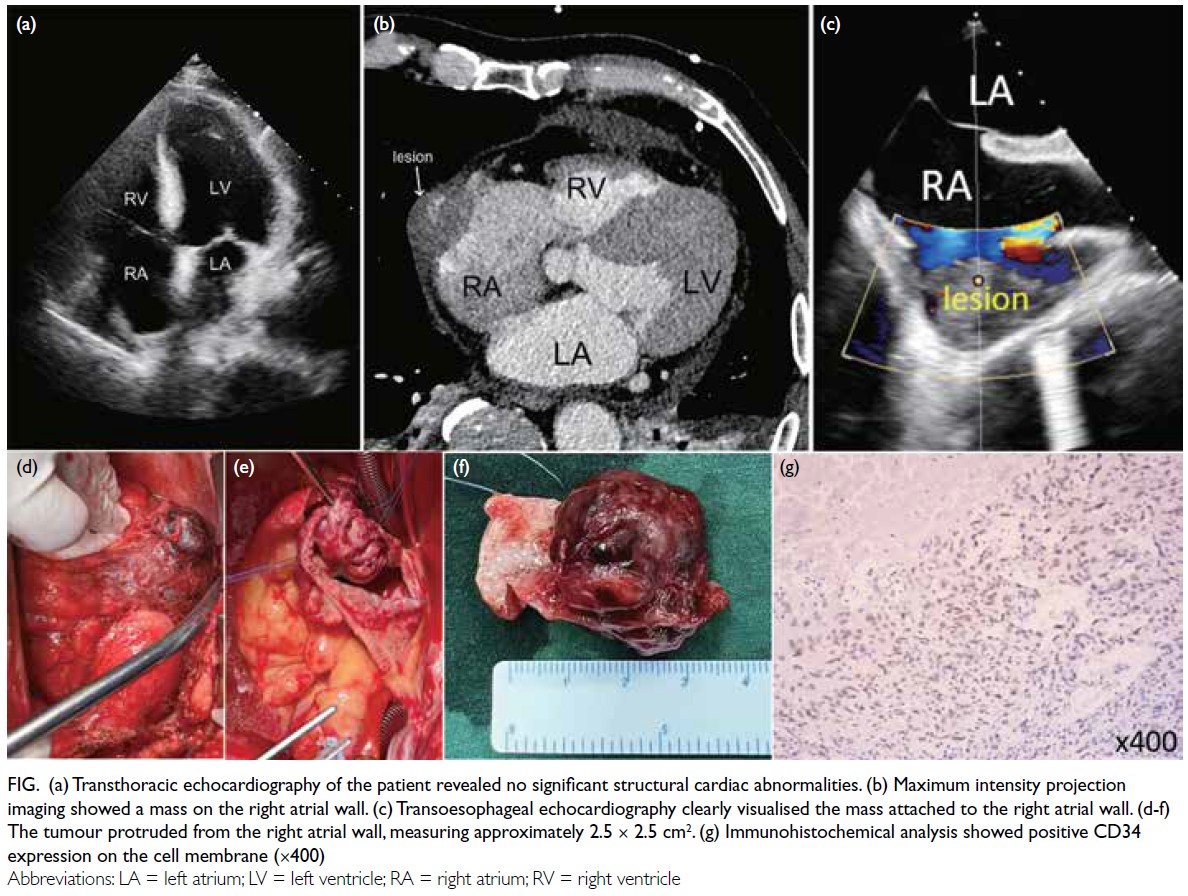
Figure. (a) Transthoracic echocardiography of the patient revealed no significant structural cardiac abnormalities. (b) Maximum intensity projection imaging showed a mass on the right atrial wall. (c) Transoesophageal echocardiography clearly visualised the mass attached to the right atrial wall. (d-f) The tumour protruded from the right atrial wall, measuring approximately 2.5 × 2.5 cm2. (g) Immunohistochemical analysis showed positive CD34 expression on the cell membrane (×400)
Given the patient’s medical history and the
initial echocardiographic findings, a preliminary
diagnosis of aortic dissection was considered.
Nonetheless a comprehensive aortic computed
tomography angiography revealed no signs of aortic
dissection or intramural haematoma. Maximum
intensity projection revealed an irregular, contrastenhancing
linear area at the lower right atrium near
the right atrial appendage, extending towards the
pericardial edge. The contrast density was reduced
and the boundary appeared indistinct, with a slightly
higher-density area within the pericardium (Fig b).
Maximum intensity projection is a high-resolution
imaging technique that effectively visualises fine
vascular structures and complex anatomical features in small volumes, making it particularly suitable for
analysing microvasculature. After multidisciplinary
discussions, the progressive pericardial effusion was
suspected to be related to an abnormality or mass
lesion in the right atrium. A decision was made to
proceed with exploratory thoracotomy.
Preoperative transoesophageal echocardiography
confirmed the presence of a mass in the right atrial
free wall (Fig c). During surgery, a 2.5 × 2.5 cm2 mass
was found protruding from the free wall of the right
atrium, infiltrating the atrial wall. The mass was
located approximately 1 cm from the atrioventricular
junction and at a sufficient distance from the superior
and inferior vena cavae (Fig d-f). The tumour was
successfully resected with a safe margin, and the
atrial wall defect repaired using a bovine pericardial
patch. Postoperative histopathological examination
confirmed the diagnosis of cardiac angiosarcoma
(Fig g). Follow-up echocardiography revealed normal
cardiac chamber structure and function. The patient
made an uneventful recovery and was discharged
on postoperative day 9 feeling well. Nonetheless,
approximately 3 months later, the patient developed
pericardial effusion again, suggesting recurrence
with possible metastatic progression. The patient did
not undergo further treatment.
Discussion
In clinical practice, chest pain associated with
acute pericardial effusion is often considered a
potential indication of aortic dissection, as was
initially suspected in this case. Nonetheless, routine
preoperative assessments for aortic dissection
revealed no structural abnormalities in the heart
or aorta, leading us to exclude this diagnosis. In
establishing a definitive diagnosis, we broadened
our differential diagnosis to include other conditions
that could cause chest pain with acute pericardial
effusion, such as cardiac tumour that is not easily
detected in clinical practice. Additionally, we
considered whether appropriate diagnostic methods had been used. Hence, we employed more specialised
and less commonly used diagnostic techniques
such as maximum intensity projection imaging and
transoesophageal echocardiography to obtain a
clearer understanding of the cardiac structures and
reach a definitive diagnosis.
The pathological diagnosis in this patient’s
tumour was angiosarcoma. Cardiac angiosarcoma,
a rare malignant cardiac tumour, accounts for
about 25% to 30% of all primary malignant cardiac
tumours.1 Most affected patients are under 65 years
of age, and the tumour most frequently originates in
the right atrium, often invading adjacent structures.2
Typical clinical manifestations include dyspnoea,
pericardial effusion, and chest pain, and these usually
appear in the advanced stages of disease, leading to a
poor prognosis.3
Conclusion
Patients with chest pain and pericardial effusion are often clinically diagnosed with aortic dissection.
Nonetheless, once initial evaluations exclude this
diagnosis, it is essential to consider cardiac tumour,
a relatively rare condition in clinical practice.
Author contributions
Concept or design: Z Xu, G Zhang.
Acquisition of data: H Yang, X Zhang.
Analysis or interpretation of data: H Yang, X Zhang, Z Chen, F Zhang.
Drafting of the manuscript: H Yang, X Zhang, Z Chen, F Zhang.
Critical revision of the manuscript for important intellectual content: Z Xu, G Zhang.
Acquisition of data: H Yang, X Zhang.
Analysis or interpretation of data: H Yang, X Zhang, Z Chen, F Zhang.
Drafting of the manuscript: H Yang, X Zhang, Z Chen, F Zhang.
Critical revision of the manuscript for important intellectual content: Z Xu, G Zhang.
All authors had full access to the data, contributed to the study, approved the final version for publication, and take responsibility for its accuracy and integrity.
Conflicts of interest
All authors have disclosed no conflicts of interest.
Funding/support
This study received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethics approval
The patient was treated in accordance with the Declaration
of Helsinki. The patient was provided with information
regarding the study and gave written informed consent for
all treatments, procedures, and publication of the case report
with the accompanying images prior to participation.
References
1. Kumari N, Bhandari S, Ishfaq A, et al. Primary cardiac
angiosarcoma: a review. Cureus 2023;15:e41947. Crossref
2. Patel SD, Peterson A, Bartczak A, et al. Primary cardiac
angiosarcoma—a review. Med Sci Monit 2014;20:103-9. Crossref
3. Chambergo-Michilot D, De la Cruz-Ku G, Sterner RM, Brañez-Condorena A, Guerra-Canchari P, Stulak J. Clinical characteristics, management, and outcomes of patients
with primary cardiac angiosarcoma: a systematic review. J
Cardiovasc Thorac Res 2023;15:1-8. Crossref

