Hong Kong Med J 2026;32:Epub 14 Apr 2026
© Hong Kong Academy of Medicine. CC BY-NC-ND 4.0
EDITORIAL
New guidance notes to drive rational prescription
of antimicrobials for community settings in Hong Kong
Edmond SK Ma, MD, FHKAM (Community Medicine)1,2; LS Ko, MB, BS2; SK Mak, FHKAM (Community Medicine)2; Martin CS Wong, MD, FHKAM (Family Medicine)3; Angus MW Chan, FHKAM (Family Medicine)4; on behalf of the
Advisory Group on Antibiotic Guidance Notes in Community Setting of the Centre for Health Protection†
1 Epidemiology Adviser, Hong Kong Medical Journal
2 Centre for Health Protection, Department of Health, Hong Kong SAR, China
3 Editor-in-Chief, Hong Kong Medical Journal
4 Immediate Past President, Hong Kong College of Family Physicians, Hong Kong SAR, China
Corresponding author: Dr Edmond SK Ma (edmond_sk_ma@dh.gov.hk)
† Members in alphabetical order: Dr Jane Chun-kwong Chan, Dr Jacky Man-chun Chan, Dr Angus Ming-wai Chan, Dr Pui-kwong Chan, Dr David Vai-kiong Chao,
Dr Hong Chen, Dr Catherine Xiao-rui Chen, Dr Yan-kit Cheung, Mr Vincent Wai-yan Chow, Dr Yat Chow, Dr Tony King-hang Ha,
Dr Pak-leung Ho, Dr Peter Ka-chung Kwan, Dr Mike Yat-wah Kwan, Dr Terence Kin-hung Kwong, Dr Kinson Kin-sang Lau,
Dr Amas Kwan-wai Leung, Dr Ada Wai-chi Lin, Dr Andrea Tin-wai Liu, Dr Leo Lui, Dr Grace Chung-yan Lui, Dr David Christopher Lung,
Prof Martin Chi-sang Wong, Prof William Chi-wai Wong, Prof Samuel Yeung-shan Wong, Prof Joyce Hoi-sze You, Ms Grace Young
Background
Antimicrobial resistance (AMR) has caused
significant mortality and morbidity globally, and
Hong Kong is no exception. It has been estimated
that from 2020 to 2030, AMR-related infections
in Hong Kong will result in 18 433 excess deaths
with a total economic cost of US$4.3 billion.1
Antimicrobial resistance is not only a problem
of resistant bacteria such as methicillin-resistant
Staphylococcus aureus (MRSA), carbapenem-resistant
Acinetobacter, vancomycin-resistant
Enterococcus and carbapenemase-producing
Enterobacterales in hospitals; rising resistance
to commonly used antibiotics has narrowed
prescribing options, leading to treatment failure
in community-acquired infections. The latest
community laboratory surveillance, conducted in
2024 by the Centre for Health Protection (CHP) of
the Department of Health, revealed that the urinary
pathogen Escherichia coli was commonly resistant
to ampicillin (67.2%), co-trimoxazole (32.2%), and
levofloxacin (36.9%), with 16.9% of specimens testing
positive for extended-spectrum beta-lactamase.2
The same surveillance programme indicated that
isolates of Streptococcus pneumoniae were resistant
to erythromycin (75.0%) and penicillin (29.2%).2 The
local threat of AMR highlights the need for robust
antibiotic stewardship. In a local study involving
19 primary care clinicians and 321 patients that
investigated help-seeking behaviour and antibiotic
prescribing for acute cough, there was a significant
difference in antibiotic prescribing rates between
private and public primary care clinicians (17.4% vs 1.6%).3 In another local study of primary care
physicians on the management of uncomplicated
urinary tract infections, the proportion of E coli
isolates matched (sensitive) to the prescribed
antibiotic (amoxicillin, ampicillin, ciprofloxacin,
co-trimoxazole, gentamicin, or nitrofurantoin) was
90.7% in the public sector and 59.2% in the private
sector, indicating that there is room for improvement
in the latter.4
The CHP has been tracking antimicrobial
supply as a proxy for consumption through
surveillance data collected from licensed wholesale
traders. Over the past decade, about half of the
antimicrobials prescribed each year have been
prescribed by private doctors in the community (Fig 1). Interestingly, a significant 27.2% reduction in the
overall defined daily dose per 1000 inhabitants per
day was observed during the three pandemic years
(2020-2022) compared with the pre–COVID-19
baseline, probably due to reduced respiratory
infections.5 Nevertheless, a rebound in defined daily
dose was noted at the start of 2023, particularly in the
private sector following the resumption of normalcy.5
Antimicrobial consumption can be grouped
according to the World Health Organization (WHO)’s
AWaRe classification—Access, Watch and Reserve—based on resistance risk and medical importance,
with the aim of improving appropriate antibiotic
use.6 According to the WHO, ‘Access’ antibiotics can
be used freely, ‘Watch’ antibiotics require caution,
and ‘Reserve’ antibiotics are reserved for last-resort
cases. The WHO has advocated increasing the use of
‘Access’ antibiotics to at least 60% of total antibiotic consumption, while preserving ‘Watch’ and ‘Reserve’
antibiotics for conditions in which they are truly
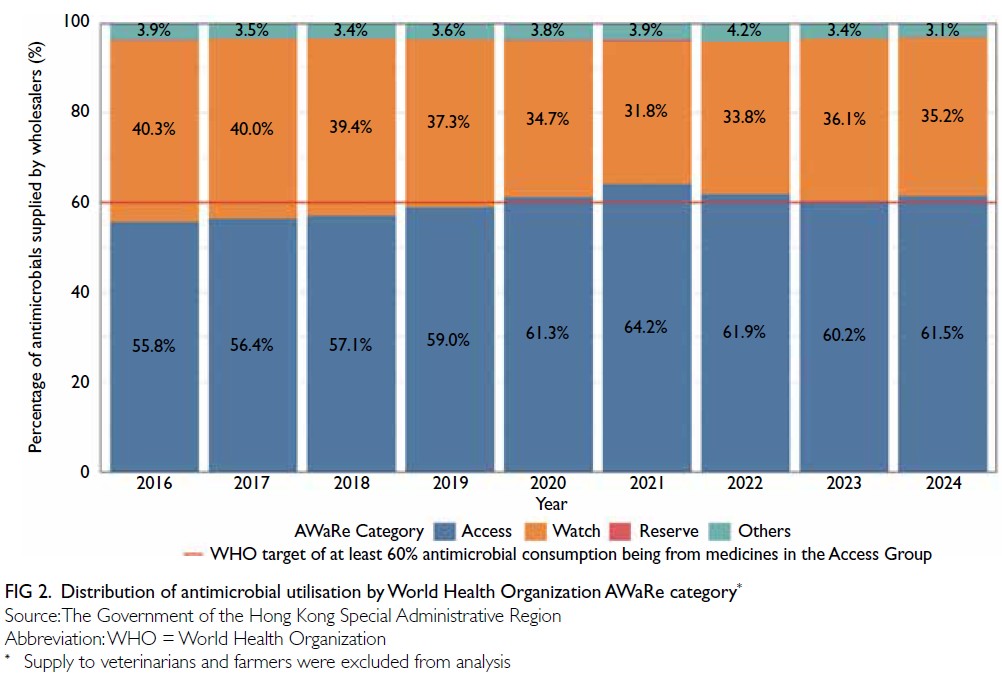
indicated.6 In Hong Kong, since 2020, the proportion
of antimicrobial use within the ‘Access’ group has met the WHO target of 60% of total consumption
(Fig 2). It is noteworthy that the proportion of
‘Access’ antibiotics prescribed is the lowest among
private doctors in the community compared with other sectors, such as the Hospital Authority and
private hospitals (Fig 3). Overuse of broad-spectrum
antibiotics is one of the main drivers of AMR and
calls for coordinated efforts to address the resistance
problem.
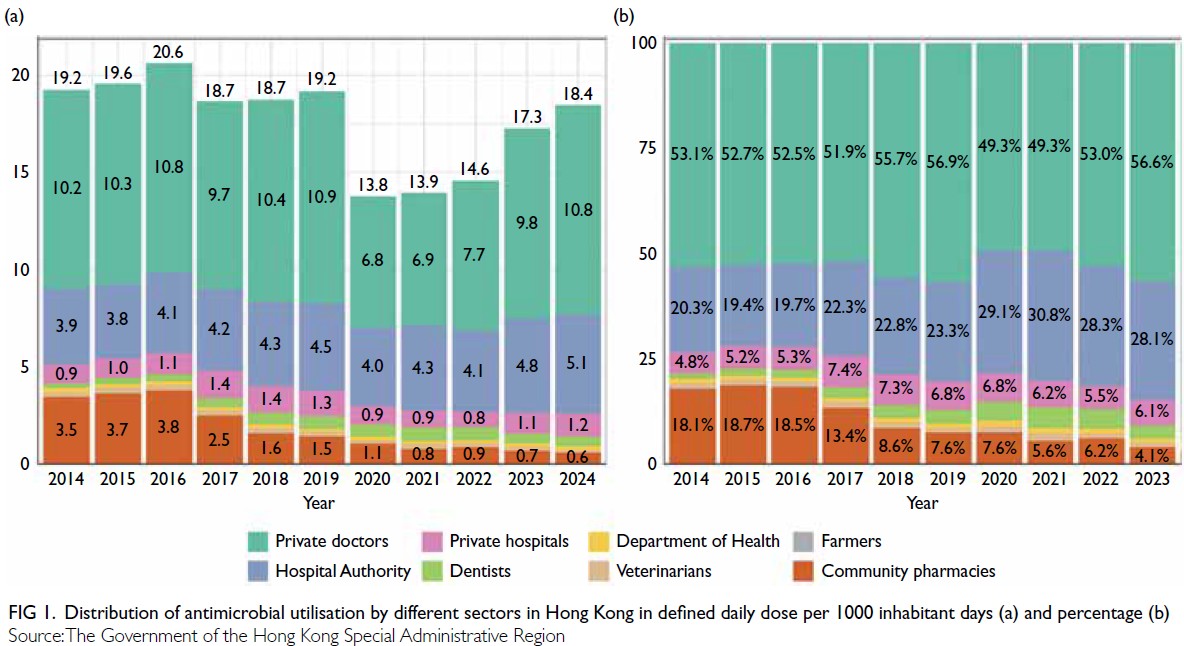
Figure 1. Distribution of antimicrobial utilisation by different sectors in Hong Kong in defined daily dose per 1000 inhabitant days (a) and percentage (b)
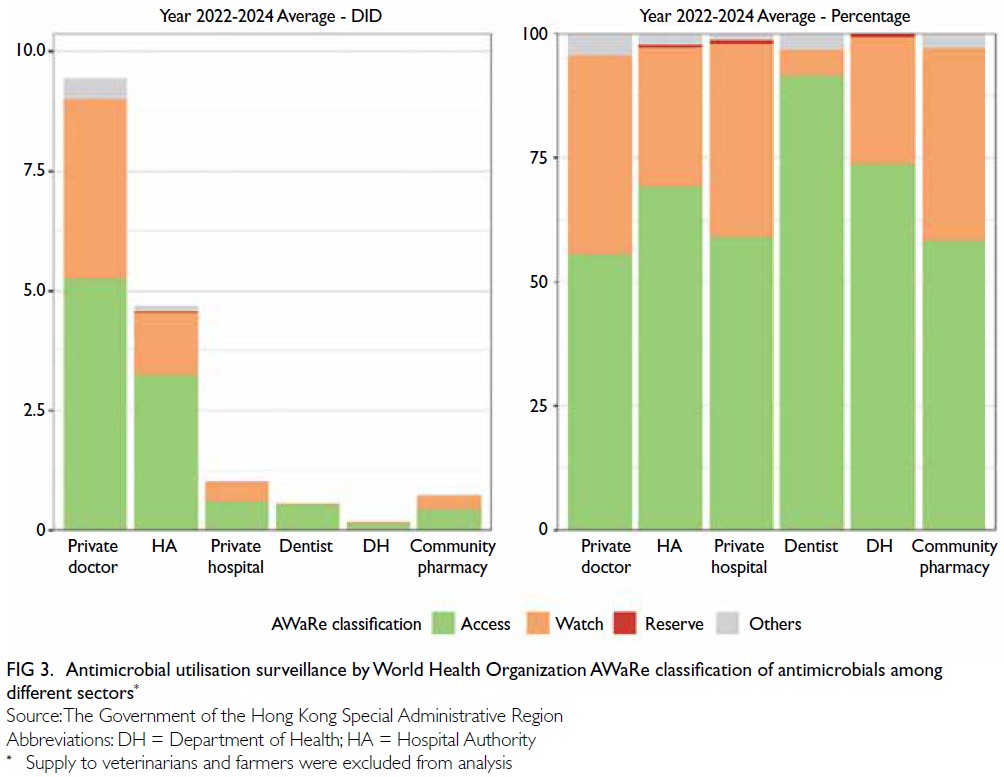
Figure 3. Antimicrobial utilisation surveillance by World Health Organization AWaRe classification of antimicrobials among different sectors
The second Hong Kong Strategy and Action
Plan on Antimicrobial Resistance (2023-2027) was
launched in 2022 to address the issue of AMR under
a One Health approach.7 Among the six key areas,
optimising the use of antimicrobials in humans
is one of the main strategic actions. In 2017 and
2018, the CHP issued guidance notes on the use
of antimicrobials for seven common conditions in
a community setting, under the leadership of an
Advisory Group on Antibiotic Guidance Notes in
Primary Care, covering acute rhinosinusitis (ARS),
acute pharyngitis, acute otitis media (AOM), acute
uncomplicated cystitis in women, community-acquired
pneumonia (CAP), acute exacerbations
of chronic obstructive pulmonary disease (COPD),
and simple (uncomplicated) skin and soft tissue
infections. With the changing epidemiology of
infectious diseases, evolving bacterial resistance
patterns, and the latest scientific evidence for the
management of different conditions, the Advisory Group has recently reviewed and updated its
guidance notes. The Advisory Group has also been
renamed the Advisory Group on Antibiotic Guidance
Notes in a Community Setting and includes new
representatives from the Hong Kong College of
Paediatricians, the Hong Kong College of Physicians,
the Hong Kong College of Otorhinolaryngologists,
and the Hong Kong Chinese Medical Association.
Similar to the previous group, the Advisory Group
also includes members from the Hong Kong Medical
Association, Hong Kong Academy of Medicine,
private medical groups, The Hong Kong Society for
Infectious Diseases, Hong Kong Doctors Union, a
representative from the Coordinating Committee
in Family Medicine of the Hospital Authority, Chief
Pharmacist’s Office of the Hospital Authority, The
Hong Kong Private Hospitals Association, and
representatives of the CHP. Five meetings were
held to deliberate on the content, from July 2024
to August 2025. In this latest edition, the Advisory
Group has revised the content of the guidance notes
with reference to international guidelines, up-to-date
scientific research, local disease epidemiology, the
latest susceptibility data from the local surveillance
network, and the availability of antibiotics in the local market. These notes serve as a key reference
to optimise the use of antibiotics in the treatment
of infections across both public and private sectors
in the community. We have extracted the relevant
content on aetiology, clinical features, and the latest
recommendations on antibiotic use for these seven
conditions. The recommended choice of antibiotics,
including first- and second-line drugs for each
condition, can be found in the online supplementary Tables 1-11. The full version of the guidance notes is
available on the CHP website: https://www.chp.gov.hk/en/features/49811.html.
Acute rhinosinusitis
Aetiology and clinical features
Rhinosinusitis refers to inflammation of the
mucosal lining of the nasal cavity, nasopharynx, and
paranasal sinuses. Acute rhinosinusitis is clinically
defined as lasting fewer than 12 weeks, whereas
rhinosinusitis that persists for 12 weeks or longer
without complete resolution of symptoms is defined
as chronic rhinosinusitis.8 Acute rhinosinusitis is
caused predominantly by viral infection, termed
acute viral rhinosinusitis or the common cold.
Adults experience approximately two to five
episodes per year, and schoolchildren seven to ten.8
When secondary bacterial infection occurs, acute
bacterial rhinosinusitis (ABRS) develops. It is more
frequent in children than in adults.9 10 11 The majority
of ARS cases are caused by viral infection, with only
about 2% complicated by bacterial infection.12 13
Streptococcus pneumoniae, Haemophilus influenzae
(non-typeable), and Moraxella catarrhalis are the
main causes of ABRS.14 Staphylococcus aureus,
streptococcal species, and anaerobes (from
odontogenic infections) may occasionally be found.15
Clinical features of rhinosinusitis include
cough, nasal symptoms, fever, halitosis, headache,
facial pain, and swelling. Cough is worse at night due
to postnasal drip. The appearance of nasal discharge
ranges from watery to purulent and cannot reliably
distinguish between bacterial and viral infection.
Fever usually resolves within 48 hours. Facial
tenderness may occur when the upper molars are
percussed or the cheekbones are pressed; this is
less common in children than in adults. Acute viral
rhinosinusitis is mostly self-limiting, typically lasting
no longer than 7 to 10 days.8 16
Management
Most ARS symptoms start to improve after 5 days,
and the majority of uncomplicated ARS cases
resolve within 2 to 3 weeks. Antibiotics are generally
not needed. Symptomatic management, such as
paracetamol, nonsteroidal anti-inflammatory
drugs, nasal decongestants, intranasal normal
saline irrigation and intranasal corticosteroids, can be considered where appropriate.8 12 17 18 19 20 Antibiotic
treatment for ABRS is only slightly beneficial. A
Cochrane review found that, out of 100 patients
treated with antibiotics, only five experienced faster
cure between days 7 and 14.20 The number needed
to benefit is 18, while the number needed to harm
is about eight.18 Antibiotic treatment causes more
adverse effects than placebo in the treatment of
ABRS.19 In addition, the use of antibiotics does
not prevent complications.21 For uncomplicated
ABRS cases, watchful waiting can be considered
after shared decision making and education about
when to return for follow-up or initiate antibiotics
(eg, if symptoms do not improve within the next
3 days or worsen rapidly or significantly at any
time).12 22 Antibiotic treatment should be reserved
for cases with features suggestive of ABRS; however,
careful patient selection is recommended to avoid
unnecessary antibiotic use and potential side-effects.
8 12
Recommended antibiotics
The recommended antibiotics for the treatment of
ABRS in adults and paediatric patients are detailed
in online supplementary Tables 1 and 2, respectively.
The first-line antibiotic is usually amoxicillin or
amoxicillin-clavulanate. The latter is a beta-lactam/beta-lactamase inhibitor combination and is
therefore active against beta-lactamase-producing
bacteria, including most H influenzae, M catarrhalis
and methicillin-sensitive S aureus. It has no added
advantage against S pneumoniae, whose beta-lactam
resistance does not rely on enzyme production. For
patients with type I hypersensitivity to penicillin,
antibiotics from a completely different class should
be used, such as doxycycline or macrolides. If
macrolides (eg, clarithromycin, erythromycin) are
prescribed, follow-up after the initial course of
treatment is recommended because of the relatively
high rate of antibiotic resistance. A 7-day course
of antibiotics is sufficient to treat acute sinusitis in
both adults and children. This takes into account
the overall evidence on efficacy and safety, as well
as the risk of AMR.12 A meta-analysis comparing
short-course treatment (3-7 days) with long-course
treatment (6-10 days) found no significant difference
in cure rate or symptom improvement.23
Acute pharyngitis
Aetiology and clinical features
Acute pharyngitis, or acute sore throat, is a mostly
self-limiting disease and usually lasts for around
1 week. Although its aetiology can be viral or
bacterial, most cases are viral and antibiotics are
inappropriate. Viral pharyngitis can be caused by
enterovirus, rhinovirus, influenza or parainfluenza
virus, coronavirus (including severe acute respiratory syndrome coronavirus 2), adenovirus, respiratory
syncytial virus, Epstein–Barr virus, herpes simplex
virus, metapneumovirus, cytomegalovirus, and
human immunodeficiency virus. Patients with acute
sore throat and associated signs and symptoms
such as conjunctivitis, coryza, cough, diarrhoea,
hoarseness, discrete ulcerative stomatitis and/or viral
exanthema are more likely to have a viral illness.24 25
Conversely, symptoms such as sudden-onset sore
throat, fever, and/or pain on swallowing, and
physical examination findings such as pharyngeal
and tonsillar erythema, an erythematous sandpaper-like
rash, tonsillar hypertrophy with or without
exudates, palatal petechiae, and/or anterior cervical
lymphadenopathy are more suggestive of a bacterial
cause.25 26 Group A streptococcus (GAS) is the
most common bacterial cause of acute pharyngitis,
accounting for about 80% of bacterial cases, with
the remainder usually caused by group C or group G
streptococci. Group A streptococcus is responsible
for 5% to 15% of sore throat consultations in adults
and 20% to 30% in children.27 28 29 Although symptoms
of GAS pharyngitis resolve without antibiotic
treatment, complications can arise and may be
suppurative (eg, cervical lymphadenitis, peritonsillar
abscess, mastoiditis, and retropharyngeal abscess) or
non-suppurative (eg, scarlet fever, acute rheumatic
fever, and post-streptococcal glomerulonephritis).25
Management and antibiotic treatment
Although GAS pharyngitis is mostly self-limiting,
antibiotics are prescribed to relieve acute symptoms,
prevention of acute and subacute complications,
and reduce transmission. Antibiotic treatment
can prevent suppurative complications and acute
rheumatic fever, and may offer protection against
the subsequent development of post-infectious
glomerulonephritis.30 Group A streptococcus is
generally sensitive to penicillin and other members
of the beta-lactam group of antibiotics, but shows
high resistance (42.3% to 60.0% from 2016 to 2020)
to erythromycin locally.31
Penicillin V or amoxicillin is the recommended
drug of choice for patients who are not allergic to
these agents (online supplementary Tables 3 and 4). Group A streptococcus resistant to penicillins
and other beta-lactams has not been reported. All
Streptococcus pyogenes isolates tested by the CHP
from 2008 to 2020 were sensitive to penicillin.31
First-generation cephalosporins (eg, cephalexin) are
the first-line agents for penicillin-allergic individuals
(ie, those without anaphylactic reactions). Other
cephalosporins (eg, cefaclor, cefuroxime) are
alternatives but are not favoured as first-line agents
because of their broader spectrum of activity. As
GAS resistance to macrolides (eg, erythromycin,
azithromycin, and clarithromycin) is known to
be common in Hong Kong, macrolides are not an appropriate first-choice antibiotic treatment.31
Regarding the duration of antibiotics, a 10-day
course is recommended by the Infectious Diseases
Society of America and the United States Centers for
Disease Control and Prevention to achieve maximal
eradication of GAS from the pharynx for the
primary prevention of acute rheumatic fever.25 The
National Institute for Health and Care Excellence
guideline recommends treatment for 5 to 10 days but
recognises that microbiological cure may be better
with a 10-day course of phenoxymethylpenicillin
compared with a 5- or 7-day course, although there
were no differences in relapse or recurrence.32 Since
routine rapid antigen detection testing for GAS is
not recommended and microbiological cure is the
goal, a 10-day course is recommended to maximise
treatment effectiveness.
Acute otitis media
Aetiology and clinical features
Acute otitis media is the acute inflammation of the
middle ear. It is a common paediatric condition
with peak prevalence at 6 to 18 months, while
AOM in adults is rare.33 It has been reported that
27% of infants and 37% of children with upper
respiratory tract infections develop AOM.34 35 After
the introduction of pneumococcal vaccination,
overseas studies showed that the incidence of AOM
decreased significantly.36 37 Acute otitis media can be
caused by viruses or bacteria, but it is often difficult
to distinguish between them as both can co-exist.
Viruses that cause upper respiratory tract infections
(eg, respiratory syncytial virus, adenovirus and
influenza viruses) are present in up to two-thirds
of cases.38 The average global distribution of
causative bacterial pathogens of AOM is as follows:
S pneumoniae (30%), H influenzae (non-typeable)
[23%], and M catarrhalis (5%).39 The remaining
cases are caused by other bacteria (eg, GAS). There
is usually a single bacterial cause, but coinfection
with other pathogens is known to occur.40 Typical
symptoms of AOM include otalgia that interferes
with normal activity or sleep, new-onset ear
discharge, fever, loss of appetite and difficulty
hearing. It may present as ear tugging or irritability
in infants and young children.
Management
Viral AOM is a mostly self-limiting infection, with
symptoms (ie, otalgia) typically lasting about 3 to
7 days.41 Most children and young people recover
within 3 days without antibiotics. In a Cochrane
review, 60% of children not treated with antibiotics
showed improvement in symptoms within 24
hours, and over 80% had symptoms that resolved
spontaneously within 3 days.42 When determining
whether to prescribe antibiotics, healthcare providers should consider the patient’s general health, the
severity of the disease, the risk of complications, and
the expected benefits of antibiotic therapy:
If the patient is not improving within 48 to 72
hours and has acute, worsening symptoms; is
systemically very unwell; has signs and symptoms
of a more serious illness or condition; or is at
high risk of complications, clinicians should
offer immediate antibiotics. The patient should
be referred to hospital if there is severe systemic
infection or complication (eg, mastoiditis,
meningitis, or facial nerve paralysis).
Red flag signs and symptoms include a fever
of 39°C or above, drowsiness, rapid breathing,
rapid heart rate, severe ear pain, and signs or
symptoms of intracranial complications (eg, neck
stiffness, altered mental status, seizures, or focal
neurological deficits).
Children under 2 years of age with bilateral
AOM, and children and young people with AOM
and otorrhoea, are more likely to benefit from
antibiotics.41
Antibiotic treatment has no early effect on
pain, a slight effect over the following days, and
only a modest effect on the number of children
with tympanic perforations, contralateral otitis
episodes and abnormal tympanometry findings in
subsequent weeks, with no difference in the rare
occurrence of severe complications.42 A Cochrane
review found that for patients with respiratory
infections (including AOM) in whom clinicians
considered it safe not to prescribe antibiotics
immediately, a non-prescribing approach with
advice to return if symptoms did not resolve (delayed
antibiotics) resulted in the least antibiotic use, while
maintaining similar patient satisfaction and clinical
outcomes compared with immediate antibiotics.43
The recommended choice of antibiotic regimen is
detailed in online supplementary Table 5. Regardless
of whether antibiotics are prescribed, patients
and caregivers should be informed about red flag
symptoms and advised to seek medical attention if
symptoms worsen rapidly.
Community-acquired pneumonia
Aetiology and clinical features
Community-acquired pneumonia refers to an acute
infection of the lung parenchyma in a patient who
has acquired the infection outside a healthcare
setting and has developed symptoms and signs in
the community. Community-acquired pneumonia
can be caused by a variety of pathogens, including
viruses and bacteria. There is increasing recognition
of viral pathogens in CAP4 of which the most
common include influenza virus, rhinovirus and
parainfluenza virus. The most frequently detected
bacterial pathogens are S pneumoniae, H influenzae, S aureus, and Mycoplasma pneumoniae.44 45 46 47 Group
A streptococcus and S aureus may cause secondary
bacterial pneumonia following influenza virus
infection. Common clinical features of CAP include
cough, fever, pleuritic chest pain, dyspnoea, and
sputum production. On physical examination,
many patients are febrile, although this finding is
frequently absent in older patients. Tachypnoea and
tachycardia are also common. Chest examination
may reveal audible crackles. Signs of consolidation,
such as decreased or bronchial breath sounds and
dullness to percussion, may be present.
Antibiotic therapy
Antibiotic therapy should be started as soon
as CAP is suspected or established48 49 50 51 52 53 (online supplementary Tables 6 and 7). When considering
the choice of antibiotic, clinicians are advised to take
into account the severity of the infection and the
risk of developing complications (eg, co-morbidities
such as severe lung disease or immunosuppression),
local AMR patterns and prevalence, as well as any
recent antibiotic use and microbiological results, if
available.54
Streptococcus pneumoniae is one of the most
common pathogens identified in local CAP.44 In
Hong Kong, there is reduced susceptibility of S pneumoniae to penicillin (23% to 51% resistance)
and to macrolides (82% resistance to erythromycin)
in the community.55 56 Risk factors include age over 65
years, beta-lactam therapy within the last 3 months,
alcoholism, multiple medical co-morbidities, and
exposure to a child in a day-care centre. Amoxicillin-clavulanate
is therefore recommended as the first-line
empirical treatment. Doxycycline can be added
if macrolide-resistant M pneumoniae infection is
suspected. For patients with co-morbidities or those
at risk of Legionella pneumonia, a macrolide can
be added. Due to poor intrinsic activity against S pneumoniae and/or low oral bioavailability, certain
oral cephalosporins (first-generation agents, cefaclor,
cefuroxime, ceftibuten, cefixime and loracarbef) are
not recommended.
Mycoplasma pneumoniae is common in
children and is also seen in adults in Hong Kong.44 57
The infection is often self-limiting without specific
antibiotic therapy. Initial empirical therapy covering
M pneumoniae is considered optional for outpatients
with mild CAP. It may be indicated if the first-line
agent has failed, if outpatients have severe CAP,
or if the patient is a child aged over 5 years or an
adolescent. Up to 40% of CAP in children aged 5 years
or above has been attributed to M pneumoniae.57 In
Hong Kong, the macrolide resistance rate among
M pneumoniae is high in the community, with
an increasing trend from 28.2% in 2018 to 61.3%
in 2024.58 Doxycycline is recommended for the
treatment of macrolide-resistant M pneumoniae–associated CAP in adults and children (regardless of
age or duration of therapy).59
Fluoroquinolones may be considered in the
treatment of CAP when the first-line agent has
failed, when an outpatient is allergic to first-line
agents, or when there is documented infection with
S pneumoniae with a penicillin minimum inhibitory
concentration (MIC) of 4 μg/mL or above (intermediate
susceptibility to penicillin). Nonetheless, excessive
use of respiratory fluoroquinolones in CAP may lead
to delayed diagnosis of tuberculosis and increased
fluoroquinolone resistance in Mycobacterium
tuberculosis. Fluoroquinolones should be reserved
for use in outpatients who have no other treatment
options. Patients should be warned of the risk of
severe adverse effects, including aortic dissection or
rupture of an aortic aneurysm, significant decreases
in blood sugar, and disabling side-effects involving
the tendons, muscles, joints, nerves, central nervous
system, and mental health.60 61 62
Duration of antibiotic therapy
Most outpatients with CAP will show an adequate
clinical response within 72 hours. For most patients,
appropriately chosen initial antibiotic therapy should
not be changed within the first 72 hours unless there
is marked clinical deterioration. Clinical judgement
is required when determining the duration of
antibiotic therapy. Factors to consider include the
patient’s clinical response, severity of infection,
causative pathogen, in vitro susceptibility of the
pathogen, and the presence of complications and
side-effects. In adults and children, a 5-day course
of antibiotics (except for doxycycline) is usually
effective for mild CAP in the outpatient setting.54 63 64 65 66
Clinicians may consider stopping treatment after 5
days unless the patient fails to improve clinically or
the microbiological results suggest the need for a
longer course.67
Acute exacerbations of chronic obstructive
pulmonary disease
Clinical features and causes of exacerbation
Chronic obstructive pulmonary disease is a
heterogeneous lung condition characterised by
chronic respiratory symptoms due to airway
and/or alveolar abnormalities. It is caused by a
combination of environmental (eg, passive smoking,
outdoor and indoor air pollution, occupational
exposure to airborne pollutants) and host factors
(eg, smoking and advancing age).68 69 In Hong Kong,
the prevalence of COPD is 0.5% among individuals
aged 15 years or above.70 It is most common among
those aged 75 to 84 years (2.2%), with a male
predominance.70 The most common respiratory symptoms include dyspnoea, cough, and/or sputum
production. Chronic obstructive pulmonary disease is diagnosed by spirometry demonstrating a postbronchodilator
ratio of FEV1/FVC (forced expiratory
volume in 1 second to forced vital capacity) of
<0.7. The disease is associated with co-morbidities
such as cardiovascular disease, hypertension, and
lung cancer.71 72 73 It may be punctuated by acute
exacerbations, defined as acute episodes of worsening
respiratory symptoms within 14 days that may be
accompanied by tachypnoea and/or tachycardia,
and are often associated with local and systemic
inflammation.74 Acute exacerbations of COPD are
mainly triggered by respiratory viral infection (eg,
influenza A and rhinovirus), although bacterial
infection and air pollution can also precipitate these
events.74 75 76 77 Common bacterial isolates in patients
hospitalised with a COPD exacerbation include H
influenzae, S pneumoniae, Pseudomonas aeruginosa,
and M catarrhalis.76 78 79 80
When to prescribe antibiotics and choice of
antibiotics
Appropriately prescribed antibiotics may shorten
recovery time and reduce the risk of early relapse,
treatment failure, and duration of hospitalisation.
Antibiotics can be prescribed when there are clinical
signs of bacterial infection. Evidence suggests
that sputum colour and purulence can predict the
presence of bacterial infection. In a pooled analysis,
green or yellow sputum showed a sensitivity of 94.7%
and a specificity of 15% for the presence of bacteria.81
Studies have also shown that a positive bacterial
culture was obtained in 77% to 84% of patients with
purulent sputum.82 83 According to the 2024 Global
Strategy for Prevention, Diagnosis and Management
of COPD report, antibiotics should be given to
patients in the community if they: (a) have three
cardinal symptoms, namely increased dyspnoea,
increased sputum volume, and increased sputum
purulence; (b) have increased sputum purulence
and one other cardinal symptom; or (c) require
mechanical ventilation.74
Empirical antibiotic therapy (online supplementary Table 8) targets likely bacterial
pathogens responsible for acute exacerbations of
COPD and takes into account local patterns of
antibiotic resistance.56 Pseudomonas aeruginosa
and/or Enterobacterales infection may occur in
outpatients with advanced COPD. Risk factors for
P aeruginosa infection include chronic colonisation
or previous isolation of P aeruginosa from sputum,
very severe COPD (forced expiratory volume in 1
second <30% predicted), bronchiectasis on chest
imaging, broad-spectrum antibiotic use within the
past 3 months, and chronic systemic glucocorticoid
use.84 85 86 87 Amoxicillin and macrolides are not
recommended because of the high resistance rates
in Hong Kong. Local community data show reduced
susceptibility of S pneumoniae to penicillin (23%-51% resistance) and to macrolides (82% resistance to
erythromycin).55 56 In addition, 50% of H influenzae
isolates were resistant to ampicillin, and nearly
all (99%) M catarrhalis isolates produced beta-lactamase.56 Amoxicillin-clavulanate or a respiratory
fluoroquinolone (eg, levofloxacin) is recommended.
In patients for whom amoxicillin-clavulanate is
contraindicated because of non-type I penicillin
allergy, a cephalosporin such as cefpodoxime or
cefuroxime may be considered. Fluoroquinolones
should be reserved for outpatients who have no other
treatment options for acute bacterial exacerbation
of chronic bronchitis because of the risk of severe
adverse effects, including aortic dissection or rupture
of an aortic aneurysm, significant decreases in blood
sugar, or disabling side-effects involving the tendons,
muscles, joints, nerves, central nervous system and
mental health.60 61 62 Regarding treatment duration,
a systematic review of outpatients with COPD
exacerbations indicated that short-course antibiotic
treatment (≤5 days) did not differ significantly
from long-course treatment (≥6 days) in terms of
clinical cure or bacterial eradication.88 These results
concurred with those of another systematic review
and meta-analysis comparing short-course (<6 days)
with long-course antibiotics (>7 days).89 In addition,
there were significantly fewer adverse events with
short-course antibiotics.88 89 90 Based on the evidence, a
5-day course of antibiotics will generally be adequate
to treat a mild-to-moderate acute exacerbation of
COPD due to bacterial infection.
Acute uncomplicated cystitis in women
Aetiology and clinical features
Acute uncomplicated cystitis is characterised by
local bladder signs and symptoms such as dysuria,
urgency, frequency and suprapubic pain. There
should be no signs or symptoms suggestive of
infection spreading beyond the bladder (eg, fever,
chills, rigors, unstable vital signs, flank pain or
costovertebral angle tenderness). Individuals
with urinary catheters are excluded from this
definition.91 92 93 94 95 96 97 Cystitis usually occurs when bacteria
from the gastrointestinal tract enter the urethra and
ascend to the bladder.98 Escherichia coli is the most
commonly isolated pathogen (~52%) in midstream
urine samples collected in the outpatient setting
of the Hospital Authority, followed by Klebsiella
pneumoniae (~9%), Proteus mirabilis (~5%), and
Streptococcus agalactiae (~3%) [unpublished data
from CHP].
Antibiotic therapy
Given the very high probability of urinary tract
infection based on typical symptoms, clinicians can
consider empirical treatment without urine culture
or dipstick urinalysis. The choice of antibiotics should take into account the symptoms, potential
complications, previous urine culture results, and
local antibiotic susceptibility patterns.98 Among
the E coli isolated from urine samples in outpatient
settings of the Department of Health56 and Hospital
Authority (unpublished data), 64% to 67% were
resistant to ampicillin, 36% to 46% to levofloxacin,
20% to cefpodoxime, 39% to cefuroxime, 31% to
32% to co-trimoxazole, 6% to 16% to amoxicillin-clavulanate,
2% to fosfomycin and 1% to 2%
to nitrofurantoin. In the same settings, 99% to
100% of Klebsiella pneumoniae were resistant to
ampicillin, 23% to 42% to nitrofurantoin, 12% to
cefpodoxime, 35% to cefuroxime, 15% to 20% to
co-trimoxazole, 10% to 17% to levofloxacin, and
8% to 14% to amoxicillin-clavulanate.56 Judicious
use of antibiotics is recommended to minimise
potential collateral damage (ecological adverse
effects of antimicrobial therapy, such as colonisation
or infection with multidrug-resistant organisms),
particularly with broad-spectrum cephalosporins
and fluoroquinolones.97
For the choice of antibiotic therapy (online supplementary Table 9), nitrofurantoin is appropriate
because of the low local resistance rate and is less likely
to select for drug-resistant organisms (the preserved
in vitro susceptibility of E coli to nitrofurantoin over
many years of use suggests that it causes only minor
collateral damage). Beta-lactam agents, including
amoxicillin-clavulanate, are appropriate choices for
therapy even in cases of intermediate susceptibility
because they achieve high urinary concentrations.
In view of disabling and potentially long-lasting or
irreversible side-effects, fluoroquinolones should
be used only when other commonly prescribed
antibiotics are considered inappropriate. Co-trimoxazole
is not recommended as a first-line agent
given the high local resistance.56 Antibiotic treatment
is not required for asymptomatic bacteriuria except
during pregnancy or prior to urological procedures
associated with mucosal trauma.91 95 99 100
Simple (uncomplicated) skin and soft tissue
infections
Aetiology and clinical features
The term ‘skin and soft-tissue infections (SSTIs)’
describes a wide variety of clinical conditions.
Simple, or uncomplicated, SSTIs refer to superficial
infections such as cellulitis, simple abscesses,
impetigo and furuncles, and require antibiotics
or surgical incision and drainage. Complicated
SSTIs include deep soft-tissue infections (eg, deep
abscesses and necrotising fasciitis) that require
significant surgical intervention. When classifying
patients with SSTIs, the necrotising or non-necrotising
nature of the infection, the anatomical
extent, the characteristics of the infection (purulent or non-purulent), and the clinical condition of the
patient should always be assessed independently.101 102
Simple SSTIs usually present with localised clinical
findings such as erythema, warmth, oedema and
pain over the affected site. They are not associated
with systemic signs or symptoms that indicate spread
(eg, fever, tachycardia, diaphoresis, fatigue, anorexia
and vomiting) or uncontrolled co-morbidities that
may complicate treatment. Simple SSTIs are usually
monomicrobial, mainly caused by S aureus and
beta-haemolytic streptococci such as S pyogenes. In
diabetic foot infection, polymicrobial infection is
more likely. Vibrio vulnificus infection is associated
with injuries related to seawater or seafood exposure.
Impetigo is usually caused by S aureus, whereas
cellulitis is usually caused by beta-haemolytic
streptococci. Nonetheless, both pathogens may
occur in combination in simple SSTIs.
Antibiotic therapy
Simple SSTIs are amenable to outpatient
management with topical or oral antibiotics.
When choosing an empirical antibiotic (online supplementary Tables 10 and 11), clinicians
should consider the severity of symptoms, site of
infection, risk of uncommon pathogens, previous
microbiological results, MRSA status, and local
resistance patterns.103 In mild and localised impetigo,
topical antibiotics are adequate treatment.104 105
In other cases of simple SSTIs, oral antibiotics are
indicated. Based on data from outpatient settings
of the Hospital Authority, resistance of S pyogenes
to penicillins and other beta-lactams has not been
reported in Hong Kong; nonetheless, 37% of isolates
were resistant to erythromycin (unpublished data
from CHP). Coverage for community-associated
MRSA (CA-MRSA) should be considered if risk
factors are present (eg, history of direct contact
with CA-MRSA–infected wounds, discharge or
soiled areas, close contact with carriers, presence of
skin lesions, poor personal hygiene, and sharing of
personal items), or if the patient does not respond
to first-line treatment.106 107 Co-trimoxazole,
doxycycline/minocycline, and clindamycin can be
considered if CA-MRSA is suspected or confirmed.
Locally, 24% to 26% of S aureus isolates are MRSA.56
In addition, patients with CA-MRSA and their
close contacts should receive topical decolonisation
therapy.107
Superficial and small abscesses usually
respond well to incision and drainage and seldom
require antibiotics, except when they are associated
with systemic signs of infection, extensive cellulitis,
rapid progression or poor response to initial
drainage; involve sites that are difficult to drain (eg,
face, hands, and genitalia); or occur in children,
older adults, or those with significant co-morbid
illness or immunosuppression.102 A 5- to 7-day course of antibiotic treatment is recommended for
simple SSTIs, but this may be extended to up to
10 days at the clinician’s discretion if the infection
does not improve after completion of the initial
course.103 106 108 109 110 111 Since the skin requires time to
return to its normal condition, full resolution should
not be expected within 5 to 7 days.103
Conclusion
Rational prescription of antimicrobials is vital to
curb the rise of resistant pathogens. At the 79th
United Nations General Assembly High-Level
Meeting on AMR held in September 2024, global
leaders approved a political declaration committing
to a clear set of targets and actions, including a 10%
reduction in the estimated 4.95 million annual human
deaths associated with bacterial AMR by 2030.112 The
declaration also aims for at least 70% of antibiotics
used in human health worldwide to belong to the
WHO Access group.112 A territory-wide survey
was conducted in 2023 to examine the awareness
and practices of the general public regarding AMR
in Hong Kong.113 The results showed that when a
doctor’s initial assessment indicated that antibiotics
were not needed, the vast majority of respondents
(94.7%) accepted the doctor’s advice to observe for
a few more days or to wait for diagnostic test results
before deciding whether to prescribe antibiotics.113
In addition, about half (49.5%) of respondents
wanted doctors to share decision making with them
regarding antibiotic prescriptions.113 We urge all
doctors in both the public and private sectors to
prescribe antibiotics only when clinically indicated
and to refer to clinical guidelines and the current
guidance notes when selecting an appropriate agent.
Whenever possible, narrow-spectrum antibiotics
should be used at optimal doses and for the shortest
effective duration.
Author contributions
All authors contributed to the editorial, approved the final
version for publication, and take responsibility for its accuracy
and integrity.
Conflicts of interest
ESK Ma and MCS Wong are members of the Hong Kong Medical Journal Editorial Board and internal review of this editorial was independently conducted by a senior editor.
Other authors have declared no conflicts of interest.
Funding/support
This editorial received no specific grant from any funding
agency in the public, commercial, or not-for-profit sectors.
Supplementary material
The supplementary material was provided by the authors,
and some information may not have been peer reviewed.
Accepted supplementary material will be published as submitted by the authors, without any editing or formatting.
Any opinions or recommendations discussed are solely those
of the author(s) and are not endorsed by the Hong Kong
Academy of Medicine or the Hong Kong Medical Association.
The Hong Kong Academy of Medicine and the Hong Kong
Medical Association disclaim all liability and responsibility
arising from any reliance placed on the content.
References
1. World Health Organization. Health and economic
impacts of antimicrobial resistance in the Western Pacific
Region, 2020-2030. 2023 June 13. Available from: https://www.who.int/publications/i/item/9789290620112.
Accessed 31 Dec 2025.
2. Centre for Health Protection, Department of Health,
Hong Kong SAR Government. Antimicrobial resistance
surveillance in human–community setting. Available
from: https://www.chp.gov.hk/en/static/101604.html.
Accessed 19 Jan 2026.
3. Wong CK, Liu Z, Butler CC, et al. Help-seeking and
antibiotic prescribing for acute cough in a Chinese primary
care population: a prospective multicentre observational
study. NPJ Prim Care Respir Med 2016;26:15080. Crossref
4. Wong CK, Kung K, Au-Doung PL, et al. Antibiotic
resistance rates and physician antibiotic prescription
patterns of uncomplicated urinary tract infections
in southern Chinese primary care. PloS One
2017;12:e0177266. Crossref
5. Ma ES, Hsu E, Chow V, et al. Rebound of antibiotic use
and respiratory infections after resumption of normalcy
from COVID-19 in Hong Kong. Infect Drug Resist
2025;18:1325-37. Crossref
6. World Health Organization. AWaRe classification of
antibiotics for evaluation and monitoring of use, 2023.
2023 July 26. Available from: https://www.who.int/publications/i/item/WHO-MHP-HPS-EML-2023.04. Accessed 16 Mar 2026.
7. Ma ES. Combating antimicrobial resistance in Hong
Kong: where are we and where should we go? Hong Kong
Med J 2022;28:424-6. Crossref
8. Fokkens WJ, Lund VJ, Hopkins C, et al. European position
paper on rhinosinusitis and nasal polyps 2020. Rhinology
2020;58(Suppl S29):1-464. Crossref
9. Wald ER, Nash D, Eickhoff J. Effectiveness of amoxicillin/clavulanate potassium in the treatment of acute bacterial sinusitis in children. Pediatrics 2009;124:9-15. Crossref
10. DeMuri GP, Gern JE, Moyer SC, Lindstrom MJ, Lynch SV,
Wald ER. Clinical features, virus identification, and
sinusitis as a complication of upper respiratory tract
illness in children ages 4-7 years. J Pediatr 2016;171:133-9.e1. Crossref
11. Marom T, Alvarez-Fernandez PE, Jennings K, Patel JA, McCormick DP, Chonmaitree T. Acute bacterial sinusitis
complicating viral upper respiratory tract infection in
young children. Pediatr Infect Dis J 2014;33:803-8. Crossref
12. National Institute for Health and Care Excellence.
Sinusitis (acute): antimicrobial prescribing. 2017 October
27. Available from: https://www.nice.org.uk/guidance/ng79. Accessed 25 Mar 2025.
13. Gwaltney JM Jr, Wiesinger BA, Patrie JT. Acute community-acquired bacterial sinusitis: the value of
antimicrobial treatment and the natural history. Clin
Infect Dis 2004;38:227-33. Crossref
14. Chow AW, Benninger MS, Brook I, et al. IDSA clinical
practice guideline for acute bacterial rhinosinusitis in
children and adults. Clin Infect Dis 2012;54:e72-112. Crossref
15. Bennett JE, Dolin R, Blaser MJ. Mandell, Douglas and
Bennett’s Infectious Disease Essentials. 8th ed. Elsevier
Health Sciences; 2016.
16. Desrosiers M, Evans GA, Keith PK, et al. Canadian clinical
practice guidelines for acute and chronic rhinosinusitis [in
English, French]. J Otolaryngol Head Neck Surg 2011;40
Suppl 2:S99-193.
17. Aring AM, Chan MM. Current concepts in adult acute
rhinosinusitis. Am Fam Physician 2016;94:97-105.
18. Smith MJ. Evidence for the diagnosis and treatment of
acute uncomplicated sinusitis in children: a systematic
review. Pediatrics 2013;132:e284-96. Crossref
19. King D, Mitchell B, Williams CP, Spurling GK. Saline nasal
irrigation for acute upper respiratory tract infections.
Cochrane Database Syst Rev 2015;2015:CD006821. Crossref
20. Segboer C, Gevorgyan, A, Avdeeva, K, et al. Intranasal
corticosteroids for non-allergic rhinitis. Cochrane
Database Syst Rev 2019;2019:CD010592. Crossref
21. Hansen FS, Hoffmans R, Georgalas C, Fokkens WJ.
Complications of acute rhinosinusitis in the Netherlands.
Fam Pract 2012;29:147-53. Crossref
22. Rosenfeld RM, Piccirillo JF, Chandrasekhar SS, et al.
Clinical practice guideline (update): adult sinusitis.
Otolaryngol Head Neck Surg 2015;152(2 Suppl):S1-39. Crossref
23. Falagas ME, Karageorgopoulos DE, Grammatikos AP,
Matthaiou DK. Effectiveness and safety of short vs. long
duration of antibiotic therapy for acute bacterial sinusitis:
a meta-analysis of randomized trials. Br J Clin Pharmacol
2009;67:161-71. Crossref
24. Chan JY, Yau F, Cheng F, Chan D, Chan B, Kwan M.
Practice recommendation for the management of
acute pharyngitis. Hong Kong J Paediatr (new series)
2015;20:156-62.
25. Centers for Disease Control and Prevention, United States
Government. Clinical guidance for group A streptococcal
pharyngitis. 2025 November 18. Available from: https://www.cdc.gov/group-a-strep/hcp/clinical-guidance/strep-throat.html. Accessed 17 Oct 2024.
26. Sauve L, Forrester AM, Top KA. Group A streptococcal
pharyngitis: a practical guide to diagnosis and treatment.
Paediatr Child Health 2021;26:319-20. Crossref
27. Shulman ST, Bisno AL, Clegg HW, et al. Clinical practice
guideline for the diagnosis and management of group A
streptococcal pharyngitis: 2012 update by the Infectious
Diseases Society of America. Clin Infect Dis 2012;55:e86-102. Crossref
28. Bisno AL. Acute pharyngitis: etiology and diagnosis.
Pediatrics 1996;97:949-54. Crossref
29. Ebell MH, Smith MA, Barry HC, Ives K, Carey M. The
rational clinical examination. Does this patient have strep
throat? JAMA 2000;284:2912-8. Crossref
30. Bateman E, Mansour S, Okafor E, Arrington K, Hong
BY, Cervantes J. Examining the efficacy of antimicrobial
therapy in preventing the development of postinfectious
glomerulonephritis: a systematic review and meta-analysis.
Infect Dis Rep 2022;14:176-83. Crossref
31. Centre for Health Protection, Department of Health, Hong Kong SAR Government. Streptococcus pyogenes (pharyngeal specimens). Available from: https://www.chp.gov.hk/en/statistics/data/10/100044/6861.html. Accessed 18 Oct 2024.
32. National Institute for Health and Care Excellence. Sore
throat (acute): antimicrobial prescribing. 2018 January
26. Available from: https://www.nice.org.uk/guidance/ng84. Accessed 25 Mar 2025.
33. Rijk MH, Hullegie S, Schilder AG, et al. Incidence and
management of acute otitis media in adults: a primary
care-based cohort study. Fam Pract 2021;38:448-53. Crossref
34. Chonmaitree T, Alvarez-Fernandez P, Jennings K, et al.
Symptomatic and asymptomatic respiratory viral
infections in the first year of life: association with acute
otitis media development. Clin Infect Dis 2015;60:1-9. Crossref
35. Chonmaitree T, Revai K, Grady JJ, et al. Viral upper
respiratory tract infection and otitis media complication
in young children. Clin Infect Dis 2008;46:815-23. Crossref
36. Mohanty S, Podmore B, Cuñado Moral A, et al. Incidence
of acute otitis media from 2003 to 2019 in children ≤17
years in England. BMC Public Health 2023;23:201. Crossref
37. Hu T, Done N, Petigara T, et al. Incidence of acute otitis
media in children in the United States before and after the
introduction of 7- and 13-valent pneumococcal conjugate
vaccines during 1998-2018. BMC Infect Dis 2022;22:294. Crossref
38. Chonmaitree T. Acute otitis media is not a pure bacterial
disease. Clin Infect Dis 2006;43:1423-5. Crossref
39. Mather MW, Drinnan M, Perry JD, Powell S, Wilson JA,
Powell J. A systematic review and meta-analysis of
antimicrobial resistance in paediatric acute otitis media.
Int J Pediatr Otorhinolaryngol 2019;123:102-9. Crossref
40. Le Saux N, Robinson JL; Canadian Paediatric Society,
Infectious Diseases and Immunization Committee.
Management of acute otitis media in children six months
of age and older [in English, French]. Paediatr Child
Health 2016;21:39-50. Crossref
41. National Institute for Health and Care Excellence. Otitis
media (acute): antimicrobial prescribing. 2022 March 11.
Available from: https://www.nice.org.uk/guidance/ng91. Accessed 25 Mar 2025.
42. Venekamp RP, Sanders, SL, Glasziou, PP, Rovers MM.
Antibiotics for acute otitis media in children. Cochrane
Database Syst Rev 2023;11:CD000219. Crossref
43. Spurling GK, Dooley L, Clark J, Askew DA.
Immediate versus delayed versus no antibiotics for
respiratory infections. Cochrane Database Syst Rev
2023;10:CD004417. Crossref
44. Lui G, To HK, Lee N, et al. Adherence to treatment
guideline improves patient outcomes in a prospective
cohort of adults hospitalized for community-acquired
pneumonia. Open Forum Infect Dis 2020;7:ofaa146. Crossref
45. Jain S, Williams DJ, Arnold SR, et al. Community-acquired
pneumonia requiring hospitalization among
U.S. children. N Engl J Med 2015;372:835-45. Crossref
46. Bénet T, Sánchez Picot V, Messaoudi M, et al.
Microorganisms associated with pneumonia in children
<5 years of age in developing and emerging countries:
the GABRIEL pneumonia multicenter, prospective, case-control
study. Clin Infect Dis 2017;65:604-12. Crossref
47. Jain S, Self WH, Wunderink RG, et al. Community-acquired
pneumonia requiring hospitalization among
U.S. adults. N Engl J Med 2015;373:415-27. Crossref
48. National Institute for Health and Care Excellence. Pneumonia in adults: diagnosis and management. 2023 October 31. Available from: https://www.nice.org.uk/guidance/cg191. Accessed 25 Mar 2025.
49. Lim WS, Baudouin SV, George RC, et al. BTS guidelines
for the management of community acquired pneumonia
in adults: update 2009. Thorax 2009;64 Suppl 3:iii1-55. Crossref
50. Levy ML, Le Jeune I, Woodhead MA, Macfarlaned JT,
Lim WS; British Thoracic Society Community Acquired
Pneumonia in Adults Guideline Group. Primary care
summary of the British Thoracic Society Guidelines for
the management of community acquired pneumonia in
adults: 2009 update. Endorsed by the Royal College of
General Practitioners and the Primary Care Respiratory
Society UK. Prim Care Respir J 2010;19:21-7. Crossref
51. Harris M, Clark J, Coote N, et al. British Thoracic Society
guidelines for the management of community acquired
pneumonia in children: update 2011. Thorax 2011;66
Suppl 2:ii1-23. Crossref
52. Mandell LA, Wunderink RG, Anzueto A, et al. Infectious
Diseases Society of America/American Thoracic Society
consensus guidelines on the management of community-acquired
pneumonia in adults. Clin Infect Dis 2007;44
Suppl 2(Suppl 2):S27-72. Crossref
53. Bradley JS, Byington CL, Shah SS, et al. The management
of community-acquired pneumonia in infants and
children older than 3 months of age: clinical practice
guidelines by the Pediatric Infectious Diseases Society
and the Infectious Diseases Society of America. Clin
Infect Dis 2011;53:e25-76. Crossref
54. National Institute for Health and Care Excellence.
Pneumonia (community-acquired): antimicrobial
prescribing. 2019 September 16. Available from: https://www.nice.org.uk/guidance/ng138. Accessed 25 Mar 2025.
55. Centre for Health Protection, Department of Health, Hong
Kong SAR Government. Ho PL, Wu TC, editors. Reducing
Bacterial Resistance with IMPACT: interhospital multidisciplinary
programme on antimicrobial chemotherapy.
6th Edition. 2025. Available from: http://www.chp.gov.hk/files/pdf/reducing_bacterial_resistance_with_impact.pdf. Accessed 30 Aug 2025.
56. Centre for Health Protection, Department of Health,
Hong Kong SAR Government. Bacterial pathogen
isolation and percentage of antimicrobial resistance—out-patient setting. Available from: https://www.chp.gov.hk/en/statistics/data/10/641/697/3345.html. Accessed 13 Mar 2025.
57. Lung DC, Lam DS, Chan E, et al. Practice recommendation
for management of community acquired pneumonia in
children. Hong Kong J Paediatr (new series) 2016;21:178-93.
58. Centre for Health Protection, Department of Health,
Hong Kong SAR Government. Detection of Mycoplasma
pneumoniae in respiratory specimens in 2024.
Available from: https://www.chp.gov.hk/en/statistics/data/10/641/642/7062.html. Accessed 13 Mar 2025.
59. Kimberlin DW, Banerjee R, Barnett ED, Lynfield R,
Sawyer MH, editors. Red Book: 2024-2027 Report of
the Committee on Infectious Diseases. Committee on
Infectious Diseases, American Academy of Pediatrics;
2024. Crossref
60. United States Food and Drug Administration. FDA
warns about increased risk of ruptures or tears in the aorta blood vessel with fluoroquinolone antibiotics in
certain patients. Available from: https://web.archive.org/web/20251214152417/https://www.fda.gov/drugs/drug-safety-and-availability/fda-warns-about-increased-risk-ruptures-or-tears-aorta-blood-vessel-fluoroquinolone-antibiotics. Accessed 25 Nov 2024.
61. United States Food and Drug Administration. FDA
reinforces safety information about serious low blood sugar
levels and mental health side effects with fluoroquinolone
antibiotics; requires label changes. Available from:
https://web.archive.org/web/20251214152856/https:/www.fda.gov/drugs/drug-safety-and-availability/fda-reinforces-safety-information-about-serious-low-blood-sugar-levels-and-mental-health-side. Accessed 11 Feb 2025.
62. Drug Office, Department of Health, Hong Kong
SAR Government. Letters to Healthcare Providers.
Fluoroquinolone antibiotics: must now only be prescribed
when other commonly recommended antibiotics are
inappropriate in the United Kingdom (Letter to Healthcare
Professionals). 2024 January 23. Available from: https://www.drugoffice.gov.hk/eps/news/showNews/Fluoroquinolone+antibiotics%3A+must+now+only+be+prescribed+when+other+commonly+recommended+antibiotics+are+inappropriate+in+the+United+Kingdom+%2-8Letter+t/healthcare_providers/2024-01-23/en/52611.html . Accessed 11 Feb 2025.
63. Kuijpers SM, Buis DT, Ziesemer KA, et al. The evidence
base for the optimal antibiotic treatment duration of
upper and lower respiratory tract infections: an umbrella
review. Lancet Infect Dis 2025;25:94-113. Crossref
64. Kuitunen I, Jääskeläinen J, Korppi M, Renko M. Antibiotic
treatment duration for community-acquired pneumonia
in outpatient children in high-income countries—a
systematic review and meta-analysis. Clin Infect Dis
2023;76:e1123-8. Crossref
65. Gao Y, Liu M, Yang K, et al. Shorter versus longer-term
antibiotic treatments for community-acquired
pneumonia in children: a meta-analysis. Pediatrics
2023;151:e2022060097. Crossref
66. Mo Y, Tan WC, Cooper BS. Antibiotic duration for common bacterial infections—a systematic review. JAC Antimicrob Resist 2025;7:dlae215. Crossref
67. Metlay JP, Waterer GW, Long AC, et al. Diagnosis and
treatment of adults with community-acquired pneumonia.
An official clinical practice guideline of the American
Thoracic Society and Infectious Diseases Society of
America. Am J Respir Crit Care Med 2019;200:e45-67. Crossref
68. Rabe KF, Watz H. Chronic obstructive pulmonary disease.
Lancet 2017;389:1931-40. Crossref
69. Mannino DM, Buist AS. Global burden of COPD:
risk factors, prevalence, and future trends. Lancet
2007;370:765-73. Crossref
70. Non-Communicable Disease Branch, Centre for Health
Protection, Department of Health, Hong Kong SAR
Government. Population Health Survey 2020-22. Available
from: https://www.chp.gov.hk/en/resources/29/100057.html. Accessed 19 Mar 2026.
71. Chen W, Thomas J, Sadatsafavi M, FitzGerald JM. Risk
of cardiovascular comorbidity in patients with chronic
obstructive pulmonary disease: a systematic review and
meta-analysis. Lancet Respir Med 2015;3:631-9. Crossref
72. de Torres JP, Marín JM, Casanova C, et al. Lung cancer in patients with chronic obstructive pulmonary disease—incidence and predicting factors. Am J Respir Crit Care
Med 2011;184:913-9. Crossref
73. Barnes PJ, Celli BR. Systemic manifestations and
comorbidities of COPD. Eur Respir J 2009;33:1165-85. Crossref
74. Global Initiative for Chronic Obstructive Lung Disease.
2024 Gold Report. Global Strategy for Prevention,
Diagnosis and Management of COPD: 2024 Report.
Available from: https://goldcopd.org/2024-gold-report/. Accessed 25 Mar 2025.
75. Papi A, Bellettato CM, Braccioni F, et al. Infections and
airway inflammation in chronic obstructive pulmonary
disease severe exacerbations. Am J Respir Crit Care Med
2006;173:1114-21. Crossref
76. Ko FW, Ip M, Chan PK, Ng SS, Chau SS, Hui DS. A one-year
prospective study of infectious etiology in patients
hospitalized with acute exacerbations of COPD and
concomitant pneumonia. Respir Med 2008;102:1109-16. Crossref
77. Ko FW, Ip M, Chan PK, et al. Viral etiology of
acute exacerbations of COPD in Hong Kong. Chest
2007;132:900-8. Crossref
78. Ko FW, Lam RK, Li TS, et al. Sputum bacteriology
in patients hospitalized with acute exacerbations of
chronic obstructive pulmonary disease and concomitant
pneumonia in Hong Kong. Intern Med J 2005;35:661-7. Crossref
79. Ko FW, Ng TK, Li TS, et al. Sputum bacteriology in
patients with acute exacerbations of COPD in Hong
Kong. Respir Med 2005;99:454-60. Crossref
80. Hui DS, Ip M, Ling T, et al. A multicentre surveillance
study on the characteristics, bacterial aetiologies and
in vitro antibiotic susceptibilities in patients with
acute exacerbations of chronic bronchitis. Respirology
2011;16:532-9. Crossref
81. Miravitlles M, Kruesmann F, Haverstock D, Perroncel R,
Choudhri SH, Arvis P. Sputum colour and bacteria in
chronic bronchitis exacerbations: a pooled analysis. Eur
Respir J 2012;39:1354-60. Crossref
82. Soler N, Agustí C, Angrill J, Puig De la Bellacasa J, Torres A.
Bronchoscopic validation of the significance of sputum
purulence in severe exacerbations of chronic obstructive
pulmonary disease. Thorax 2007;62:29-35. Crossref
83. Stockley RA, O’Brien C, Pye A, Hill SL. Relationship of
sputum color to nature and outpatient management of
acute exacerbations of COPD. Chest 2000;117:1638-45. Crossref
84. Garcia-Vidal C, Almagro P, Romaní V, et al. Pseudomonas
aeruginosa in patients hospitalised for COPD exacerbation:
a prospective study. Eur Respir J 2009;34:1072-8. Crossref
85. Parameswaran GI, Sethi S. Pseudomonas infection in
chronic obstructive pulmonary disease. Future Microbiol
2012;7:1129-32. Crossref
86. Gallego M, Pomares X, Espasa M, et al. Pseudomonas
aeruginosa isolates in severe chronic obstructive
pulmonary disease: characterization and risk factors.
BMC Pulm Med 2014;14:103. Crossref
87. Boixeda R, Almagro P, Díez-Manglano J, et al. Bacterial
flora in the sputum and comorbidity in patients with acute
exacerbations of COPD. Int J Chron Obstruct Pulmon Dis
2015;10:2581-91. Crossref
88. Llor C, Moragas A, Miravitlles M, Mesquita P, Cordoba G.
Are short courses of antibiotic therapy as effective as
standard courses for COPD exacerbations? A systematic
review and meta-analysis. Pulm Pharmacol Ther
2022;72:102111. Crossref
89. Stolbrink M, Amiry J, Blakey JD. Does antibiotic
treatment duration affect the outcomes of exacerbations
of asthma and COPD? A systematic review. Chron Respir
Dis 2018;15:225-40. Crossref
90. National Institute for Health and Care Excellence. Chronic
obstructive pulmonary disease (acute exacerbation):
antimicrobial prescribing. 2018 December 5. Available
from: https://www.nice.org.uk/guidance/ng114. Accessed 25 Mar 2025.
91. SA Health, Government of South Australia. Urinary
Tract Infections (adult): Empirical Treatment Clinical
Guideline. September 2023. Available from: https://www.sahealth.sa.gov.au/wps/wcm/connect/public+content/sa+health+internet/about+us/governance/policy+governance/policies/urinary+tract+infections+adult+empirical+treatment+clinical+guideline . Accessed 26 May 2025.
92. National Institute for health and Care Excellence. Urinary
tract infections in adults. 2023 February 15. Available
from: https://www.nice.org.uk/guidance/qs90. Accessed 25 Mar 2025.
93. Kranz J, Bartoletti R, Bruyère F, et al. European Association
of Urology Guidelines on Urological Infections: summary
of the 2024 Guidelines. Eur Urol 2024;86:27-41. Crossref
94. United States Food and Drug Administration.
Uncomplicated urinary tract infections: developing drugs
for treatment guidance for industry. August 2019. Available
from: https://www.fda.gov/regulatory-information/search-fda-guidance-documents/uncomplicated-urinary-tract-infections-developing-drugs-treatment-guidance-industry . Accessed 12 Aug 2025.
95. British Columbia. Urinary tract infections in the primary
care setting–investigation. 2020 July 29. Available from:
https://www2.gov.bc.ca/gov/content/health/practitioner-professional-resources/bc-guidelines/urinary-tract-infections. Accessed 21 Jul 2025.
96. Trautner BW, Cortés-Penfield NW, Gupta K, et al. IDSA
2025 Guideline update on complicated urinary tract
infections. 2025 July 17. Available from: https://www.idsociety.org/practice-guideline/complicated-urinary-tract-infections/. Accessed 12 Aug 2025.
97. Gupta K, Hooton TM, Naber KG, et al. International
clinical practice guidelines for the treatment of acute
uncomplicated cystitis and pyelonephritis in women:
a 2010 update by the Infectious Diseases Society of
America and the European Society for Microbiology and
Infectious Diseases. Clin Infect Dis 2011;52:e103-20. Crossref
98. National Institute for Health and Care Excellence.
Urinary tract infection (lower): antimicrobial prescribing.
2018 October 31. Available from: https://www.nice.org.uk/guidance/ng109. Accessed 25 Mar 2025.
99. Nicolle LE, Gupta K, Bradley SF, et al. Clinical Practice
Guideline for the Management of Asymptomatic
Bacteriuria: 2019 update by the Infectious Diseases
Society of America. Clin Infect Dis 2019;68:e83-110. Crossref
100. UK Health Security Agency. Diagnosis of urinary tract
infections: quick reference tools for primary care.
Updated 2025 July 7. Available from: https://www.gov.uk/government/publications/urinary-tract-infection-diagnosis/diagnosis-of-urinary-tract-infections-quick-reference-tools-for-primary-care. Accessed 12 Aug 2025.
101. Sartelli M, Guirao X, Hardcastle TC, et al. 2018 WSES/SIS-E consensus conference: recommendations for the management of skin and soft-tissue infections. World J
Emerg Surg 2018;13:58. Crossref
102. Ramakrishnan K, Salinas RC, Agudelo Higuita NI. Skin and soft tissue infections. Am Fam Physician 2015;92:474-83.
103. National Institute for Health and Care Excellence.
Cellulitis and erysipelas: antimicrobial prescribing. 2019
September 27. Available from: https://www.nice.org.uk/guidance/ng141. Accessed 25 Mar 2025.
104. Stevens DL, Bisno AL, Chambers HF, et al. Practice
guidelines for the diagnosis and management of skin
and soft tissue infections: 2014 update by the Infectious
Diseases Society of America. Clin Infect Dis 2014;59:e10-52. Crossref
105. National Institute for Health and Care Excellence.
Impetigo: antimicrobial prescribing. 2020 February 26.
Available from: https://www.nice.org.uk/guidance/ng153. Accessed 25 Mar 2025.
106. Leung YH, Lai RW, Chan AC, et al. Risk factors for community-associated methicillin-resistant Staphylococcus aureus infection in Hong Kong. J Infect 2012;64:494-9. Crossref
107. Centre for Health Protection, Department of Health,
Hong Kong SAR Government. Community-associated
methicillin-resistant Staphylococcus aureus (CA-MRSA)
infection. 2026 February 9. Available from: https://www.chp.gov.hk/en/healthtopics/content/24/5392.html. Accessed 19 Mar 2026.
108. Sartelli M, Coccolini F, Kluger Y, et al. WSES/GAIS/WSIS/SIS-E/AAST global clinical pathways for patients with skin and soft tissue infections. World J Emerg Surg
2022;17:3. Crossref
109. Stevens DL, Bisno AL, Chambers HF, et al. Practice
guidelines for the diagnosis and management of skin
and soft tissue infections: 2014 update by the Infectious
Diseases Society of America. Clin Infect Dis 2014;59:147-59. Crossref
110. The Royal Children’s Hospital Melbourne. Cellulitis and
other bacterial skin infections. March 2020. Available
from: https://www.rch.org.au/clinicalguide/guideline_index/cellulitis_and_skin_infections/. Accessed 18 Aug 2025.
111. Burkett E, Cranitch E, Bandiera D, Ward T. Improving the
quality and choice of care setting for residents of aged care
facilities with acute healthcare needs steering committee.
Management of acute care needs of RACF residents: a suite
of collaborative pathways for general practitioners and
registered nurses. Available from: https://www.health.qld.gov.au/clinical-practice/guidelines-procedures/clinical-pathways/residential-aged-care-clinical-pathways/all-pathways/cellulitis. Accessed 18 Aug 2025.
112. World Health Organization. World leaders commit
to decisive action on antimicrobial resistance. 2024
September 26. Available from: https://www.who.int/news/item/26-09-2024-world-leaders-commit-to-decisive-action-on-antimicrobial-resistance. Accessed 19 Jan 2026.
113. Ma E, Hsu E, Chow T, Ko LS, Lau KY, Ho B. Change in
public knowledge, attitude and practice on antibiotic
use after a territory-wide health promotion campaign in
Hong Kong. J Infect Dis Ther 2024;12:604.