Hong Kong Med J 2026;32:Epub 13 Apr 2026
© Hong Kong Academy of Medicine. CC BY-NC-ND 4.0
MEDICAL PRACTICE
Consensus statement on the use of Alzheimer’s
disease biomarkers and anti-amyloid therapies in Hong Kong
CK Shum, FHKAM (Medicine), FRCP1 #; YF Shea, FRCP, FHKAM (Medicine)2 #; TW Au Yeung, MD, FHKAM (Medicine)3; Cherry CY Chan, FHKAM (Radiology), FHKCR4; WC Chan, MB, ChB, FHKAM (Psychiatry)5; WK Cheng, MB, ChB, FHKAM (Medicine)6; Nelson YF Cheung, FRCP, FHKAM (Medicine)7; Danny HY Cho, MB, BS, FHKAM (Radiology)4; TK Chow, FHKCR, FHKAM (Radiology)8; Gardian CY Fong, MD, FRCP9; Bonaventure YM Ip, FHKAM (Medicine), FRCP10; Julio SH Kwok, FHKCR, FHKAM (Radiology)11; Billy MH Lai, MB, BS, FHKAM (Radiology)12; Linda CW Lam, MB, ChB, FHKAM (Psychiatry)13; Allen TC Lee, MD, FHKAM (Psychiatry)13; CM Lok, MB, ChB, FHKCR14; KY Mok, MB, BS, FHKAM (Medicine)15,16; David KK Ng, MB, BS, FHKAM (Radiology)17; Deyond YW Siu, FRCR, FHKCR18; PY Yeung, MB, ChB, FHKAM (Medicine)19; Stanley KF Tam, FHKAM (Medicine), FRCP20
1 Department of Medicine and Geriatrics, Tuen Mun Hospital, Hong Kong SAR, China
2 Department of Medicine, Queen Mary Hospital, Hong Kong SAR, China
3 The Jockey Club Institute of Ageing, The Chinese University of Hong Kong, Hong Kong SAR, China
4 Dtepartment of Diagnostic and Interventional Radiology, Kwong Wah Hospital, Hong Kong SAR, China
5 Department of Psychiatry, Li Ka Shing Faculty of Medicine, The University of Hong Kong, Hong Kong SAR, China
6 Department of Medicine, Kwong Wah Hospital, Hong Kong SAR, China
7 Department of Medicine, Queen Elizabeth Hospital, Hong Kong SAR, China
8 Department of Radiology and Nuclear Medicine, Nuclear Medicine Unit, Tuen Mun Hospital, Hong Kong SAR, China
9 813 Medical Centre, Hong Kong SAR, China
10 Department of Medicine and Therapeutics, The Chinese University of Hong Kong, Hong Kong SAR, China
11 Nuclear Medicine and PET Centre, Hong Kong Baptist Hospital, Hong Kong SAR, China
12 Department of Imaging and Interventional Radiology, Prince of Wales Hospital, Hong Kong SAR, China
13 Department of Psychiatry, The Chinese University of Hong Kong, Hong Kong SAR, China
14 Assure Medical Imaging, Hong Kong SAR, China
15 Division of Life Science, The Hong Kong University of Science and Technology, Hong Kong SAR, China
16 Hong Kong Center for Neurodegenerative Diseases, Hong Kong SAR, China
17 Nuclear Medicine Unit, Queen Elizabeth Hospital, Hong Kong SAR, China
18 Scanning Department, St Teresa’s Hospital, Hong Kong SAR, China
19 Department of Medicine, Tung Wah Eastern Hospital, Hong Kong SAR, China
20 Department of Medicine, Hong Kong Buddhist Hospital, Hong Kong SAR, China
Corresponding author: Dr CK Shum (sck375@ha.org.hk)
Abstract
Alzheimer’s disease (AD) is the most common
aetiology of cognitive impairment worldwide and
in Hong Kong. There have been rapid advances in
the use of biomarkers for the diagnosis of AD and
in the availability of anti-amyloid therapies (AAT)
to slow cognitive and functional decline. At present,
there is no consensus in Hong Kong regarding the
application of AD biomarkers or the use of AAT. A
multidisciplinary group of 20 medical specialists from
five professional societies discussed issues related to
the application of biomarkers for the diagnosis of
AD pathology and the use of AAT, and reviewed the
evidence in the context of local experience to inform
recommendations. A modified Delphi approach
was adopted to finalise the recommendations.
Consensus was defined as ≥75% agreement on a
9-point Likert scale among panellists. The panel
finalised 26 consensus statements addressing the
use of AD biomarkers, including neuroimaging and
fluid biomarkers, as well as the use of AAT, including
inclusion criteria, serial neuroimaging monitoring
during treatment, and management of infusion
reactions. These recommendations are relevant to
the Hong Kong healthcare setting and may serve as
guidance for doctors across specialties to facilitate
appropriate management of AD.
Introduction
There is increasing demand for accurate diagnosis
of Alzheimer’s disease (AD) pathology with the
availability of anti-amyloid therapies (AAT).1 2
However, diagnostic accuracies for AD pathology
in primary care and specialised memory clinics
are approximately 61% and 73%, respectively.3
In Hong Kong, patients with memory problem
are often referred to specialised clinics. Plasma
biomarkers and neuroimaging have revolutionised
the diagnostic approach to AD. Recently, AAT have
emerged as disease-modifying treatments targeting
the underlying pathology of AD.1 2 They have been
shown to slow cognitive and functional decline and
reduce amyloid-beta plaque burden.1 2 Nevertheless,
amyloid-related imaging abnormalities (ARIA),
frequent intravenous dosing, magnetic resonance
imaging (MRI) monitoring, treatment costs, and
healthcare infrastructure to support the new AAT
treatment in Hong Kong remain important concerns
in clinical practice.1 2
In Hong Kong, there is currently no consensus
regarding the use of AD biomarkers or AAT. This
article presents the findings of an expert panel
convened to formulate the first multispecialty
consensus recommendations on the use of AD
biomarkers and AAT, with the aim of providing
practical local guidance for healthcare practitioners
based on current evidence and expert opinion. This
consensus statement comprises two parts, namely,
the use of AD biomarkers and the use of AAT.
Methods
The joint consensus panel comprised 20 medical
specialists from Hong Kong: four geriatricians
(CK Shum, YF Shea, TW Au Yeung, PY Yeung)
representing the Hong Kong Geriatrics Society;
three psychiatrists (WC Chan, Linda CW Lam, Allen
TC Lee) and one neurologist (KY Mok) representing
the Chinese Dementia Research Association; four
nuclear medicine physicians (TK Chow, Julio SH
Kwok, CM Lok, David KK Ng) representing the Hong
Kong Society of Nuclear Medicine and Molecular
Imaging; four neurologists (WK Cheng, Nelson YF
Cheung, Gardian CY Fong, Bonaventure YM Ip)
representing the Hong Kong Neurological Society;
and four radiologists (Cherry CY Chan, Danny HY
Cho, Billy MH Lai, Deyond YW Siu) representing
the Hong Kong Society of Diagnostic Radiologists.
The panellists were nominated by the respective
professional societies and had relevant knowledge
and experience in the field.
Literature searches were conducted in PubMed
and Ovid to identify relevant articles. The keywords
used were ‘biomarkers’, ‘Alzheimer’s disease’,
‘plasma’, ‘amyloid positron emission tomography’,
‘memory clinic’, ‘lecanemab’, and ‘donanemab’. In
total, 49 articles were selected, including 10 major
guidelines,4 5 6 7 8 9 10 11 12 13 four meta-analyses or systematic
reviews,14 15 16 17 two randomised controlled trials,1 2 32 original articles,3 18 19 20 21 22 23 24 25 26 27 28 29 30 31 32 33 34 35 36 37 38 39 40 41 42 43 44 45 46 47 48 and one case report.49
Consensus statements were developed using
a modified Delphi process (Fig 1).50 Panellists
evaluated each drafted statement on a 9-point Likert
scale (1=strongly agree; 2=agree; 3=moderately
agree; 4=mildly agree; 5=neutral; 6=mildly disagree;
7=moderately disagree; 8=disagree; 9=strongly
disagree). A statement was considered ‘accepted’ if
at least 75% of panellists rated it 1-3, and ‘rejected’
if at least 75% rated it 7-9. Statements with less than
75% agreement were rephrased and subjected to
further voting. The level of evidence was determined
according to the Oxford Centre for Evidence-Based
Medicine 2011 Levels of Evidence.51
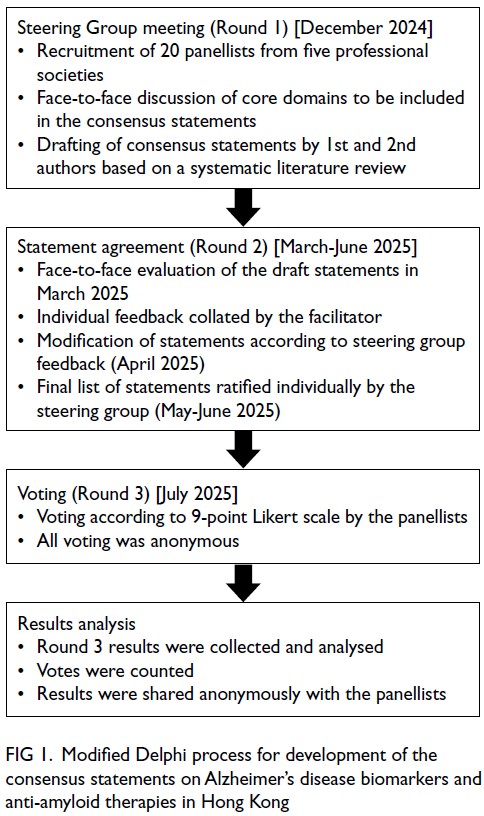
Figure 1. Modified Delphi process for development of the consensus statements on Alzheimer’s disease biomarkers and anti-amyloid therapies in Hong Kong
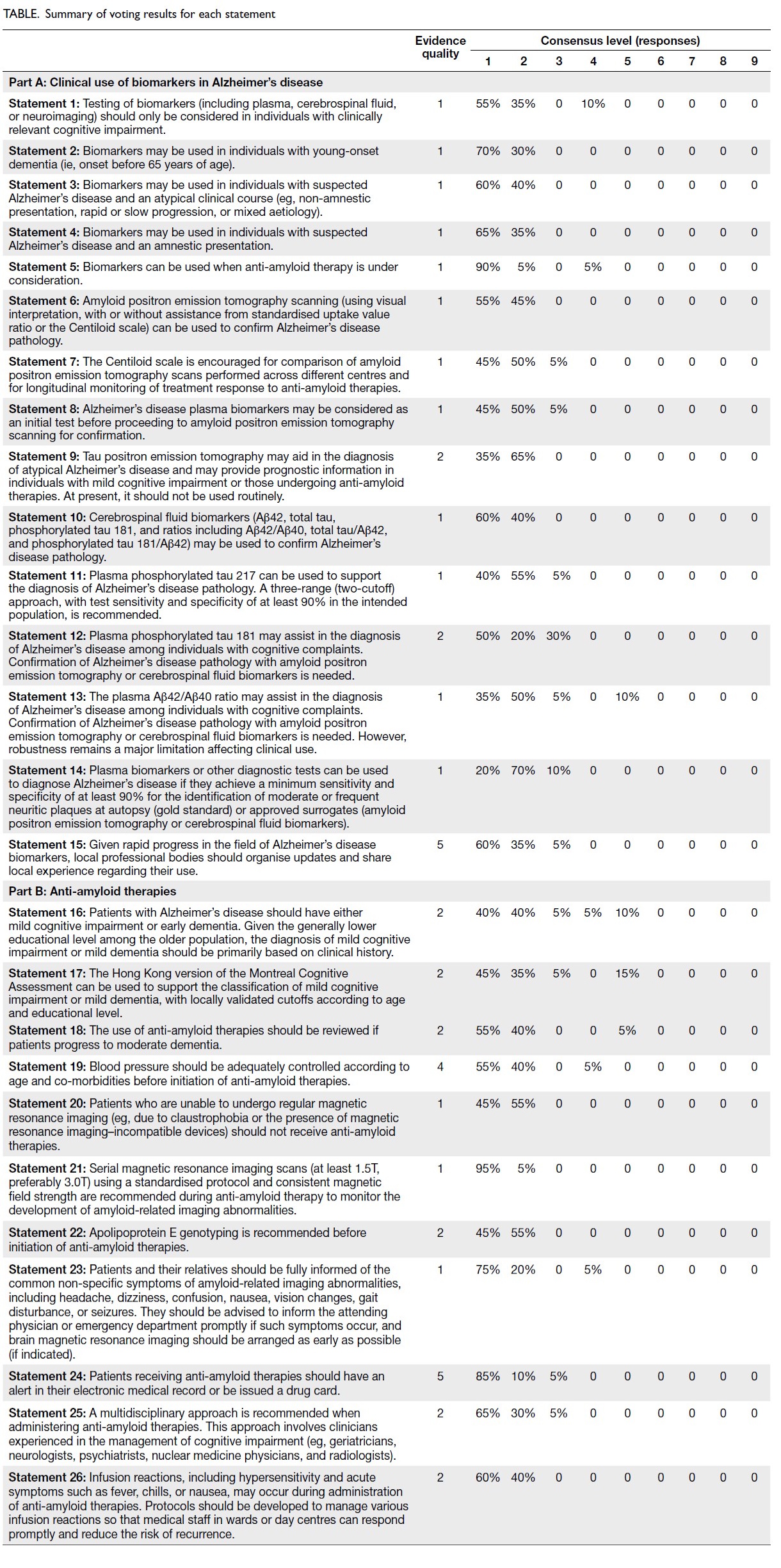
Results
In total, 26 statements met the threshold for
consensus and were accepted after the first round
of voting; these are summarised in the Table. No
statements required rephrasing or further voting.
Part A: Clinical use of biomarkers in
Alzheimer’s disease
Target population
Statement 1: Testing of biomarkers (including
plasma, cerebrospinal fluid, or neuroimaging)
should only be considered in individuals with
clinically relevant cognitive impairment.
Two recent diagnostic criteria for AD7 9
emphasise avoiding AD biomarker testing in
individuals without cognitive complaints (ie,
asymptomatic individuals in the community
who may harbour AD pathology). It remains
uncertain whether such individuals will progress
to mild cognitive impairment (MCI) or dementia.7
Furthermore, there is no evidence that treatment
in these individuals prevents future cognitive
decline. Ongoing clinical trials aim to address these
questions.
Clinical indications
Statement 2: Biomarkers may be used in individuals with young-onset dementia (ie, onset before 65 years
of age).
Statement 3: Biomarkers may be used in individuals
with suspected Alzheimer’s disease and an atypical
clinical course (eg, non-amnestic presentation, rapid
or slow progression, or mixed aetiology).
Individuals with young-onset dementia may
have various potential underlying aetiologies.
Moreover, young-onset AD (ie, before 65 years of age) is more likely to present with atypical clinical
features, including prominent agnosia (posterior
cortical atrophy), predominant language impairment
(logopenic variant primary progressive aphasia),
marked behavioural manifestations (eg, disinhibition
in behavioural variant AD), or atypical parkinsonism,
as seen in corticobasal syndrome.10 16 Confirmation
of underlying AD pathology is important when
considering AD-specific treatment.
Statement 4: Biomarkers may be used in individuals with suspected Alzheimer’s disease and an amnestic presentation.
The diagnostic accuracy of a clinical diagnosis
of AD is approximately 77%, even among dementia
specialists.3 Observational studies and drug trials
have shown that 15% to 20% of individuals clinically
diagnosed with late-onset AD dementia had negative
amyloid positron emission tomography (PET)
results.10 The prevalence of amyloid PET positivity
decreases with advancing age among patients with
typical amnestic dementia, reflecting an increasing
prevalence of non-AD pathologies (eg, limbic-predominant
age-related TDP-43 encephalopathy)
that can clinically mimic AD.5
Statement 5: Biomarkers can be used when anti-amyloid therapy is under consideration.
Initiation of AAT requires confirmation
of AD pathology in accordance with current
recommendations.6 11 At present, either amyloid PET
imaging or cerebrospinal fluid (CSF) biomarkers is
recommended.6 11
Amyloid positron emission tomography imaging
Statement 6: Amyloid positron emission tomography
scanning (using visual interpretation, with or
without assistance from standardised uptake value
ratio or the Centiloid scale) can be used to confirm
Alzheimer’s disease pathology.
Statement 7: The Centiloid scale is encouraged
for comparison of amyloid positron emission
tomography scans performed across different centres
and for longitudinal monitoring of treatment
response to anti-amyloid therapies.
Statement 8: Alzheimer’s disease plasma biomarkers
may be considered as an initial test before
proceeding to amyloid positron emission tomography
scanning for confirmation.
Amyloid PET imaging provides direct
visualisation of amyloid plaques in the brain, enabling
confirmation of amyloid pathology.10 Currently, only
11C-Pittsburgh compound B, 18F-flutemetamol, and
18-florbetaben are available locally. In the IDEAS
study (Imaging Dementia—Evidence for Amyloid
Scanning), which involved 11 409 individuals, patient management changed in 60.2% of those with MCI
and 63.5% of those with dementia.30 The diagnosis
changed from AD to non-AD in 25.1% of patients
and from non-AD to AD in 10.5%.30 Amyloid PET
has been included as a core biomarker of amyloid
deposition.9 It should be performed before initiation
of AAT and may be used to confirm AD pathology
in individuals with inconclusive plasma or CSF
biomarker results.9 12 17 However, high cost and
limited availability may restrict its routine use.
Therefore, measurement of plasma AD biomarkers
as an initial test is recommended before proceeding
to amyloid PET imaging (eg, in the context of
financial constraints).12 17
Given the availability of multiple centres
offering amyloid PET imaging in Hong Kong and the
potential need for serial scans to monitor response
to AAT, reporting of amyloid burden using the
Centiloid scale is encouraged.21
Tau positron emission tomography imaging
Statement 9: Tau positron emission tomography
may aid in the diagnosis of atypical Alzheimer’s disease and may provide prognostic information in
individuals with mild cognitive impairment or those
undergoing anti-amyloid therapies. At present, it
should not be used routinely.
Only one tau PET tracer is currently available
in Hong Kong: 18F-T807 (also known as 18F-AV-1451
or 18F-flortaucipir). In donanemab trials, individuals
with a lower tau burden on tau PET demonstrated
greater slowing of cognitive decline compared
with those with higher tau deposition.2 A previous
study has also shown that tau deposition follows
characteristic patterns in atypical AD, such as
increased uptake in the posterior regions in posterior
cortical atrophy and in the language-dominant left
hemisphere in logopenic variant primary progressive
aphasia.32
Cerebrospinal fluid biomarkers
Statement 10: Cerebrospinal fluid biomarkers
(Aβ42, total tau, phosphorylated tau 181, and
ratios including Aβ42/Aβ40, total tau/Aβ42, and
phosphorylated tau 181/Aβ42) may be used to
confirm Alzheimer’s disease pathology.
It is well established that individuals with AD
have approximately 50% lower CSF Aβ42 levels
than non-AD controls, owing to amyloid plaque
deposition, and approximately twofold higher
total tau (T-tau) or phosphorylated tau (p-tau181)
levels, reflecting neuronal injury and release of
neurofibrillary tangles into the CSF.15 Ratios such
as Aβ42/Aβ40, T-tau/Aβ42, and p-tau181/Aβ42
improve diagnostic accuracy for AD pathology.15
A local study demonstrated that the sensitivity and
specificity of the CSF Aβ42/T-tau and Aβ42/p-tau181
ratios were 96% and 83%, and 92% and 83%,
respectively.31 Cerebrospinal fluid biomarkers may
be used to confirm AD pathology in individuals with
inconclusive plasma biomarker results.6 9 11
Plasma biomarkers
Statement 11: Plasma phosphorylated tau 217 can
be used to support the diagnosis of Alzheimer’s
disease pathology. A three-range (two-cutoff)
approach, with test sensitivity and specificity
of at least 90% in the intended population, is
recommended.
Alzheimer’s disease is characterised by
neurofibrillary tangles composed of p-tau species,
including p-tau181 and p-tau217.14 These are
released following neuronal injury and death.14
Plasma p-tau217 has emerged as a robust
biomarker for AD pathology and is elevated by
250% to 600% in patients with AD compared
with non-AD individuals.3 14 17 19 22 25 26 Multiple
measurement techniques are available, including
immunoprecipitation mass spectrometry (IP-MS),
the Meso Scale Discovery platform, and single
molecule array. Increasing evidence suggests that
the diagnostic accuracy of plasma p-tau217 is
comparable to that of CSF biomarkers or amyloid
PET imaging.25 26 33 A three-range (two-cutoff)
strategy has been proposed, comprising a lower
threshold to rule out AD (90%-95% sensitivity)
and a higher threshold to rule in AD (90%-95%
specificity).25 A recent guideline has included plasma
p-tau217 as a core biomarker for the diagnosis of AD
pathology.9
Statement 12: Plasma phosphorylated tau 181 may
assist in the diagnosis of Alzheimer’s disease among
individuals with cognitive complaints. Confirmation
of Alzheimer’s disease pathology with amyloid
positron emission tomography or cerebrospinal fluid
biomarkers is needed.
Similar to p-tau217, plasma p-tau181 is
elevated in individuals with AD.17 34 Levels can be
measured using IP-MS, single molecule array, or
other ultra-sensitive immunoassays.17 34 Previously
reported values of area under the receiver operating
characteristic curve (AUC) for the detection of AD pathology range from 0.86 to 0.89.17 34 Confirmation
of AD pathology with amyloid PET imaging or CSF
biomarker assessment remains necessary.
Statement 13: The plasma Aβ42/Aβ40 ratio may
assist in the diagnosis of Alzheimer’s disease among
individuals with cognitive complaints. Confirmation
of Alzheimer’s disease pathology with amyloid
positron emission tomography or cerebrospinal fluid
biomarkers is needed. However, robustness remains
a major limitation affecting clinical use.
Plasma Aβ42 levels and the Aβ42/Aβ40 ratio
are reduced in individuals with positive amyloid
PET findings.27 The Aβ42/Aβ40 ratio can be measured by IP-MS (AUC=0.86-0.89)17 or by ultra-sensitive
immunoassays (AUC=0.69-0.78).27 The
plasma Aβ42/Aβ40 ratio correlates with cerebral
amyloid PET and CSF amyloid measurements, and
its diagnostic accuracy is relatively high across
the AD spectrum.17 27 The ratio is more strongly
associated with amyloid deposition than individual
plasma Aβ42 or Aβ40 values. However, the plasma
Aβ42/Aβ40 ratio is reduced by only 8% to 15% in
individuals harbouring AD pathology,17 27 compared
with a 40% to 60% reduction in CSF, owing to
peripheral production of Aβ in extracerebral
tissues.17 Consequently, there is greater overlap in
plasma Aβ42/Aβ40 ratios between AD and non-AD
individuals than in corresponding CSF ratios. The
robustness of plasma Aβ measurements declines
substantially with minor increases in the coefficient
of variation, and plasma Aβ is highly sensitive to
small measurement biases.18 29 The Aβ42/Aβ40
ratio may also be affected by medications such as
sacubitril/valsartan.17
Statement 14: Plasma biomarkers or other
diagnostic tests can be used to diagnose Alzheimer’s
disease if they achieve a minimum sensitivity and
specificity of at least 90% for the identification of
moderate or frequent neuritic plaques at autopsy
(gold standard) or approved surrogates (amyloid
positron emission tomography or cerebrospinal fluid
biomarkers).
The Lumipulse G pTau217/β-amyloid 1-42
plasma ratio in vitro diagnostic test (Fujirebio,
Tokyo, Japan) has been approved by the United States
Food and Drug Administration for the detection
of Alzheimer’s pathology.52 It adopts a three-range
approach; confirmatory testing (amyloid PET
imaging or CSF biomarkers) is required if results
are indeterminate, which occurs in fewer than 20%
of patients.52 The reported positive predictive value
is 92% and the negative predictive value is 97%.52
Other international organisations have similarly
recommended performance thresholds of at least
90% sensitivity and 90% specificity for confirmatory
tests of AD pathology.12 13
Role of professional bodies
Statement 15: Given rapid progress in the field of
Alzheimer’s disease biomarkers, local professional
bodies should organise updates and share local
experience regarding their use.
Local professional bodies may organise
workshops or regular updates to guide clinicians.
Local assays are in development and have shown
encouraging results (eg, PlasmarkAD; Cognitact,
Hong Kong SAR, China); they may have future
clinical utility if they meet the minimum performance
requirements for the test assay in the intended
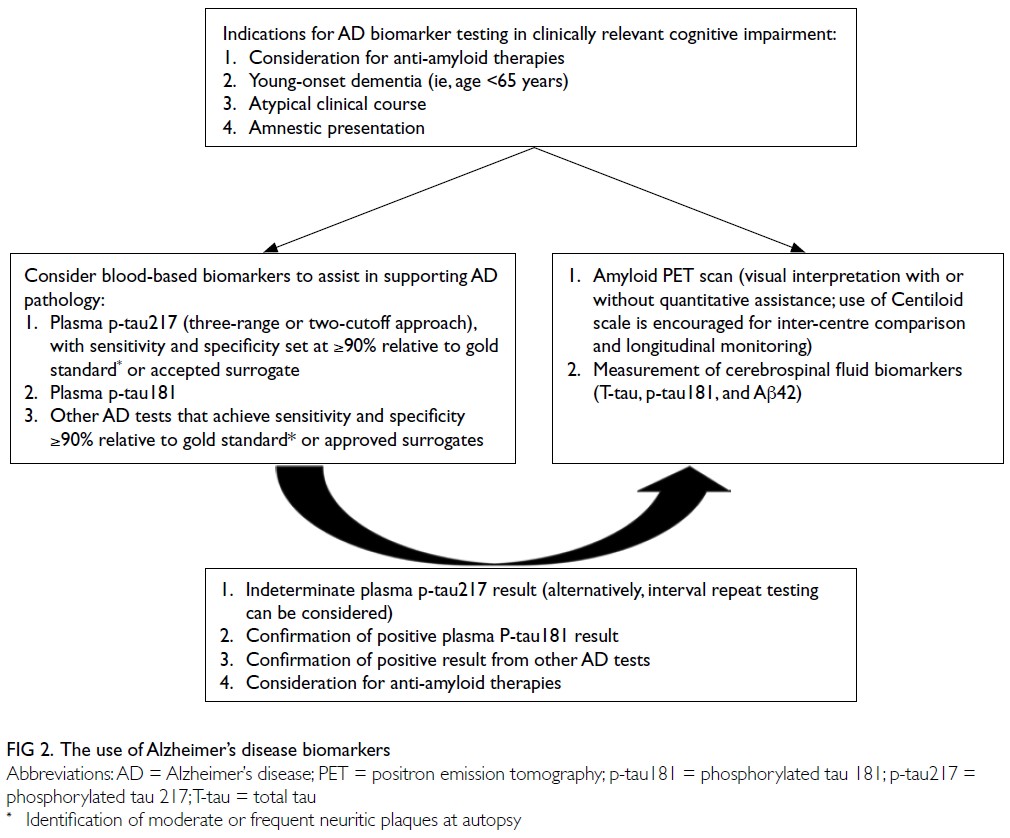
population.24 The use of AD biomarkers suggested in
the consensus statement was summarised in Figure 2.
Part B: Anti-amyloid therapies
Target population
Statement 16: Patients with Alzheimer’s disease
should have either mild cognitive impairment
or early dementia. Given the generally lower
educational level among the older population, the
diagnosis of mild cognitive impairment or mild dementia should be primarily based on clinical history.
Statement 17: The Hong Kong version of the
Montreal Cognitive Assessment can be used
to support the classification of mild cognitive
impairment or mild dementia, with locally
validated cutoffs according to age and educational
level.
Statement 18: The use of anti-amyloid therapies
should be reviewed if patients progress to moderate
dementia.
Older adults in Hong Kong often have lower
levels of formal education. Clinical history obtained
from a primary caregiver should be relied upon when
assessing cognitive impairment, including staging as
MCI or dementia. Mild cognitive impairment should
be diagnosed according to the Petersen criteria,28
with preservation of activities of daily living. In
patients with dementia, activities of daily living are
impaired. The Hong Kong version of the Montreal
Cognitive Assessment has been locally validated
and may be used as supportive evidence for the
diagnosis of MCI or mild dementia.11 35 Alternatively, the Cantonese version of the Mini–Mental State
Examination may be used in accordance with the
Clarity-AD study, which included patients with the
scores between 22 and 30.6 20
Blood pressure
Statement 19: Blood pressure should be adequately
controlled according to age and co-morbidities
before initiation of anti-amyloid therapies.
Poorly controlled blood pressure has been
associated with an increased risk of ARIA.8
Magnetic resonance imaging
Statement 20: Patients who are unable to undergo
regular magnetic resonance imaging (eg, due
to claustrophobia or the presence of magnetic
resonance imaging–incompatible devices) should not
receive anti-amyloid therapies.
Statement 21: Serial magnetic resonance imaging
scans (at least 1.5T, preferably 3.0T) using a
standardised protocol and consistent magnetic field
strength are recommended during anti-amyloid
therapy to monitor the development of amyloid-related
imaging abnormalities.
Magnetic resonance imaging is important for
monitoring ARIA during treatment with AAT.6 11
Accurate detection and follow-up are critical. This
is particularly relevant for sequences used to detect
microhaemorrhages, including susceptibility-weighted
imaging, T2*-weighted gradient echo (T2*
GRE), and susceptibility-weighted angiography
(SWAN); the first two are more commonly used
in Hong Kong. Susceptibility-weighted imaging
is reportedly more sensitive than T2* GRE for
detecting haemosiderin deposition. Nevertheless,
T2* GRE has been preferred in clinical trials because
of lower inter-scanner variability.4 A previous study
comparing SWAN with T2*-weighted imaging
indicated that SWAN was similar or superior,
particularly for detecting microbleeds or lesions
near the skull base.23 Serial MRI scans should be
performed using standardised imaging protocols
and, whenever possible, the same scanner to
minimise inter-scan variability.
Relevant to amyloid-related imaging abnormalities
Statement 22: Apolipoprotein E genotyping is
recommended before initiation of anti-amyloid
therapies.
Lecanemab was first approved in China in
January 2024, based on the Clarity-AD study,53 which
did not include participants of Chinese ethnicity.
Donanemab was approved in China in December
2024.54 Consequently, data regarding the use of AAT
in the Chinese population remain limited. Given
the early stage of local implementation, patients and their relatives should be fully informed of the
increased risk of ARIA associated with AAT. To
facilitate informed decision-making, apolipoprotein
E genotyping should be performed.6 11
Statement 23: Patients and their relatives should be
fully informed of the common non-specific symptoms
of amyloid-related imaging abnormalities, including
headache, dizziness, confusion, nausea, vision
changes, gait disturbance, or seizures. They should
be advised to inform the attending physician or
emergency department promptly if such symptoms
occur, and brain magnetic resonance imaging should
be arranged as early as possible (if indicated).
Statement 24: Patients receiving anti-amyloid
therapies should have an alert in their electronic
medical record or be issued a drug card.
Fatal outcomes have been reported following
inadvertent administration of intravenous
recombinant tissue plasminogen activator to
patients receiving AAT who presented to the
emergency department with stroke-like symptoms.49
Patients and their relatives should inform attending
physicians of ongoing AAT, and brain MRI should
be arranged as early as possible when clinically
indicated.49 An alert within the electronic medical
record system or provision of a drug card may
enable attending doctors, particularly emergency
physicians, to be promptly informed of treatment
status. Physicians planning to initiate local AAT
services are also encouraged to notify colleagues in
the stroke team and emergency department.49
Multidisciplinary advice
Statement 25: A multidisciplinary approach is
recommended when administering anti-amyloid
therapies. This approach involves clinicians
experienced in the management of cognitive
impairment (eg, geriatricians, neurologists,
psychiatrists, nuclear medicine physicians, and
radiologists).
Experienced clinicians (eg, geriatricians,
neurologists and psychiatrists) can identify
suitable candidates, arrange baseline evaluations,
including apolipoprotein E genotyping, amyloid
PET imaging and brain MRI, assess the severity of
cognitive impairment, and manage behavioural
and psychological symptoms. They can also advise
on the management of ARIA, particularly in severe
amyloid-related imaging abnormalities–oedema/effusion, which may require pulse corticosteroids,
immunosuppressive therapy, or seizure
management.6 11 Nuclear medicine physicians
play a key role in reviewing amyloid and tau PET
imaging and interpreting treatment-related amyloid
clearance. Radiologists are important for monitoring
ARIA and reviewing baseline imaging, including assessment of the number of microhaemorrhages,
the presence of superficial siderosis, and the number
of lacunar infarcts.6 11
Infusion reactions
Statement 26: Infusion reactions, including
hypersensitivity and acute symptoms such as fever,
chills, or nausea, may occur during administration
of anti-amyloid therapies. Protocols should be
developed to manage various infusion reactions
so that medical staff in wards or day centres can
respond promptly and reduce the risk of recurrence.
Management of infusion reactions should follow
the relevant appropriate use recommendations.6 11
The development of clear protocols enables healthcare
professionals to address these manifestations with
confidence and to implement appropriate preventive
measures.6 11 General management may include
temporary interruption of infusion, intramuscular
adrenaline injection, bronchodilators, intravenous
hydrocortisone, second-generation antihistamines,
and paracetamol.6 11 Preventive measures for
future infusions may include premedication with
paracetamol or non-steroidal anti-inflammatory
drugs, second-generation antihistamines, or
intravenous hydrocortisone prior to the next AAT
infusion.6 11
Conclusion
It is hoped that this consensus statement will
provide as practical guidance for local clinicians in
the management of patients with AD. Regarding
biomarkers in the diagnosis of AD, amyloid PET
and CSF biomarkers remain the surrogate gold
standards, while other biomarkers that achieve a
minimum sensitivity and specificity of at least 90%
in the intended population may also be used. For the
administration of AAT, clinicians should be aware
of contraindications, and relevant risks should be
explained to patients and their caregivers. For future
implementation of AAT in public system, accessibility
to AD biomarkers (eg, plasma biomarkers and
amyloid PET), MRI capacity for ARIA monitoring,
healthcare infrastructure to support the new
treatment, and cost-effectiveness considerations are
obstacles that need to be addressed.
Author contributions
All authors contributed to the concept or design, drafting of
the manuscript, and critical revision of the manuscript for
important intellectual content. All authors had full access to
the data, contributed to the study, approved the final version
for publication, and take responsibility for its accuracy and
integrity.
Conflicts of interest
YF Shea reported acting as a member of the Eisai Hong Kong Lecanemab Advisory Board in 2024. Other authors disclosed no conflicts of interest.
Acknowledgement
The authors thank Dr Colin Hing-tung Lui, President of the
Hong Kong Neurological Society, and Dr John Boom-ting
Kung, President of Hong Kong Society of Nuclear Medicine
and Molecular Imaging, for their coordination of panellist
selection.
Funding/support
This study received no specific grant from any funding agency
in the public, commercial, or not-for-profit sectors.
References
1. Sims JR, Zimmer JA, Evans CD, et al. Donanemab in early symptomatic Alzheimer disease: the TRAILBLAZER-ALZ 2 randomized clinical trial. JAMA 2023;330:512-27. Crossref
2. van Dyck CH, Swanson CJ, Aisen P, et al. Lecanemab in early Alzheimer’s disease. N Engl J Med 2023;388:9-21. Crossref
3. Palmqvist S, Tideman P, Mattsson-Carlgren N, et al. Blood biomarkers to detect Alzheimer disease in primary care and secondary care. JAMA 2024;332:1245-57. Crossref
4. Cogswell PM, Andrews TJ, Barakos JA, et al. Alzheimer disease anti-amyloid immunotherapies: imaging recommendations and practice considerations for monitoring of amyloid-related imaging abnormalities. AJNR Am J Neuroradiol 2025;46:24-32. Crossref
5. Corriveau-Lecavalier N, Botha H, Graff-Radford J, et al. Clinical criteria for a limbic-predominant amnestic neurodegenerative syndrome. Brain Commun 2024;6:fcae183. Crossref
6. Cummings J, Apostolova L, Rabinovici GD, et al. Lecanemab: appropriate use recommendations. J Prev Alzheimers Dis 2023;10:362-77. Crossref
7. Dubois B, Villain N, Schneider L, et al. Alzheimer disease as a clinical-biological construct—an international working group recommendation. JAMA Neurol 2024;81:1304-11. Crossref
8. Greenberg SM, Aparicio HJ, Furie KL, et al. Vascular neurology considerations for antiamyloid immunotherapy: a science advisory from the American Heart Association. Stroke 2025;56:e30-8. Crossref
9. Jack CR Jr, Andrews JS, Beach TG, et al. Revised criteria for diagnosis and staging of Alzheimer’s disease: Alzheimer’s Association Workgroup. Alzheimers Dement 2024;20:5143-69. Crossref
10. Rabinovici GD, Knopman DS, Arbizu J, et al. Updated appropriate use criteria for amyloid and tau PET: a report from the Alzheimer’s Association and Society for Nuclear Medicine and Molecular Imaging Workgroup. J Nucl Med 2025;66:S5-31. Crossref
11. Rabinovici GD, Selkoe DJ, Schindler SE, et al. Donanemab: appropriate use recommendations. J Prev Alzheimers Dis 2025;12:100150. Crossref
12. Schindler SE, Galasko D, Pereira AC, et al. Acceptable performance of blood biomarker tests of amyloid pathology—recommendations from the Global CEO Initiative on Alzheimer’s Disease. Nat Rev Neurol 2024;20:426-39. Crossref
13. Palmqvist S, Whitson HE, Allen LA, et al. Alzheimer’s Association clinical practice guideline on the use of blood-based biomarkers in the diagnostic workup of suspected Alzheimer’s disease within specialized care settings. Alzheimers Dement 2025;21:e70535. Crossref
14. Mielke MM, Fowler NR. Alzheimer disease blood biomarkers: considerations for population-level use. Nat Rev Neurol 2024;20:495-504. Crossref
15. Olsson B, Lautner R, Andreasson U, et al. CSF and blood biomarkers for the diagnosis of Alzheimer’s disease: a systematic review and meta-analysis. Lancet Neurol 2016;15:673-84. Crossref
16. Shea YF, Pan Y, Mak HK, et al. A systematic review of atypical Alzheimer’s disease including behavioural and psychological symptoms. Psychogeriatrics 2021;21:396-406. Crossref
17. Teunissen CE, Verberk IM, Thijssen EH, et al. Blood-based biomarkers for Alzheimer’s disease: towards clinical implementation. Lancet Neurol 2022;21:66-77. Crossref
18. Benedet AL, Brum WS, Hansson O, et al. The accuracy and robustness of plasma biomarker models for amyloid PET positivity. Alzheimers Res Ther 2022;14:26. Crossref
19. Cecchetti G, Agosta F, Rugarli G, et al. Diagnostic accuracy of automated Lumipulse plasma pTau-217 in Alzheimer’s disease: a real-world study. J Neurol 2024;271:6739-49. Crossref
20. Chiu HF, Lee HC, Chung WS, Kwong PK. Reliability and validity of the Cantonese version of Mini–Mental State Examination—a preliminary study. J Hong Kong Coll Psychiair 1994;4:25-8.
21. Collij LE, Bollack A, La Joie R, et al. Centiloid recommendations for clinical context-of-use from the AMYPAD consortium. Alzheimers Dement 2024;20:9037-48. Crossref
22. Dyer AH, Dolphin H, O’Connor A, et al. Performance of plasma p-tau217 for the detection of amyloid-beta positivity in a memory clinic cohort using an electrochemiluminescence immunoassay. Alzheimers Res Ther 2024;16:186. Crossref
23. Hayashida Y, Kakeda S, Hiai Y, et al. Diagnosis of
intracranial hemorrhagic lesions: comparison between
3D-SWAN (3D T2*-weighted imaging with multi-echo
acquisition) and 2D-T2*-weighted imaging. Acta Radiol 2014;55:201-7. Crossref
24. Jiang Y, Zhou X, Ip FC, et al. Large-scale plasma proteomic profiling identifies a high-performance biomarker panel for Alzheimer’s disease screening and staging. Alzheimers Dement 2022;18:88-102. Crossref
25. Mattsson-Carlgren N, Collij LE, Stomrud E, et al. Plasma biomarker strategy for selecting patients with Alzheimer disease for antiamyloid immunotherapies. JAMA Neurol 2024;81:69–78. Crossref
26. Mendes AJ, Ribaldi F, Lathuiliere A, et al. Head-to-head study of diagnostic accuracy of plasma and cerebrospinal fluid p-tau217 versus p-tau181 and p-tau231 in a memory clinic cohort. J Neurol 2024;271:2053-66. Crossref
27. Nakamura A, Kaneko N, Villemagne VL, et al. High performance plasma amyloid- β biomarkers for Alzheimer’s disease. Nature 2018;554:249-54. Crossref
28. Petersen RC, Smith GE, Waring SC, Ivnik RJ, Tangalos EG, Kokmen E. Mild cognitive impairment: clinical characterization and outcome. Arch Neurol 1999;56:303-8. Crossref
29. Rabe C, Bittner T, Jethwa A, et al. Clinical performance and robustness evaluation of plasma amyloid- β42/40 prescreening. Alzheimers Dement 2023;19:1393-402. Crossref
30. Rabinovici GD, Gatsonis C, Apgar C, et al. Association of amyloid positron emission tomography with subsequent change in clinical management among Medicare beneficiaries with mild cognitive impairment or dementia. JAMA 2019;321:1286-94. Crossref
31. Shea YF, Chu LW, Zhou L, et al. Cerebrospinal fluid biomarkers of Alzheimer’s disease in Chinese patients: a pilot study. Am J Alzheimers Dis Other Demen 2013;28:769-75. Crossref
32. Tetzloff KA, Graff-Radford J, Martin PR, et al. Regional distribution, asymmetry, and clinical correlates of tau uptake on [18F]AV-1451 PET in atypical Alzheimer’s disease. J Alzheimers Dis 2018;62:1713-24. Crossref
33. Therriault J, Servaes S, Tissot C, et al. Equivalence of plasma p-tau217 with cerebrospinal fluid in the diagnosis of Alzheimer’s disease. Alzheimers Dement 2023;19:4967-77. Crossref
34. Thijssen EH, La Joie R, Wolf A, et al. Diagnostic value of plasma phosphorylated tau181 in Alzheimer’s disease and frontotemporal lobar degeneration. Nat Med 2020;26:387-97. Crossref
35. Wong A, Law LS, Liu W, et al. Montreal Cognitive Assessment: one cutoff never fits all. Stroke 2015;46:3547-50. Crossref
36. Kwon HS, Lee EH, Kim HJ, et al. Predicting amyloid PET positivity using plasma p-tau181 and other blood-based biomarkers. Alzheimers Dement (Amst) 2023;15:e12502. Crossref
37. Karikari TK, Pascoal TA, Ashton NJ, et al. Blood phosphorylated tau 181 as a biomarker for Alzheimer’s disease: a diagnostic performance and prediction modelling study using data from four prospective cohorts. Lancet Neurol 2020;19:422–33. Crossref
38. Cheung CY, Ran AR, Wang S, et al. A deep learning model for detection of Alzheimer’s disease based on retinal photographs: a retrospective, multicentre case-control study. Lancet Digit Health 2022;4:e806-15. Crossref
39. Fan X, Cai Y, Zhao L, et al. Machine learning–derived MRI-based neurodegeneration biomarker for Alzheimer’s disease: a multi-database validation study. J Alzheimers Dis 2024;97:883-93. Crossref
40. Liu W, Au LW, Abrigo J, et al. MRI-based Alzheimer’s disease-resemblance atrophy index in the detection of preclinical and prodromal Alzheimer’s disease. Aging (Albany NY) 2021;13:13496-514. Crossref
41. Jiang Y, Uhm H, Ip FC, et al. A blood-based multipathway biomarker assay for early detection and staging of Alzheimer’s disease across ethnic groups. Alzheimers Dement 2024;20:2000-15. Crossref
42. Huang KL, Hsiao IT, Huang CW, et al. The Taiwan-ADNI workflow toward integrating plasma p-tau217 into prediction models for the risk of Alzheimer’s disease and tau burden. Alzheimers Dement 2025;21:e14297. Crossref
43. Thijssen EH, La Joie R, Strom A, et al. Plasma phosphorylated tau 217 and phosphorylated tau 181 as biomarkers in Alzheimer’s disease and frontotemporal lobar degeneration: a retrospective diagnostic performance study. Lancet Neurol 2021;20:739-52. Crossref
44. Ashton NJ, Brum WS, Di Molfetta G, et al. Diagnostic accuracy of a plasma phosphorylated tau 217 immunoassay for Alzheimer disease pathology. JAMA Neurol 2024;81:255-63. Crossref
45. Figdore DJ, Griswold M, Bornhorst JA, et al. Optimizing cutpoints for clinical interpretation of brain amyloid status using plasma p-tau217 immunoassays. Alzheimers Dement 2024;20:6506-16. Crossref
46. Kivisäkk P, Fatima HA, Cahoon DS, et al. Clinical evaluation of a novel plasma pTau217 electrochemiluminescence immunoassay in Alzheimer’s disease. Sci Rep 2024;14:629. Crossref
47. Thanapornsangsuth P, Booncharoen K, Khieukhajee J, et al. The Bayesian approach for real-world implementation of plasma p-tau217 in tertiary care memory clinics in Thailand. Alzheimers Dement 2024;20:6456-67. Crossref
48. Arranz J, Zhu N, Rubio-Guerra S, et al. Diagnostic performance of plasma pTau217, pTau181, Aβ1-42 and Aβ1-40 in the LUMIPULSE automated platform for the detection of Alzheimer disease. Alzheimers Res Ther 2024;16:139. Crossref
49. Reish NJ, Jamshidi P, Stamm B, et al. Multiple cerebral hemorrhages in a patient receiving lecanemab and treated with t-PA for stroke. N Engl J Med 2023;388:478-9. Crossref
50. Jünger S, Payne SA, Brine J, Radbruch L, Brearley SG. Guidance on Conducting and REporting DElphi Studies (CREDES) in palliative care: recommendations based on a methodological systematic review. Palliat Med 2017;31:684-706. Crossref
51. Centre for Evidence-Based Medicine. OCEBM Levels of Evidence. Available from: https://www.cebm.ox.ac.uk/resources/levels-of-evidence/ocebm-levels-of-evidence. Accessed 23 Mar 2026.
52. United States Food and Drug Administration. FDA clears first blood test used in diagnosing Alzheimer’s disease. New test provides less invasive option, reduces reliance on PET scans and increases diagnosis accessibility [press release]. 2025 May 16. Available from: https://www.fda.gov/news-events/press-announcements/fda-clears-first-blood-test-used-diagnosing-alzheimers-disease. Accessed 1 Sep 2025.
53. Biogen. LEQEMBI (Lecanemab) approved for the treatment of Alzheimer’s disease in China [press release]. 2024 January 9. Available from: https://investors.biogen.com/news-releases/news-release-details/leqembir-lecanemab-approved-treatment-alzheimers-disease-china. Accessed 1 Sep 2025.
54. Eli Lilly. Lilly’s Kisunla (donanemab-azbt) approved in China for the treatment of early symptomatic Alzheimer’s disease [press release]. 2024 December 17. Available from: https://investor.lilly.com/news-releases/news-release-details/lillys-kisunlatm-donanemab-azbt-approved-china-treatment-early. Accessed 1 Sep 2025.