Hong Kong Med J 2026;32:Epub 15 Apr 2026
© Hong Kong Academy of Medicine. CC BY-NC-ND 4.0
ORIGINAL ARTICLE
Descriptive analysis of platelet-rich plasma
injection therapy in chronic musculoskeletal pain
Mandy HM Chu, MB, ChB, FANZCA1; WS Chan, FANZCA, FHKCA (Pain Medicine)2; Ara CY Li, FANZCA, FHKCA (Pain Medicine)3; Henry MK Wong, MB, ChB, FANZCA1; HL Wong, HDip, BSc1; KM Ho, FANZCA, FCICM1
1 Department of Anaesthesia and Intensive Care, The Chinese University
of Hong Kong, Hong Kong SAR, China
2 Peter Hung Pain Specialist Clinic, CUHK Medical Centre, Hong Kong SAR, China
3 Department of Anaesthesia, Pain and Perioperative Medicine, Prince of
Wales Hospital, Hong Kong SAR, China
Corresponding author: Prof Mandy HM Chu (hiumanchu@cuhk.edu.hk)
Abstract
Introduction: Platelet-rich plasma (PRP) injections
have been used to manage various chronic pain
conditions. However, evidence remains limited due
to poor standardisation across practices. In this
descriptive study, we aimed to characterise current
PRP practice patterns at a university-affiliated
private pain clinic in Hong Kong, focusing on case
mix and treatment outcomes in patients with chronic
musculoskeletal pain.
Methods: This retrospective descriptive
study included patients with diverse chronic
musculoskeletal pain conditions aged 18 years or
older who attended the Peter Hung Pain Specialist
Clinic and received PRP injection therapy between
January 2023 and December 2024. Improvements in
pain and changes in oral analgesic use were recorded.
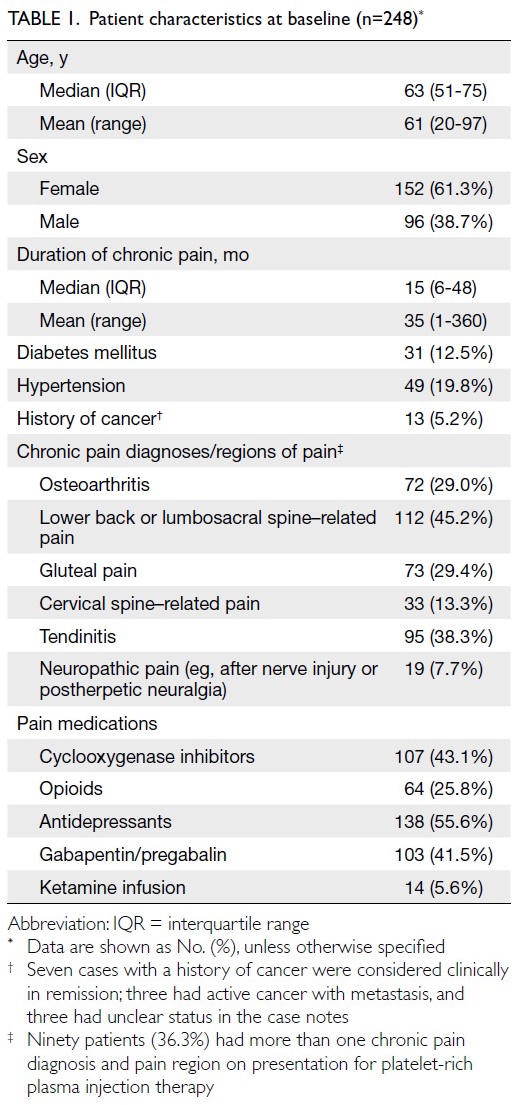
Results: In total, 248 patients were included. Prior
to PRP treatment, over 70% required multiple oral
analgesics for pain control, including 55.6% taking
antidepressants, 41.5% gabapentin or pregabalin,
and 25.8% oral opioids. At first follow-up (median:
4 weeks, range: 1-20), more than 60% reported
‘moderate’ or ‘much’ improvement in pain symptoms.
By 12 months post-treatment, fewer than 10% of
patients in each category continued to require oral opioids, antidepressants, gabapentin, or pregabalin.
Of the 26 patients (10.5%) who required a second
PRP session, only one reported no improvement.
Conclusion: These results highlight the potential
utility of PRP in managing chronic musculoskeletal
pain and underscore the need for randomised
controlled trials to confirm its long-term impact on
quality of life of patients.
New knowledge added by this study
- Musculoskeletal pain is a common clinical manifestation in Hong Kong.
- Leukocyte-rich platelet-rich plasma (PRP) provided convincing pain improvement across various types of musculoskeletal pain, with reduction or discontinuation of oral analgesics.
- The effect of PRP injection was more pronounced in patients with a shorter duration of chronic pain.
- Randomised controlled trials with standardised PRP preparation in specific patient groups are needed.
- Platelet-rich plasma injection may be beneficial for chronic pain; however, further evidence is required.
Introduction
Platelet-rich plasma (PRP) refers to plasma with
a platelet concentration higher than that found in
whole blood. It is classified into four types based
on its leukocyte and fibrin content: leukocyte-rich
or -poor, and fibrin-rich or -poor.1 Initially used by
haematologists in the 1970s as platelet transfusions
for thrombocytopenia, PRP gained traction in the
1980s in maxillofacial surgery and sports medicine
due to its potential anti-inflammatory effects.2 Since
then, its applications have extended to regenerative
medicine and pain management because of its abundance of growth factors and cytokines.3
Despite its widespread use in degenerative and pain
conditions—such as osteoarthritis, low back pain,
and tendinitis—evidence for PRP efficacy in humans
remains limited and controversial.4 5 Understanding
of PRP practice and efficacy among anaesthetists is
particularly scarce. This study aimed to characterise
current PRP practice patterns at a university-affiliated
private pain clinic in Hong Kong, focusing
on case mix and treatment outcomes in patients
with chronic musculoskeletal pain.
Methods
Study population
This study included patients aged 18 years or older
who attended the Peter Hung Pain Specialist Clinic
and received PRP injection therapy for chronic pain
between January 2023 and December 2024. Patients
were excluded if they were younger than 18 years,
had been diagnosed with cancer-related pain, or did
not proceed with PRP therapy after pain assessment.
Leukocyte-rich PRP was prepared by
collecting autologous blood and subjecting it to
two centrifugation cycles using sterile technique
and an Eppendorf Centrifuge 5702 (Eppendorf
SE, Hamburg, Germany). Whole blood was first
centrifuged at 3800 rpm for 2 minutes to sediment
red blood cells and form a buffy coat-rich plasma
layer, without excessive platelet loss into the
erythrocyte fraction. The plasma/buffy coat fraction
was then transferred and centrifuged again at 3800
rpm for 5 minutes, applying the same centrifugal
force in longer duration to this less viscous,
cell-reduced plasma to achieve further platelet
sedimentation and centration while minimising
platelet damage or premature activation. The same
force with longer duration is considered having the
same effect as higher force with shorter duration
by the manufacturer. Each PRP injection session targeted all painful regions deemed suitable by the
same attending pain specialist under monitored
anaesthesia care or general anaesthesia.
Data were extracted from the pain clinic and
hospital databases, including patient age, sex, sites
and types of chronic pain, duration of pain prior
to PRP therapy, use of pain medications, history of
surgical or interventional procedures for pain, and
the indications, locations, and dosage for each PRP
treatment. Standard pain assessment for patients
undergoing PRP therapy included evaluation of
overall and site-specific pain improvement at follow-up
visits. In this study, pain improvement at the
first follow-up after PRP therapy was specifically
assessed and categorised as ‘no improvement’,
‘mildly improved’, ‘moderately improved’, or ‘much
improved’. Additional outcomes included the
proportion of patients able to discontinue oral
analgesics and identification of factors associated
with a favourable pain relief response to PRP therapy.
Statistical analyses
Results are presented as numbers (with percentages)
and medians (with interquartile ranges [IQRs] and
ranges). Categorical variables before and after PRP
therapy were compared using the McNemar test.
One-way analysis of variance was used to assess the
association between the duration of chronic pain
prior to PRP therapy and the likelihood of reporting
‘much improved’ pain at the first follow-up. Logistic
regression analysis was conducted to determine
whether specific pain pathologies or anatomical
regions were associated with better responses to
PRP therapy. All statistical tests were two-tailed
and performed using SPSS (Windows version
29.0; IBM Corp, Armonk [NY], United States). A P
value <0.05 was considered statistically significant.
Results
A total of 248 patients aged 20 to 97 years who
received at least one session of PRP therapy during
the study period were included. Of these, 61.3% were
women. The duration of pain prior to PRP therapy
varied widely, ranging from 1 month to over 20 years,
with a median of 15 months. More than 70.2% of
patients (n=174) were taking two or more analgesics
before PRP therapy. Fourteen patients (5.6%) had
previously received ketamine infusions for pain; two
of these had undergone interventional procedures
such as radiofrequency thermocoagulation of
the trigeminal ganglion, ultrasound- and X-ray–guided radiofrequency with injection to the left
glossopharyngeal nerve, and X-ray–guided steroid
injection at the L4/5 level of the lumbar spine.
Ninety patients (36.3%) reported pain in more than
one anatomical region. Baseline characteristics,
including analgesic use, are summarised in Table 1.
Platelet-rich plasma therapy was administered
to 404 anatomical sites across the 248 patients. The
median volume of PRP injected during the first
session was 17.0 mL (mean=18.3, IQR=10.0-20.5;
range, 2-50). Twenty-six patients (10.5%) required
more than one PRP session, with a median interval
of 4.0 months (mean=4.0, IQR=2.8-5.0; range, 1-15).
Seven patients (2.8%) received three sessions during
the 2-year study period.
The median time to first follow-up after PRP
therapy was 4.0 weeks (mean=4.5, IQR=4.0-4.5;
range, 1-20). Thirty patients (12.1%) did not return
for follow-up. Among the remaining 218 patients,
over 60% reported their pain as either ‘moderately
improved’ or ‘much improved’. The distribution of pain relief levels is shown in Table 2. Among all
factors assessed, only the duration of chronic pain
prior to PRP therapy was significantly associated
with the likelihood of reporting ‘much improved’
pain at first follow-up. Specifically, longer pain
duration was inversely associated with improvement
(odds ratio=0.91 per 6-month increment in pain
duration prior to PRP therapy, 95% confidence
interval=0.85-0.98; P=0.008) [Table 3]. Patients with
chronic pain lasting less than 2 years appeared to
respond best to PRP therapy (Fig). The volume of
PRP injected was not significantly associated with
reporting ‘much improved’ pain.
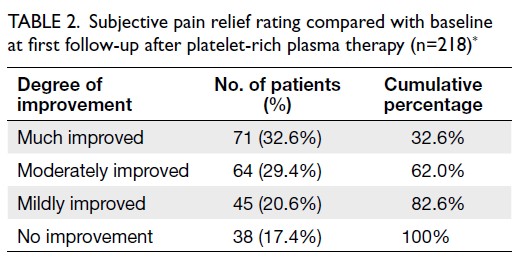
Table 2. Subjective pain relief rating compared with baseline at first follow-up after platelet-rich plasma therapy (n=218)
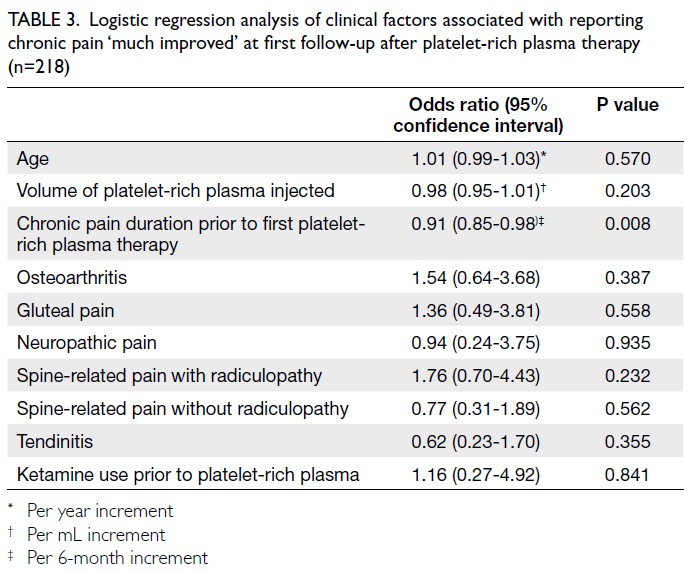
Table 3. Logistic regression analysis of clinical factors associated with reporting chronic pain ‘much improved’ at first follow-up after platelet-rich plasma therapy (n=218)
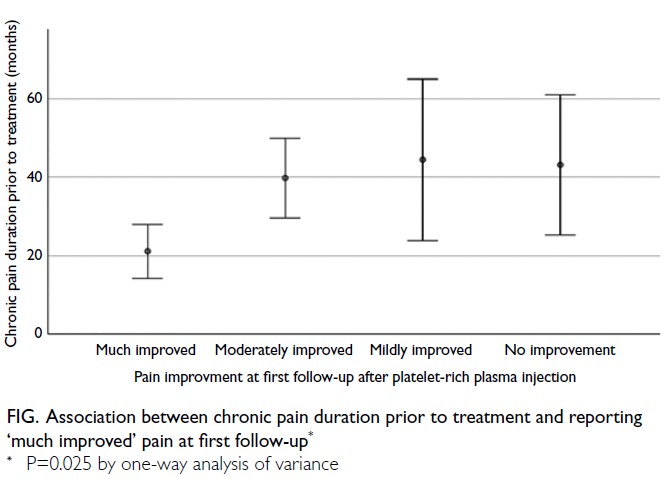
Figure. Association between chronic pain duration prior to treatment and reporting ‘much improved’ pain at first follow-up
The median time to second follow-up was 9.0
weeks (mean=9.7, IQR=8.0-11.0; range, 2-32). Over
the 12-month period after PRP therapy, a substantial
number of patients were able to discontinue oral
analgesics (Table 4).
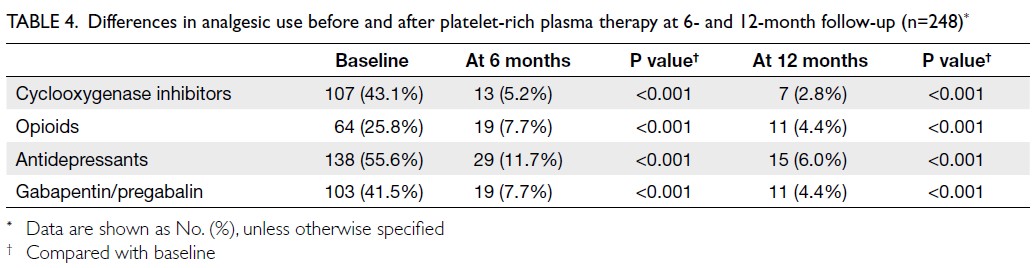
Table 4. Differences in analgesic use before and after platelet-rich plasma therapy at 6- and 12-month follow-up (n=248)
Discussion
In this descriptive study, over 60% of patients
reported moderate to significant improvement
in pain symptoms after their first PRP treatment
session. Among the 26 patients who received a
second session, only one reported no improvement,
suggesting a favourable response to repeated
treatment. This improvement was accompanied by
a substantial reduction in the use of oral analgesics.
Given the known adverse effects associated
with polypharmacy—particularly involving
antidepressants, gabapentinoids, and opioids—this
reduction may contribute to improved quality of life
of patients.
Our cohort included patients with a wide
range of chronic pain conditions affecting various
anatomical sites. Intriguingly, there was no apparent
correlation between the number of pain sites
and the degree of pain relief, suggesting that PRP
may have broad applicability across multiple pain
syndromes. However, due to the heterogeneity of
pain presentations and the presence of multiple
pain regions in many patients, we were unable to
determine whether PRP was more effective for
specific types or anatomical regions of pain. This
highlights the need for future randomised controlled trials involving more homogeneous patient
populations to confirm the long-term impact of PRP
on quality of life according to pain pathology.
Although PRP therapy is considered a
form of regenerative therapy,3 its mechanisms
of action are not yet fully understood.5 Platelets
contain granules that release a variety of bioactive
substances, including growth factors, antimicrobial
proteins, metalloproteases, coagulation factors, and
membrane glycoproteins that influence the synthesis
of interleukins and chemokines. Other bioactive
molecules, including neurotransmitters such as
serotonin, dopamine, adenosine diphosphate,
adenosine triphosphate, and histamine, may also
play roles in tissue modulation and regeneration.4
Some research suggests that leukocyte-rich
PRP has stronger anti-inflammatory effects and
higher concentrations of growth factors, which
may be important for conditions such as knee
osteoarthritis.6 7 However, evidence as to whether
leukocyte-rich PRP is superior to leukocyte-poor
PRP remains inconclusive. One of the largest
randomised controlled trials assessing PRP for knee
osteoarthritis concluded that leukocyte-poor PRP
was not significantly more effective than placebo
in improving symptoms or joint structure among
patients with mild to moderate knee osteoarthritis
over 12 months.8 Conversely, a recent systematic
review and meta-analysis found that leukocyte-poor
PRP provided moderate pain relief compared with
other active treatments; no significant difference
was observed between leukocyte-rich PRP and other
therapies.5 In the present study, all patients received
leukocyte-rich PRP; therefore, we were unable to
compare the efficacy of different PRP formulations.
The data showed that 13 patients with a
history of cancer (either active with metastasis, in
remission, or with unclear status) received PRP for
pain clinically unrelated to their cancers (Table 1).
Currently, there is no strong evidence regarding the
safety of PRP use in patients with cancer. A recent
formal consensus from the International Research
Group on Platelet Injections recommended that
PRP may be performed in patients with cancers in
remission or with metastasis—after discussion with
an oncologist—although the supporting evidence
is contradictory or inconclusive and largely based
on expert opinion, with very limited or absent
literature.9
Limitations
This study has several limitations. First, without a
control group, placebo effects cannot be excluded.
Second, PRP dosing was not standardised; the volume
administered varied according to the number and
size of pain sites, and detailed documentation of per-site
dosing was unavailable. Additionally, over 10% of
patients did not return for follow-up, and reasons for
loss to follow-up were not documented, introducing
potential selection bias. The heterogeneity of pain
conditions further complicated data interpretation.
Although current evidence suggests that leukocyte-rich
PRP may cause greater initial flare-up than
leukocyte-poor PRP for intra-articular injections,10
any initial worsening of symptoms was not captured
in our study because of variability in the timing
of first follow-up. Finally, pain improvement
was assessed using non-standardised, subjective
descriptors (‘much improved’, ‘moderately improved’,
or ‘mildly improved’). These terms reflect patient
satisfaction but do not permit precise quantification
of pain reduction. Future studies should incorporate
validated quantitative assessment tools, such as the
Brief Pain Inventory, to enhance the reliability of
outcome measurement.
Conclusion
Leukocyte-rich PRP appeared effective in improving
chronic musculoskeletal pain. The majority of
patients reported meaningful symptom relief,
and many were able to reduce or discontinue oral
analgesics—an outcome that may substantially
improve quality of life, particularly given the adverse
effects associated with polypharmacy involving
antidepressants, gabapentinoids, and opioids. Well-designed
randomised controlled trials focusing on
chronic musculoskeletal pain of less than 2 years’
duration—and incorporating standardised protocols
for leukocyte-rich PRP preparation, injection volume,
and patient selection criteria—are needed to confirm
its long-term impact on quality of life of patients.
Author contributions
Concept or design: MHM Chu, WS Chan, KM Ho.
Acquisition of data: WS Chan, HL Wong.
Analysis or interpretation of data: MHM Chu, KM Ho.
Drafting of the manuscript: MHM Chu, KM Ho.
Critical revision of the manuscript for important intellectual content: WS Chan, ACY Li, HMK Wong.
Acquisition of data: WS Chan, HL Wong.
Analysis or interpretation of data: MHM Chu, KM Ho.
Drafting of the manuscript: MHM Chu, KM Ho.
Critical revision of the manuscript for important intellectual content: WS Chan, ACY Li, HMK Wong.
All authors had full access to the data, contributed to the study, approved the final version for publication, and take responsibility for its accuracy and integrity.
Conflicts of interest
All authors have disclosed no conflicts of interest.
Acknowledgement
We are grateful to the staff in Peter Hung Pain Specialist
Clinic and Operating Theatre of CUHK Medical Centre for
their assistance in patient care and study logistics.
Funding/support
This research received no specific grant from any funding
agency in the public, commercial, or not-for-profit sectors.
Ethics approval
This research was approved by the Clinical Research Ethics
Committee of CUHK Medical Centre, Hong Kong (Ref No.:
CREC-202409). A waiver of patient consent was approved by
the Committee due to the retrospective and observational
nature of the study. Only de-identified data were used for
analysis.
References
1. Arnoczky SP, Delos D, Rodeo SA. What is platelet-rich plasma? Oper Tech Sports Med 2011;19:142-8.
Crossref
2. Cao Y, Zhu X, Zhou R, He Y, Wu Z, Chen Y. A narrative review of the research progress and clinical application of platelet-rich plasma. Ann Palliat Med 2021;10:4823-9.
Crossref
3. Arita A, Tobita M. Current status of platelet-rich plasma therapy under the act on the safety of regenerative medicine in Japan. Regen Ther 2023;23:37-43.
Crossref
4. Cole BJ, Seroyer ST, Filardo G, Bajaj S, Fortier LA. Platelet-rich plasma: where are we now and where are we going? Sports Health 2010;2:203-10.
Crossref
5. Wang F, Meng F, Chan TC, Wong SS. Platelet-rich plasma for treating chronic noncancer pain: a systematic review and meta-analysis of randomized controlled trials. Pain Ther 2025;14:1169-88.
Crossref
6. Jayaram P, Mitchell PJ, Shybut TB, Moseley BJ, Lee B. Leukocyte-rich platelet-rich plasma is predominantly anti-inflammatory compared with leukocyte-poor platelet-rich plasma in patients with mild-moderate knee osteoarthritis: a prospective, descriptive laboratory study. Am J Sports Med 2023;51:2133-40.
Crossref
7. Lin KY, Chen P, Chen AC, Chan YS, Lei KF, Chiu CH. Leukocyte-rich platelet-rich plasma has better stimulating effects on tenocyte proliferation compared with leukocyte-poor platelet-rich plasma. Orthop J Sports Med 2022;10:23259671221084706.
Crossref
8. Bennell KL, Paterson KL, Metcalf BR, et al. Effect of intra-articular platelet-rich plasma vs placebo injection on pain and medial tibial cartilage volume in patients with knee osteoarthritis: the RESTORE randomized clinical trial. JAMA 2021;326:2021-30.
Crossref
9. Eymard F, Louati K, Noël É, et al. Indications and contraindications to platelet-rich plasma injections in musculoskeletal diseases in case of infectious, oncological and haematological comorbidities: a 2025 formal consensus from the GRIIP (International Research Group on Platelet Injections). Knee Surg Sports Traumatol Arthrosc 2025;33:2293-306.
Crossref
10. Lu J, Li H, Zhang Z, Xu R, Wang J, Jin H. Platelet-rich plasma in the pathologic processes of tendinopathy: a review of basic science studies. Front Bioeng Biotechnol 2023;11:1187974.
Crossref