Hong Kong Med J 2026;32:Epub 15 Apr 2026
© Hong Kong Academy of Medicine. CC BY-NC-ND 4.0
CASE REPORT
Subacute or chronic neurological toxicity
following acute diquat poisoning: a case report
Xiaojun Jin, MD1,2; Yuanqiang Lu, MD, PhD1,2
1 Department of Emergency Medicine, The First Affiliated Hospital, Zhejiang University School of Medicine, Zhejiang, China
2 Zhejiang Key Laboratory for Diagnosis and Treatment of Physic-chemical and Aging-related Injuries, Zhejiang, China
Corresponding author: Prof Yuanqiang Lu (luyuanqiang@zju.edu.cn)
Case presentation
In January 2025, a 40-year-old male with no significant
medical history ingested 200 mL of diquat (DQ) and
required urgent medical intervention. He presented
with dizziness, fatigue, and throat discomfort.
Treatment at a local centre in China included gastric
lavage, cathartics, fluid resuscitation, and alternating
haemoperfusion (4-hour sessions) and continuous
renal replacement therapy (20-hour cycles).
Laboratory tests revealed a serum creatinine level of
187 μmol/L and elevated DQ concentrations in both
blood (122 ng/mL) and urine (2535 ng/mL).
Three days post-ingestion, the patient was
transferred to our emergency department with
ongoing throat discomfort. Subsequent laboratory
tests revealed a further increase in DQ levels
(205.66 ng/mL in blood and 4278.52 ng/mL in urine),
while paraquat (PQ) levels were undetectable. A
computed tomography scan of the brain (Fig 1a)
showed no abnormalities. Due to the lack of a
specific antidote for DQ toxicity, management
prioritised enhanced elimination through sustained
haemoperfusion-continuous renal replacement
therapy cycling (a 7-day course until blood DQ was undetectable), along with corticosteroids
(methylprednisolone 80 mg intravenously every
8 hours, tapered gradually over a 4-week period)
and antioxidant therapy (intravenous reduced
glutathione 1.2 g once daily until discharge). Lumbar
puncture on days 4 and 16 indicated elevated
intracranial pressure (230-250 mm H2O), with
an initial cerebrospinal fluid (CSF) DQ level of
111.06 ng/mL, which later became undetectable.
Mannitol and a glycerol-fructose compound were
administered as dehydrating agents to manage
intracranial pressure, although follow-up magnetic
resonance imaging (MRI) of the brain showed
no pathological changes (Fig 1b). Following
comprehensive treatment, the patient was discharged
in a stable condition.
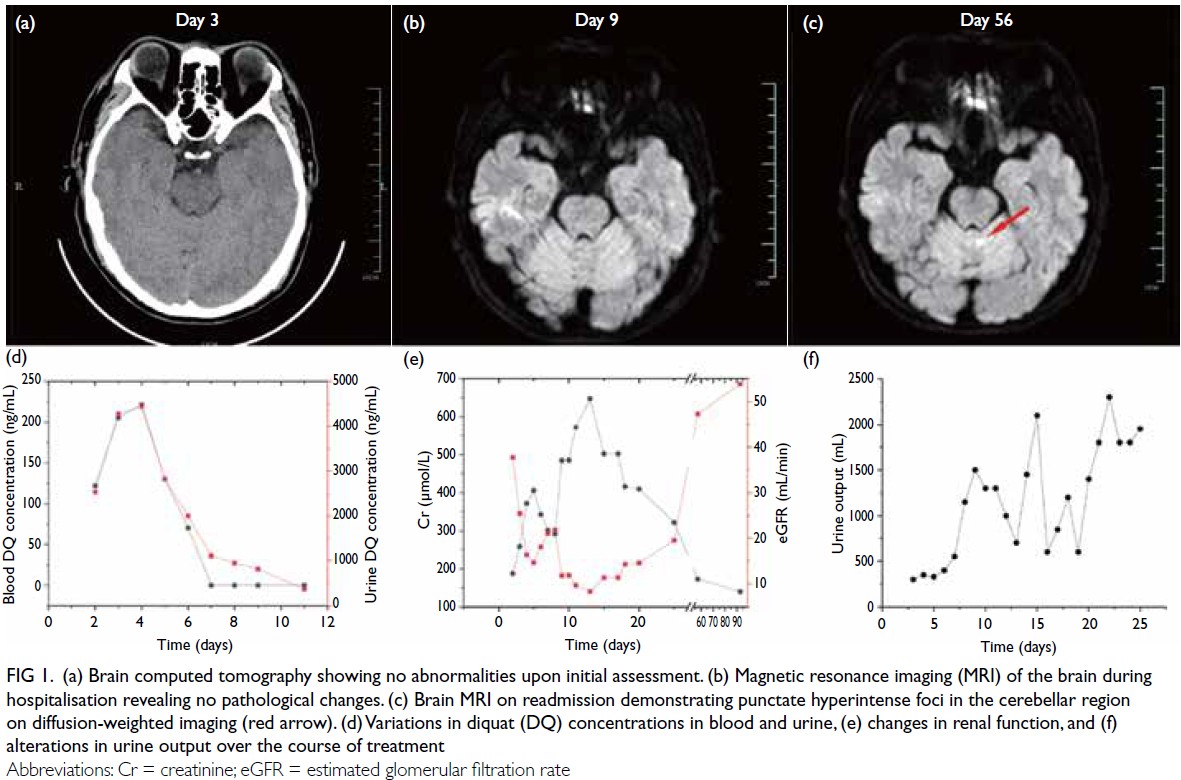
Figure 1. (a) Brain computed tomography showing no abnormalities upon initial assessment. (b) Magnetic resonance imaging (MRI) of the brain during hospitalisation revealing no pathological changes. (c) Brain MRI on readmission demonstrating punctate hyperintense foci in the cerebellar region on diffusion-weighted imaging (red arrow). (d) Variations in diquat (DQ) concentrations in blood and urine, (e) changes in renal function, and (f) alterations in urine output over the course of treatment
On day 57, the patient returned with
exacerbated headaches and intermittent blurred
vision that had developed over the preceding week.
Brain MRI revealed cerebellar hyperintensities (Fig 1c). Neurophysiological assessment demonstrated
a marked reduction in the right-sided P40-N50
amplitude of somatosensory evoked potentials
(0.86 μV vs 2.35 μV contralaterally, 63.4%
interhemispheric asymmetry), bilateral prolongation
of visual evoked potentials latencies (122 ms right
vs 126 ms left, 3.3% latency disparity), and left-lateralised
attenuation of wave I/III/V complex
amplitudes in auditory evoked potentials,
indicative of multisensory pathway dysfunction.
The patient commenced a 1-month course of oral
methylprednisolone (4 mg daily) combined with long-term neurotrophic support, including vitamin
B complex (1 tablet 3 times daily) and mecobalamin
(500 μg 3 times daily). By day 93 following exposure,
he reported less intense headaches, although
intermittent blurred vision persisted, alongside
new symptoms of anosmia and ageusia. Follow-up
assessments demonstrated partial improvement,
including somatosensory evoked potential right-sided
N50 amplitude recovery (right: 2.24 μV; left:
3.29 μV) and reduced right-sided visual evoked
potential latency (right: 114 ms; left: 128 ms). The
patient was advised to continue oral neurotrophic
therapy (vitamin B complex and mecobalamin),
with ongoing follow-up to monitor clinical recovery.
Changes in DQ levels in blood and urine are
illustrated in Figure 1d, while alterations in renal
function and urine output are depicted in Figure 1e and f, respectively. The patient’s clinical progression is summarised in Fig 2.
Discussion
Diquat is an organic herbicide characterised by a
heterocyclic structure and has increasingly replaced
PQ following the latter’s ban in China in 2016.1 This
regulatory shift has correlated with a rise in DQ
poisoning incidents. Although the toxicological
mechanisms of DQ are still being elucidated,
current hypotheses suggest that its harmful effects
may mimic those of PQ, while also producing
distinct organ-specific consequences. Although
DQ is systemically distributed, it primarily targets the kidneys, with markedly lower concentrations
detected in the brain.2 In previous clinical studies,
Yu et al3 and Zhou and Lu4 observed that exposure
to DQ can adversely affect the nervous system,
often presenting as acute toxic encephalopathy. The
onset of acute encephalopathy typically occurs 24
to 72 hours post-exposure. Research indicates that
the neuroinflammatory response triggered by DQ
involves multiple mechanisms, including oxidative
stress, mitochondrial dysfunction, and neuronal
degeneration.5 An experimental study6 demonstrated
that neuroinflammation plays a critical role in DQ-induced
toxic encephalopathy and is exacerbated
by disrupted autophagic processes in microglial
cells. While earlier investigations have focused on
the immediate and severe clinical manifestations
observed in hospital settings, the potential for
delayed neurological deficits during long-term
follow-up remains underexplored.
In the current case, the patient initially
displayed no significant neurological symptoms, and
both computed tomography and MRI scans of the
brain were unremarkable. Routine biochemical tests
revealed an isolated elevation of serum creatinine,
with all other parameters within normal ranges. To
further assess potential neurotoxicity, CSF analysis
from lumbar puncture was performed. Cerebrospinal
fluid profiles showed normal routine and biochemical
parameters, while serial measurements indicated a
rapid decline in DQ concentration. Nevertheless,
approximately 50 days post-exposure, the patient
began experiencing headaches, intermittent
blurred vision, and loss of smell and taste. This
case offers important insights into the neurological
complications following DQ exposure, highlighting
the potential for delayed deficits even when CSF DQ
levels fall below detectable thresholds. A limitation
of this report is its focus on a single case, which
limits the ability to draw broader conclusions or
conduct systematic investigations involving larger
patient cohorts.
Previous studies7 8 provides limited guidance
on the management of post-acute DQ toxicity, which
may contribute to misdiagnosis in patients presenting
with neurological or ophthalmic symptoms post-discharge.
This report highlights the urgent need
for healthcare professionals to recognise that DQ
poisoning can result in delayed subacute or chronic
neurological complications, emphasising the critical
importance of early detection, timely intervention,
and longitudinal follow-up to optimise long-term
outcomes.
Author contributions
Concept or design: Y Lu.
Acquisition of data: X Jin.
Analysis or interpretation of data: Both authors.
Drafting of the manuscript: X Jin.
Critical revision of the manuscript for important intellectual content: Both authors.
Acquisition of data: X Jin.
Analysis or interpretation of data: Both authors.
Drafting of the manuscript: X Jin.
Critical revision of the manuscript for important intellectual content: Both authors.
Both authors had full access to the data, contributed to the
study, approved the final version for publication, and take
responsibility for its accuracy and integrity.
Conflicts of interest
Both authors have disclosed no conflicts of interest.
Acknowledgement
The authors are grateful to Dr Mengxiao Feng (Department of
Emergency Medicine, The First Affiliated Hospital, Zhejiang
University School of Medicine) for her expertise in preparing
scientific figures.
Funding/support
This study received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethics approval
This study was approved by the Clinical Research Ethics
Committee of the First Affiliated Hospital, Zhejiang University
School of Medicine (Ref No.: 2025B-0359). Written informed
consent was obtained from the patient for publication of this
case report and any accompanying images.
References
1. Zhang Y, Chen X, Du H, Zhao M, Jiang X. Association between initial diquat plasma concentration, severity index and in-hospital mortality in patients with acute diquat poisoning: a retrospective cohort study. Clin Toxicol (Phila) 2024;62:557-63.
Crossref
2. Wu Y, Cui S, Wang W, Jian T, Kan B, Jian X. Kidney and lung injury in rats following acute diquat exposure. Exp Ther Med 2022;23:275.
Crossref
3. Yu G, Jian T, Cui S, Shi L, Kan B, Jian X. Acute diquat poisoning resulting in toxic encephalopathy: a report of three cases. Clin Toxicol (Phila) 2022;60:647-50.
Crossref
4. Zhou JN, Lu YQ. Lethal diquat poisoning manifests as acute central nervous system injury and circulatory failure: a retrospective cohort study of 50 cases. EClinicalMedicine 2022;52:101609.
Crossref
5. Ren Y, Guo F, Wang L. Imaging findings and toxicological mechanisms of nervous system injury caused by diquat. Mol Neurobiol 2024;61:9272-83.
Crossref
6. Wang P, Song CY, Lu X, et al. Diquat exacerbates oxidative stress and neuroinflammation by blocking the autophagic flux of microglia in the hippocampus. Ecotoxicol Environ Saf 2024;286:117188.
Crossref
7. Jones GM, Vale JA. Mechanisms of toxicity, clinical features, and management of diquat poisoning: a review. J Toxicol Clin Toxicol 2000;38:123-8.
Crossref
8. Magalhães N, Carvalho F, Dinis-Oliveira R. Human and experimental toxicology of diquat poisoning: toxicokinetics, mechanisms of toxicity, clinical features, and treatment. Hum Exp Toxicol 2018;37:1131-60.
Crossref

