Hong Kong Med J 2026;32:Epub 17 Apr 2026
© Hong Kong Academy of Medicine. CC BY-NC-ND 4.0
ORIGINAL ARTICLE
Prevalence of mild and major neurocognitive
disorders in community and residential care
homes in Hong Kong: considerations for
multidimensional risk factor evaluation and
intervention in primary care
Linda CW Lam, MD, FHKAM (Psychiatry)1; WC Chan, MB, ChB, FHKAM (Psychiatry)2; Allen TC Lee, MD, FHKAM (Psychiatry)1; Zhaohua Huo, MSc, PhD1; Vicky C Lin, MSSc, MPhil1; Ada WT Fung, MSc, PhD3; SL Ma, MPhil, PhD1; Calvin PW Cheng, MB, BS, FHKAM (Psychiatry)2; ST Cheng, PhD4; Frank HY Lai, MSc, PhD5; Benjamin HK Yip, BSc, PhD6; Samuel YS Wong, MD, FHKAM (Community Medicine)6
1 Department of Psychiatry, The Chinese University of Hong Kong, Hong Kong SAR, China
2 Department of Psychiatry, The University of Hong Kong, Hong Kong SAR, China
3 Department of Applied Social Sciences, Hong Kong Baptist University, Hong Kong SAR, China
4 Department of Health and Physical Education, The Education University
of Hong Kong, Hong Kong SAR, China
5 Department of Social Work, Education and Community Wellbeing, Faculty of Health and Life Sciences, Northumbria University, Newcastle, United Kingdom
6 The Jockey Club School of Public Health and Primary Care, The Chinese University of Hong Kong, Hong Kong SAR, China
Corresponding author: Prof Linda CW Lam (cwlam@cuhk.edu.hk)
Abstract
Introduction: Given the rapid population ageing,
the cognitive healthcare needs of older adults
warrant attention. This study reports on the findings
of the Hong Kong Mental Morbidity Survey for
Older People (HKMMSOP), which evaluated the
prevalence of neurocognitive disorders (NCD) and
associated health factors that may inform primary
care risk assessment and intervention.
Methods: The HKMMSOP recruited 4871
participants aged 60 years or above through
random sampling in Hong Kong during 2019 and
2023, including 4368 community-dwelling and 503
residential care home residents. Participants were
assessed for cognitive function and mental status
and completed health and lifestyle questionnaires.
The prevalence of NCD and associated factors were
evaluated with reference to the 2022 Hong Kong
population.
Results: The age- and gender-adjusted prevalences
of mild and major NCD were 21.8% and 9.7%,
respectively, among adults aged 60 years or above
in Hong Kong. Approximately 70% of residents
in long-term care (LTC) had major NCD. Chronic
diseases, sarcopenia risk, sensory impairments,
and specific lifestyle habits were associated with
cognitive function in logistic regression analyses
adjusted for demographic confounders (P<0.05).
Specialised medical services, including psychiatric
care, were used by approximately 40% of community
participants with major NCD.
Conclusion: A range of NCD is prevalent in both
community and LTC settings, with the highest
rates observed among the oldest-old. To improve
functional independence, community primary healthcare should prioritise early cardiovascular
disease management, physical health maintenance,
correction of sensory impairments, and promotion
of intellectual and social engagement. For effective
healthcare planning for frail older adults living in
LTC, the complex needs of nursing home residents
with NCD should be addressed.
New knowledge added by this study
- The study revealed that 9.7% of older adults had major neurocognitive disorders (NCD), and nearly one in five older adults had mild NCD in Hong Kong. The high prevalence of mild NCD warrants attention, particularly from the perspective of early management to reduce progression from functionally independent mild NCD to dependent states of major NCD.
- The lifestyle evaluation from the study highlighted that regular participation in physical and intellectual activities, being socially active, and maintaining good sleep quality were associated with better cognitive function.
- Specialised medical services utilisation among community-dwelling participants with major NCD was relatively low (<40%). Promoting awareness of early assessment may help reduce the risk of secondary complications and improve long-term health outcomes associated with NCD.
- Primary care platforms focused on early detection and management of chronic diseases should adopt a multidimensional approach—particularly addressing cardiovascular health, stroke prevention, sensorimotor function, physical activity, sleep hygiene, and leisure engagement—to achieve long-term cognitive benefits.
- Over 70% of residents in long-term care had major NCD. To improve quality of life and the caregiving environment, service provision and planning should be integrated to address the combined physical, cognitive, and mental health needs of these residents.
Introduction
Ageing, characterised by a progressive loss of
physiological integrity leading to impaired function
and increased vulnerability to death, is a major
health concern for global populations.1 The older
population (threshold defined by the World Health
Organization as aged ≥60 years) in Hong Kong, one
of the most rapidly ageing communities worldwide, is expected to increase from 2.28 million (30.5% of
the total population) in 2023 to 3.31 million (40.4%)
in 2046, with the steepest growth occurring in
advanced old age.2 Major neurocognitive disorders
(NCD) [also referred to as dementia] are the most
common neurodegenerative disorders associated
with ageing and exert a substantial impact on
healthcare systems. A global projection estimated
that the number of people living with dementia would
increase almost threefold, from 57.4 million cases in
2019 to 152.8 million in 2050.3 In the most recent
epidemiological study of dementia in Hong Kong
conducted in 2008, over 8.9% of community-dwelling
older adults had mild dementia, and 8.5% had mild
cognitive impairment (MCI), a synonym for mild
NCD.4 Given that these estimates are over a decade
old, demographic changes (eg, higher educational
attainment and evolving health conditions among
older adults) warrant re-evaluation of prevalences
and associated factors.5
Factors affecting cognitive decline are best
understood through a life-course perspective.
While genetic and early-life predisposing factors
are not readily modifiable, mid- and late-life health
and lifestyle factors are increasingly recognised as
modulators of cognitive impairment. A 2024 Lancet review6 suggested that 14 potentially modifiable
lifestyle and health factors, including cardiovascular
risk, hearing and vision loss, air pollution, and
mental health and lifestyle factors (eg, smoking,
alcohol consumption, physical inactivity, social
isolation, and depression), accounted for 45% of the
population-attributable risk of dementia. Growing
research interest has also focused on other potential
risk factors, such as sleep, diet, dental disease, and
frailty, which are also important determinants of
cognitive health.6
Investigating secular trends in dementia is
essential to understand the full spectrum of the condition in general populations and to identify risk
factors across populations and life stages.7 Recent
estimates from various populations have reported
lower-than-expected prevalence rates, possibly
due to improvements in education, environmental
enrichment, and healthcare, with resulting
reductions in cerebrovascular risk.6 8 In the local
context, an updated prevalence study facilitates
systematic evaluation of the evolving occurrence and
modulating factors of cognitive decline from physical
health to psychosocial perspectives.6 Such evaluation
is essential for developing context-, culture-, and
practice-tailored preventive strategies targeting
the identified risk factors, as well as for optimising
treatment and management.6 7 Particularly with
advancing age and rising rates of physical and mental
co-morbidities among older adults in Hong Kong,
the burden of care and service demands related to
cognitive impairment require more comprehensive
assessment and practical guidance.
This study aimed to estimate the current
prevalence of mild and major NCD in Hong Kong
and to identify their multidimensional associated
factors, based on the Hong Kong Mental Morbidity
Survey for Older People (HKMMSOP). We also
discuss how these findings may inform healthcare
interventions for early risk modification, reduction
of cognitive decline, and optimisation of care.
Methods
Study design and setting
The HKMMSOP was a commissioned study funded
by the Advisory Committee on Mental Health
through the Health and Medical Research Fund.
Conducted from January 2019 to January 2023, it
was designed as a territory-wide, population-based,
cross-sectional survey to examine the prevalence
and modulating factors of NCD among older adults
in Hong Kong. The study settings of HKMMSOP
included both community households and long-term
care (LTC) institutions. For clinical assessments,
HKMMSOP adopted a two-phase design. Phase
1 interviews comprised cognitive assessments for
mild and major NCD, evaluation of neuropsychiatric
syndromes and functioning, as well as physical health
and psychosocial measurements. Phase 2 involved
clinician interviews for diagnostic assessment and
subtyping of NCD.
Study size calculation
Sample size was determined based on previous
prevalence studies of dementia (2008)4 and common
mental disorders (2010-2013)9 in Hong Kong. For
an estimated NCD prevalence of approximately 2%
among adults aged 60 to 74 years, 3012 participants
were required to achieve a recommended
precision of 0.005. For an estimated prevalence of approximately 5% among those aged 75 years or
above, 1168 participants were required to achieve a
recommended precision of 0.0125. The HKMMSOP
ultimately recruited and completed assessments
for 3560 participants aged 60 to 74 years and 1311
participants aged 75 years or above.
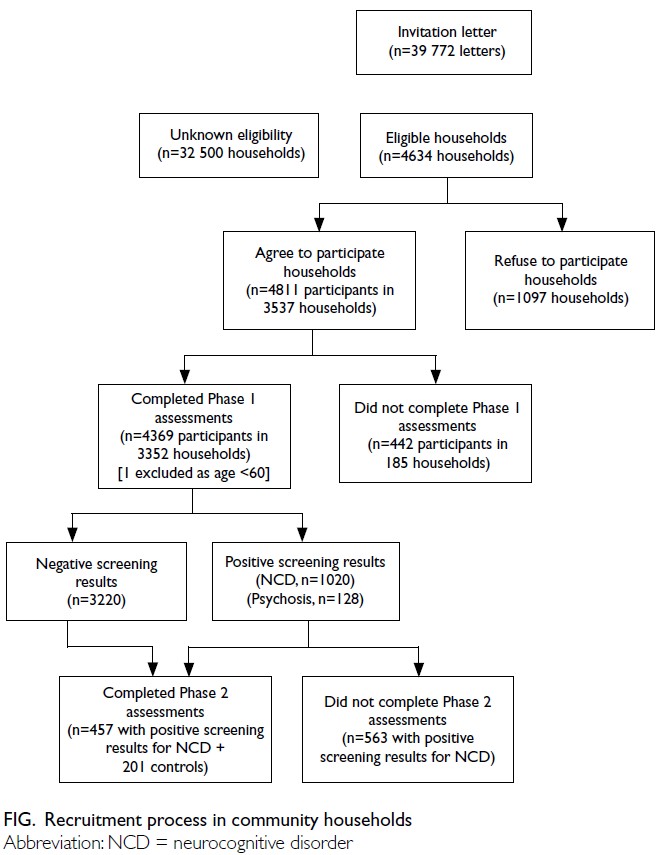
Sampling and subject recruitment
To recruit a representative sample of the older adult
population in Hong Kong, we adopted a multi-stage
random sampling method commonly used
in household surveys. A random list of addresses
(sampling frame) was generated by the Census
and Statistics Department of the Hong Kong SAR
Government, then stratified by geographical location
and residential type (private versus public housing).
For each address, an invitation letter introducing
the survey and a consent form for assessment were
enclosed. A telephone hotline, designated website,
and email contact were provided for enquiries and
to document refusals. Up to three invitation letters
were sent within 6 months to improve recruitment
success and reduce response bias; participants who
responded to the third contact had higher rates of
active employment compared with the rest of the
sample (P<0.05).
When households responded and included
residents aged 60 years or above, we invited them
for interviews; there could be one or more eligible
residents per household. If individuals agreed to
participate but were unable to provide complete
information (eg, due to profound sensory deficits),
data were obtained from their first-degree relatives
and categorised as proxy. Households with no
eligible participants (ie, all residents aged under
60 years) were invited to notify the research team
of their ineligibility through convenient contact
channels (email, text messages, or direct phone
calls). From January 2019 to January 2023, a total
of 39 772 invitation letters were sent to randomly
generated addresses. Of these, 3352 households
with 4369 eligible community-dwelling participants
consented to and completed the survey. Eligibility
and demographic characteristics were unknown
for the remaining 36 420 households. The flow of
participant sampling, recruitment, and assessment
is depicted in the Figure.
To obtain representative statistics for people
residing in LTC institutions, we adopted a two-stage
cluster sampling method. Superintendents of 600
residential care homes for the elderly (ie, LTCs),
randomly selected from the master list of registered
old age homes in Hong Kong, were first approached
for participation. When residential care homes
agreed to assist with recruitment, eligible residents
were randomly selected. Following institutional
consent and consent from participants and/or their
family members, assessments were completed for 503 residents from 51 registered old age homes
across Hong Kong. The prison population was not
included in this survey.
Data collection and measurements
Phase 1 study
Phase 1 assessments were conducted by trained
research assistants during visits to participants’
residential addresses or at the department’s research
centre. Due to social distancing and infection control
policies during the COVID-19 pandemic, phone
assessments were offered as an alternative and were
utilised by 28.7% of community participants. Proxy-based
assessments were conducted for participants
with profound physical or mental deficits (2.7%
of community participants and 34.2% of LTC
participants).
Socio-demographic information was
collected, including age, gender, birthplace, housing
type, education level, marital status, household
composition, current employment status, family
income, receipt of government financial subsidies, and religious affiliation.
Cognitive function was assessed using two
locally validated tools: the Chinese Abbreviated
MCI test10 and the Hong Kong version of Montreal
Cognitive Assessment (HK-MoCA).11 12 For
participants with moderate to severe major NCD
in both domiciliary and institutional settings, an
abridged version of the cognitive and mental state
assessment was used based on the HK-MoCA
5-minute protocol. All interviewers were trained
to administer the Clinical Dementia Rating (CDR)
to each participant, and satisfactory concordance
was achieved between interviewer-rated and
clinician-rated results (Spearman’s correlation:
0.668; P<0.001).13 Neuropsychiatric symptoms
were screened in all participants using the
Neuropsychiatric Inventory Questionnaire.14
Physical health was assessed using
questionnaires on chronic disease burden
(Cumulative Illness Rating Scale [CIRS]),15 along
with health screening measures including blood
pressure, body mass index, oral health, grip strength,
hearing and vision, postural balance, and sarcopenia
(SARC-F: Strength, Assistance with walking, Rising
from a chair, Climbing stairs, and Falls).16 Activities
of daily living were evaluated using the Chinese
version of the Disability Assessment for Dementia.17
Lifestyle factors were assessed using
questionnaires covering smoking and alcohol
consumption, physical and non-physical leisure
activities, sleep quality (Pittsburgh Sleep Quality
Index), use of drugs and vitamins, dietary intake of
fruits and vegetables, loneliness, and quality of life.18 19 20 21
Phase 2 study
Among the 1020 participants whose scores crossed
the threshold for mild or major NCD, clinician
interviews were conducted for NCD subtyping,
including assessment of cerebrovascular risk and
neuroimaging. Of these, 457 participants (response
rate: 44.8%) completed face-to-face structured
assessments at the department’s research centre,
where blood sampling facilities were available
(Figure). For consenting participants, fasting lipid
profiles, glycated haemoglobin, and apolipoprotein
E4 genotypes were analysed. Diagnoses and
subtypes of NCD were established according to
the DSM-5 (Diagnostic and Statistical Manual of
Mental Disorders, Fifth Edition)22 through clinician
assessment supplemented by laboratory results and
structural magnetic resonance imaging of the brain.
Additionally, 201 participants with negative
NCD screening results (CDR=0) were interviewed
by research team psychiatrists to evaluate the
sensitivity and specificity of Phase 1 assessments.
The sensitivity of the Phase 1 screening tools for
detecting mild or major NCD was 96.7%, while the
specificity for correctly identifying participants without NCD was 81%. The positive predictive value
was 90% and the negative predictive value was 93%.
Supplementary study for long-term care
institutions
Due to strict COVID-19 visitor policies in
place during the fieldwork period (2022-2023),
videoconference assessments were conducted
with participants in some LTC facilities (62.6%).
Individual health assessments were not performed
because of infection control policies in effect during
data collection in 2022.
Statistical analysis
Sample representativeness
In the two-phase prevalence study design, selection
bias could potentially arise if heterogeneity in
characteristics (eg, age, education level, family
structure, physical and mental morbidity) existed
between responders and non-responders. During
the study invitation and Phase 1 assessment,
demographic and clinical information for non-responders
and non-completers was unavailable.
Therefore, we compared Phase 2 completers (n=457)
and non-completers (n=563) among participants
with positive NCD screening results in Phase
1. Non-completers were more likely to be older
(>75 years: 49.9% vs 36.1%; P<0.001), have a higher
burden of chronic conditions (CIRS score: 4.8 ± 3.0
vs 4.4 ± 2.4; P=0.007), show greater levels of cognitive
impairment (CDR ≥1: 20.5% vs 12.2%; P<0.001),
exhibit more impaired daily functioning (score of
the Chinese version of the Disability Assessment for
Dementia: 90.6 ± 20.5 vs 96.0 ± 9.6; P<0.001), and
have a prior diagnosis of dementia before joining
the study (5.9% vs 1.6%; P=0.001). The potential
impact of participant imbalance in Phase 2 on study
findings is addressed in the Discussion section. No
imputation was performed for missing data.
Prevalences of mild and major neurocognitive disorders
Prevalence estimates of NCD were stratified by age-group,
gender and recruitment setting. Weighted
prevalence was calculated using sampling weights
that reflected the proportion of participants in
each stratum, with reference to the population
distribution by age and gender, as well as community
and LTC statistics (year-end population, 2022).23 24
Diagnoses included mild NCD and major NCD. For
each disorder, prevalence was expressed as the rate
per 100 persons, with corresponding 95% confidence
interval (95% CI). Sampling errors were estimated
using a bootstrap strategy implemented in Stata
software (StataCorp, College Station [TX], US). One
thousand samples were randomly drawn from the
original dataset through sampling with replacement. The standard error of weighted prevalence was
calculated as the square root of the sample variance
across these resamples.
Factors associated with neurocognitive disorders
The psychosocial, physical health, and lifestyle
correlates of NCD were evaluated. Crude and adjusted
associations (odds ratios) between the prevalence
of NCD and three categories of associated factors
were examined: (1) demographic characteristics,
including age, gender, years of education, marital
status (married, cohabiting, widowed, divorced,
separated or single), monthly household income
(<HK$6000, HK$6000-14 999, HK$15 000-29 999,
≥HK$30 000), and housing type (owned or rented);
(2) physical health status, including history of
hypertension, postural balance test (pass or fail),
corrected vision (normal or abnormal), corrected
hearing (normal or abnormal), oral health problems
(yes or no), sarcopenia (SARC-F score), and chronic
disease burden (CIRS score); and (3) lifestyle habits,
including smoking and alcohol consumption (never,
former, current), fruit intake (≥two portions/day
or not), vegetable intake (≥three portions/day or
not),18 25 sleep quality (score of the Pittsburgh Sleep
Quality Index21), regular participation (once or more
per week) in aerobic, resistance, and intellectual
activities,20 and social loneliness.19 Multivariable
logistic regression analyses were conducted to
identify independent associations between physical
health and lifestyle factors and NCD, adjusting for
confounders previously linked to NCD, including
age, gender, years of education, marital status,
and socio-economic status.5 6 8 All analyses were
performed using SPSS (Windows version 22.0; IBM
Corp, Armonk [NY], US), and differences were
considered statistically significant at P<0.05.
Results
Participant characteristics
Basic characteristics of participants living in the
community and in LTC settings are presented in
Table 1. The mean age was 69.6 ± 7.5 years (range,
60-105). Just over half of the respondents (53.6%)
were born in Hong Kong. A large proportion had
attained a secondary school education or above
(66.1%) and were married or co-habiting (67.5%).
Most older adults were economically inactive
(retired, homemaker, or never worked) [78.1%].
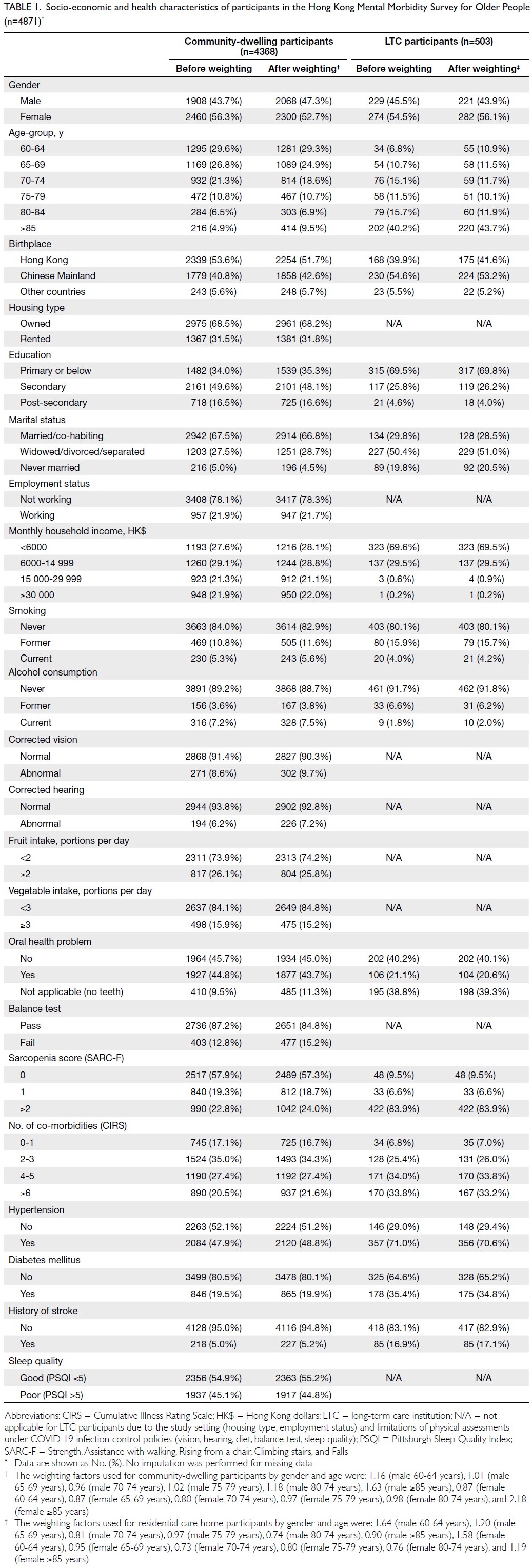
Table 1. Socio-economic and health characteristics of participants in the Hong Kong Mental Morbidity Survey for Older People (n=4871)
Of the 503 participants residing in LTC
facilities, 274 (54.5%) were women and the mean age
was 80.3 ± 11.0 years (range, 60-106). More than half
(54.6%) were born in Chinese Mainland; 30.4% had
attained secondary education or above; and 50.4%
were widowed, divorced or separated. Compared
with the LTC sample, the community sample
included a higher proportion of women (56.3% vs 54.5%) and younger-old adults (aged <75 years:
77.7% vs 32.6%), whereas the LTC sample included
more men (45.5% vs 43.7%) and adults aged ≥75
years (67.4% vs 22.2%). After sample weighting by
age and gender, both samples were comparable with
the overall older population in Hong Kong (Table 1).
Cognitive function and neuropsychiatric
symptoms
The distribution of CDR before sample weighting is
presented by 5-year age intervals and gender in both
community and LTC settings (Table 2). Older age
was associated with a higher prevalence and greater
severity of NCD, as measured by CDR (P<0.001).
Montreal Cognitive Assessment scores stratified
by gender and grouped into 5-year age intervals are
also presented for community-dwelling participants
(Table 3). Older age was associated with lower
HK-MoCA scores (P<0.001), while men had higher
HK-MoCA scores within 5-year age-groups >70
years (P<0.05).
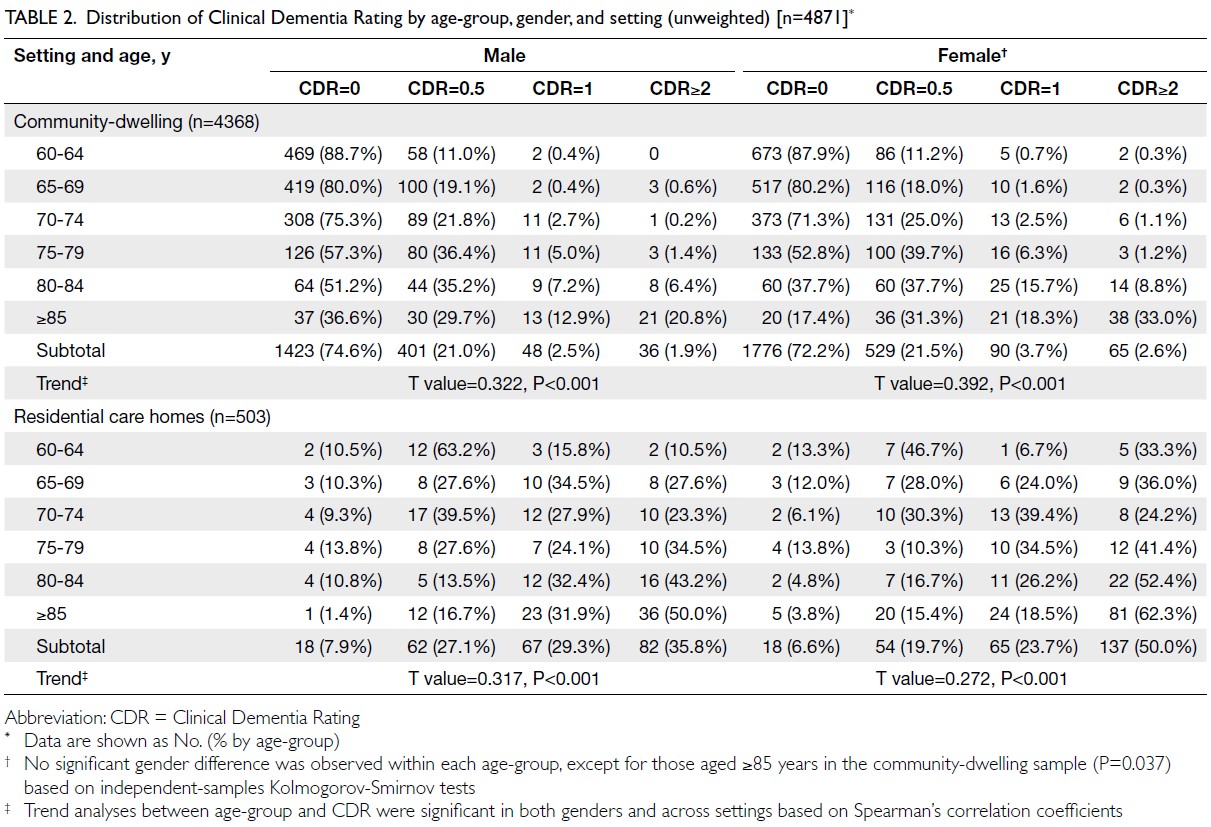
Table 2. Distribution of Clinical Dementia Rating by age-group, gender, and setting (unweighted) [n=4871]
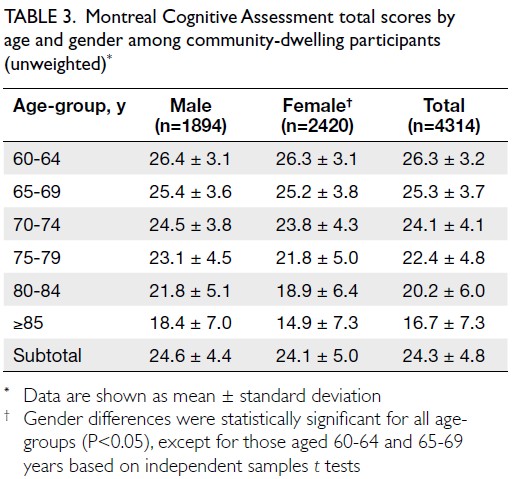
Table 3. Montreal Cognitive Assessment total scores by age and gender among community-dwelling participants (unweighted)
The proportion of neuropsychiatric symptoms
increased with worsening cognitive impairment. Among community-dwelling participants, the
prevalence of psychotic symptoms rose from 2.1% in
those with normal cognition to 6.3% in those with
mild NCD, and 21.3% in those with major NCD (P<0.001). Similarly, the prevalence of depression
and anxiety also increased with higher CDR scores
(P<0.001). Sleep disturbances were common across
all cognitive groups, affecting 32.2% of participants
with normal cognition, 43.5% with mild NCD, and
40.2% with major NCD.
Prevalence of neurocognitive disorders
The unweighted prevalence of mild and major NCD
is presented in online supplementary Table. As
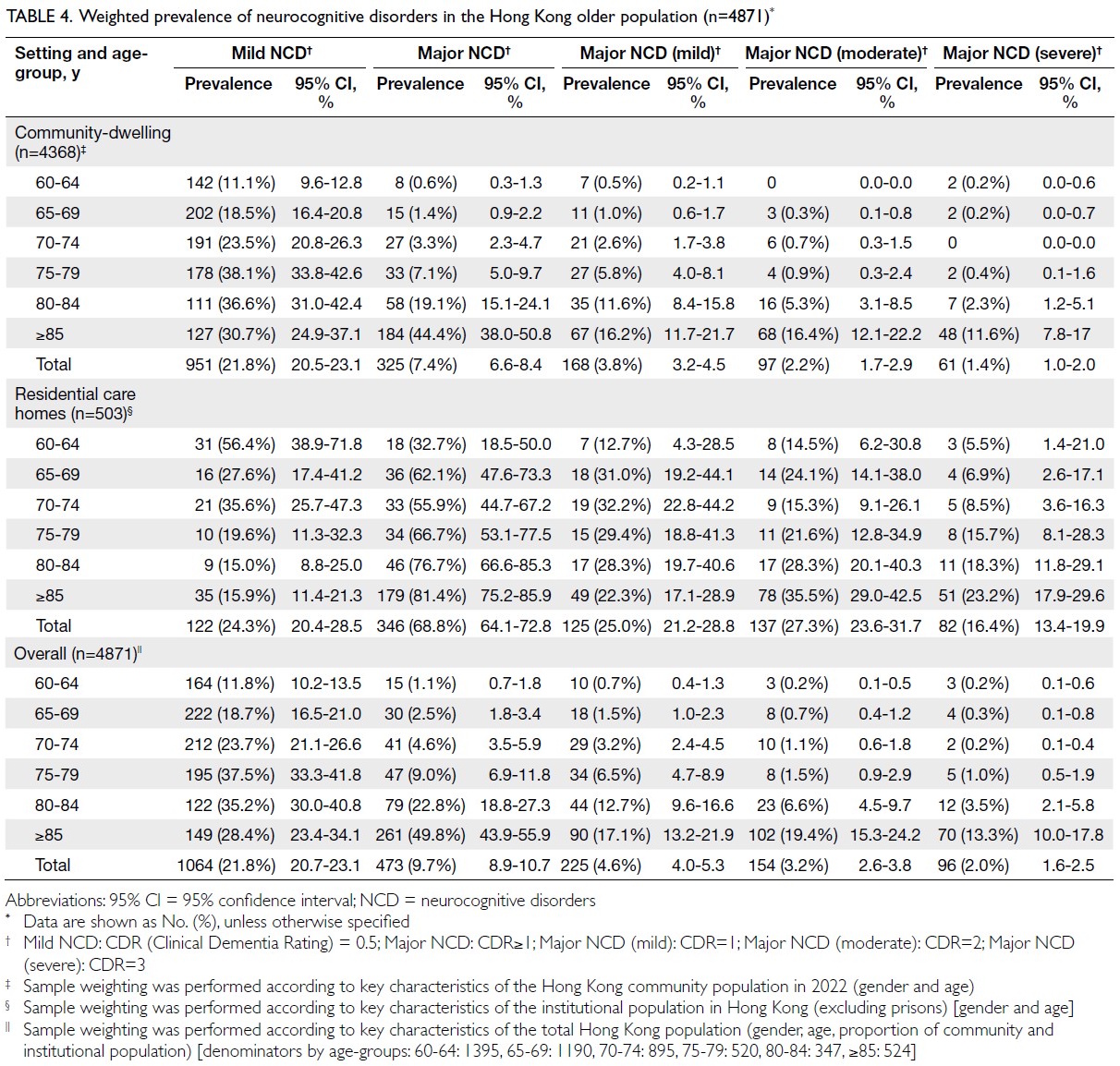
shown in Table 4, the weighted prevalence of mild
NCD was 21.8% among community-dwelling older
adults, 24.3% among those living in LTC facilities, and 21.8% overall in Hong Kong. For major NCD,
prevalence was 7.4% in the community, significantly
higher at 68.8% in LTC settings, and 9.7% overall.
Among Phase 2 community-dwelling
participants with mild or major NCD, 22.0% met the
DSM-5 criteria for Alzheimer’s disease, 23.7% had
mixed vascular NCD and AD, and 43.5% had vascular
NCD. Neurocognitive disorders due to Lewy body
disease and frontotemporal lobar degeneration
accounted for 1.2% and 0.5% of cases, respectively.
The apolipoprotein E4 genotype was identified in
17% of cognitively normal participants, 19% of those
with mild NCD, and 22% of those with major NCD. No significant differences in apolipoprotein E4
distribution were observed across cognitive function
groups.
Physical health and lifestyle correlates of neurocognitive disorders
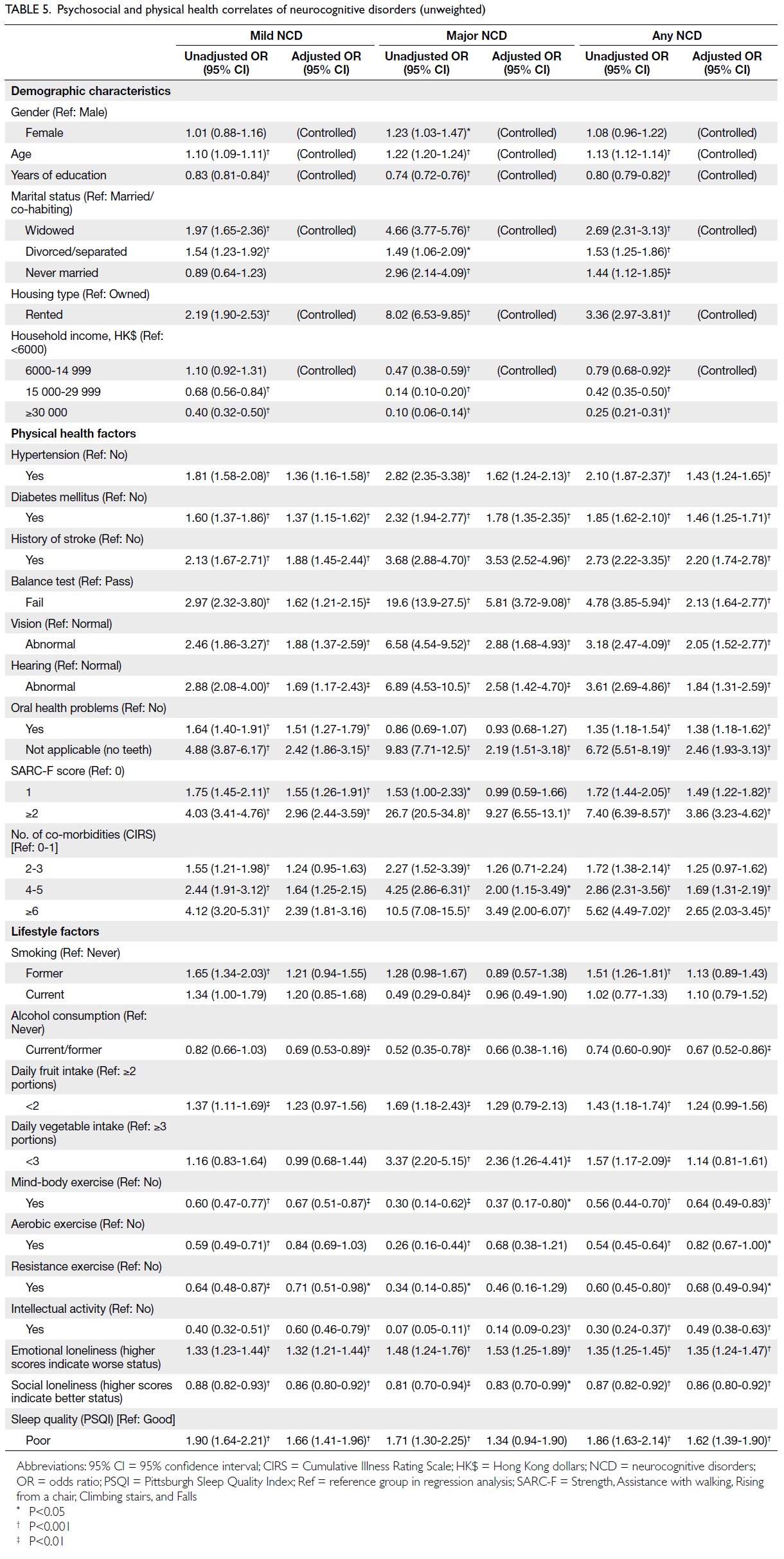
Mild neurocognitive disorders
In unadjusted analyses, older age, fewer years of
education, being widowed, divorced or separated,
living in rented housing, and lower household
income were significantly associated with a higher
prevalence of mild NCD. These factors were
subsequently controlled for in the logistic regression
analyses.
After controlling for the above demographic
confounders, hypertension, diabetes mellitus,
history of stroke, poor postural balance, higher
SARC-F scores, visual or hearing impairment, and
dental problems were associated with significantly
higher adjusted ORs for mild NCD (P<0.05). Physical
exercise (mind-body and resistance), engagement
in intellectual activities, and better subjective sleep
quality were associated with lower adjusted ORs for
mild NCD (P<0.05). In contrast, individuals with
mild NCD had significantly higher loneliness scores
(P<0.05) [Table 5].
Major neurocognitive disorders
Similar demographic risk factors, such as older
age, female gender, fewer years of education, being
widowed, divorced, separated or never married,
living in rented housing, and lower household income
level were associated with a higher prevalence of
major NCD. These factors were controlled for in the
logistic regression analyses (Table 5).
After controlling for demographic confounders,
hypertension, diabetes mellitus, history of stroke,
poor postural balance, abnormal vision or hearing,
edentulism, high SARC-F scores (≥2), and multiple
co-morbidities (≥4 chronic diseases) were associated
with higher adjusted ORs for major NCD (P<0.05)
[Table 5].
Less loneliness, participation in mind-body
physical exercise, engagement in intellectual
activities, and consumption of three portions of
vegetables or more per day were associated with
a lower likelihood of major NCD after adjustment
for demographic confounders (P<0.05). Poor sleep
quality was associated with a higher risk of major
NCD in unadjusted analyses (Table 5).
Family history
A family history of dementia was reported by 27%
of cognitively normal participants, 25% of those
with mild NCD, and 23% of those with major NCD.
Pearson Chi squared test showed no significant
differences across groups.
Service use
Self-reported service utilisation among community-dwelling
participants with NCD was assessed in 488
individuals who completed the Phase 2 assessment.
Participants with mild and major NCD reported
higher use of inpatient, accident and emergency, and
outpatient services compared with those with normal
cognition. Notably, participants with major NCD
reported significantly more psychiatric (15.7%) and
neurological (23.0%) consultations in the preceding
3 months than those with normal cognition (2.5%
and 2.7%, respectively) and those with mild NCD
(6.3% and 4.2%, respectively) [P<0.001].
Discussion
Main findings
Prevalence of neurocognitive disorders in Hong Kong compared with other Asian economies
As population ageing accelerates, the Asia-Pacific
region is projected to experience more than a
threefold increase in the number of people living
with dementia over the next three decades, rising
from 23 million in 2015 to 71 million by 2050.26 This
territory-wide, population-based study provides up-to-date prevalence estimates of NCD among adults
aged ≥60 years in Hong Kong in 2022. The prevalence
of mild NCD was 21.8% (21.8% in the community;
24.3% in LTC settings), while the prevalence of major
NCD was 9.7% (7.4% in the community; 68.8% in
LTC settings).26
A nationwide population-based study
conducted in Chinese Mainland in 2020 reported
overall age- and gender-adjusted prevalences of 15.5%
(95% CI=15.2-15.9) for MCI and 6.0% (95% CI=5.8-6.3) for dementia.5 The relatively higher prevalence
observed in Hong Kong may be related to differences
in population demographics and healthcare systems.
First, the present study included older adults residing
in residential care homes in both recruitment and
prevalence estimates. The higher proportion of
older adults living in LTC facilities in Hong Kong
(3.7% of the older population)27 compared with
Chinese Mainland (<1%),28 combined with the high
prevalence of major NCD among LTC residents
(68.8%), contributed to an increased overall
prevalence of major NCD in Hong Kong. Second,
even after excluding LTC residents, the prevalence
of NCD among community-dwelling older adults
in Hong Kong remained higher. This difference
may be attributed to longer life expectancy (83.7 vs
78.6 years) and an age structure characterised by a
greater proportion of the oldest-old (population
aged ≥80 years: 5.3% vs 2.3%) in Hong Kong29 30
compared with the Chinese Mainland. Nevertheless,
comparison of age-specific prevalence rates revealed
a lower prevalence of major NCD in younger-old
groups in Hong Kong29 30 (age 60-69 years: 1.0% vs 2.9%; age 70-79 years: 4.7% vs 8.4%5), but a higher
prevalence among the oldest-old (age ≥80 years:
33.7% vs 16.1%5). In contrast, the prevalence of mild
NCD was consistently higher across all age-groups
in Hong Kong (age 60-69 years: 14.5% vs 11.8%;
age 70-79 years: 28.8% vs 19.2%; age ≥80 years:
33.1% vs 25.0%).5 These patterns may reflect better
management of cardiovascular diseases and other
risk factors (eg, lower rates of smoking and alcohol
consumption) among the younger-old population,
the approximately twofold higher proportion of the
oldest-old among older adults (age ≥85 years: 10.7%
vs 4.9%5), and longer survival following dementia
onset in Hong Kong.
Compared with other developed Asian
economies, including Japan, South Korea, Taiwan,
and Singapore, the prevalence of dementia in Hong
Kong is also relatively high, particularly among
those aged 80 years or above (online supplementary Fig).31 Although the high proportion of oldest-old
individuals partly contributes to this observation
(Hong Kong: 10.7%, Japan: 15.2%, Taiwan: 7.7%,
Singapore: 6.6%, South Korea: 6.5%), another
important explanation may be the substantial burden
of cerebrovascular disease, which is associated with a
higher prevalence of dementia (online supplementary Fig).31 In Hong Kong, cerebrovascular risk factors
strongly contribute to dementia cases (eg, 43.5% vs
26.7% in Chinese Mainland5), whereas among the
oldest-old population, cerebral small vessel disease
is highly prevalent.
Trends in the prevalence of neurocognitive disorders in Hong Kong
In comparison with the 2008 community-based
prevalence study in Hong Kong,4 a decrease in the
overall prevalence of mild dementia (mild stage
of major NCD) was observed (from 5.4% in 2008 to 3.8% in 2022 [Table 4]), with reductions noted
across all age-groups. This decline may be attributed
to improved management of physical health risk
factors, enabling more older adults to remain within
the mild NCD range. Contributing factors may
include higher educational attainment, reduced
smoking and alcohol consumption, and better
control of cerebrovascular disease.
Regarding mild NCD (MCI or CDR=0.5),
the prevalences of very mild dementia (5.8%) and
MCI (23.8%) reported in 20084 are not directly
comparable to the prevalence of mild NCD
observed in HKMMSOP (21.8%). First, different
cognitive screening tools were employed. The 2008
study utilised the CMMSE and AMT,4 whereas
HKMMSOP adopted the HK-MoCA as the
primary screening instrument. The HK-MoCA was
designed to be more sensitive in detecting early
executive dysfunction associated with vascular or
non-Alzheimer’s pathology.12 In practice, the HK-MoCA12
demonstrates comparable sensitivity (97%)
but higher specificity (81% vs 72%) and negative
predictive value (93% vs 81%) compared with the
CMMSE.4 Second, the diagnostic threshold for mild
NCD, labelled as very mild dementia (CDR=0.5) in
the 2008 study,4 has shifted over the past decade in
favour of an early-detection paradigm. Advances in
screening accuracy and diagnostic algorithms may
have contributed to a higher detection rate of mild
NCD in HKMMSOP. Finally, due to methodological
limitations in the 2008 study,4 comparisons across the
full spectrum of cognitive impairment are restricted.
The CDR assessments were only conducted among
participants who screened positive for cognitive
impairment and proceeded to Phase 2 clinician
evaluation. In contrast, CDR scores in HKMMSOP
were determined for all participants during Phase
1 by trained research assistants and subsequently
corroborated by experienced psychiatrists.
Finally, optimisation of dementia risk
management, along with changes in the population
age structure, may also help explain the increased
concentration of moderate and severe cases among
the oldest-old participants in HKMMSOP.
Multidimensional associated factors
This study identified several risk factors associated
with NCD, many of which were common to both
mild and major NCD. These included increasing
age, lower educational attainment, being widowed,
divorced or separated, and poorer socio-economic
status as indicated by living in rented housing and
reporting a lower household income. These findings
are consistent with previous studies.5 8 32 Female
gender was associated with major NCD, but not with
mild NCD, among older adults in Hong Kong.
Regarding physical health conditions, in
addition to cardiovascular disease, poorer postural balance, higher sarcopenia scores, visual and
hearing impairment, and oral health problems were
associated with the presence of NCD. A greater
number of co-morbidities was associated with major
NCD, while poor sleep quality was associated with
an increased risk of mild NCD. With respect to
potentially modifiable lifestyle factors, consuming
three portions of vegetables or more per day
was associated with a lower prevalence of major
NCD. Regular physical exercise, engagement in
intellectual activities, and lower levels of loneliness
were associated with a reduced prevalence of NCD.
The cross-sectional associations observed between
potentially modifiable factors and NCD in this study
may enrich the existing evidence base and provide
converging directions for future research into causal
relationships. These findings may also inform policy
development aimed at the dementia prevention
worldwide.
Limitations
The findings of HKMMSOP should be interpreted
in light of several limitations. First, this was a cross-sectional
study; therefore, causal relationships
between NCD and associated factors cannot be
inferred. Second, sampling bias is an inherent
limitation in prevalence studies, as individuals
with an existing diagnosis and ongoing treatment
may be less likely to participate (due to reduced
activity levels resulting in underestimation) or
more motivated to enrol, potentially leading to
overestimation). Hard-to-reach populations may
also have been underrepresented due to factors
such as poor physical or cognitive function, limited
mobility, or the absence of family caregivers. The
COVID‑19 pandemic likely exacerbated sample bias
in this study: the household response rate was merely
8.4%, and the sample over‑represented women and
younger‑old adults, potentially underestimating
NCD severity among the oldest‑old.
Third, participants who did not attend Phase
2 assessments were older and had a greater burden
of physical morbidity. The main reasons for non-participation
among individuals with positive
screening results in Phase 1 included ‘assessment
centre too far from home’, ‘too old or too frail’,
‘no accompanying caregiver’, and ‘no perceived
necessity’. While Phase 1 assessments demonstrated
satisfactory positive and negative predictive values
for NCD diagnosis (90% and 93%, respectively),
differences in participation profiles between those
who did and did not complete Phase 2 may have
influenced prevalence estimates.
Finally, HKMMSOP was conducted during
periods when Hong Kong was affected by various
phases of the COVID-19 pandemic. Infection
control measures adversely impacted participant
recruitment (low household response rate: 8.4%) and interview arrangements. Surveys involving
residents of care homes and hostels were particularly
restricted due to stringent lockdown policies. As a
result, only a limited number of in-person, telephone,
or online assessments could be conducted with
older participants. Most information was obtained
from family caregivers or formal carers within the
respective institutions, which may have influenced
the reliability and validity of the assessment
instruments.
Implications
Despite these limitations, the findings of this
study remain valuable for informing future clinical
practice, public health interventions, and research
priorities. First, the HKMMSOP revealed that nearly
one in five older adults in Hong Kong had mild NCD.
This pattern is likely not unique to Hong Kong and
may also apply to other Asian metropolitan cities
characterised by increasing life expectancy and
a high burden of physical co-morbidities among
older populations. Mild NCD represents an at-risk
state with variable clinical trajectories. In a 5-year
prospective study of Chinese older adults with MCI,
approximately 30% progressed to dementia, while
others either remained stable or improved to normal
cognitive function.33 Given the spectrum from normal
cognition to mild and major NCD encountered in
primary care, prevention and timely intervention
should address a broad range of associated health
factors, particularly common and modifiable ones
operating across the life course of cognitive health,
such as educational attainment, socio-economic
status, sensorimotor function, physical exercise, and
intellectual activity.7 34 Although the cross-sectional
design of the HKMMSOP does not permit causal
inference, early intervention and management
of these modifiable health risks may help reduce
progression to major NCD, improve quality of
life and functional capacity among individuals
living with MCI, and yield meaningful clinical and
economic benefits.
Within Hong Kong, the development of
District Health Centres provides an opportunity
to support primary care providers in planning
screening and early intervention programmes for
cognitive and mental health. Optimal management
of cardiovascular disease and related risk factors
from midlife is essential as cerebral small vessel
disease contributes to—and may play a causal
role in—a substantial proportion of vascular and
mixed dementia cases.35 Additionally, sensory
function, oral health, and musculoskeletal integrity
should be emphasised and integrated into primary
healthcare screening. Lifestyle interventions also
warrant attention, as clinicians may promote
cognitive benefits through various forms of
physical exercise and intellectual activities, as well as interventions targeting sleep hygiene and social
connectedness.25 36 37 38
Second, considering the high prevalence
of major NCD (dementia) among the oldest-old
population, an integrated medico-social support
system should be established. Among participants
with major NCD in the HKMMSOP, utilisation
rates of specialist services (psychiatric, neurological,
and psychological outpatient care) were far from
optimal (<40%). This gap may substantially hinder
the timely treatment and management of cognitive
or behavioural complications, thereby increasing
family, economic, and societal burdens.39 At
present, primary care consultations specifically
addressing cognitive decline are not well established
in Hong Kong. While not all individuals with NCD
require specialist medical attention, the observed
60% service gap underscores the importance of
strengthening primary care management to optimise
cognitive function in the community.39 Given that
NCD comprises heterogeneous neurodegenerative
conditions, appropriate and tiered medical
assessments and interventions across both primary
and specialist settings play a critical role in accurate
subtype diagnosis, personalised manageme
Third, approximately 70% of residents in LTC
facilities were affected by major NCD. Coordinated
efforts and the integration of multidisciplinary care
are essential to recognise and address the complex
cognitive, physical, and mental health needs of
individuals living in LTC facilities, as well as those of
their caregivers.
Finally, considering the limitations of this
cross-sectional prevalence study, further research is
warranted in the following two areas: (1) investigation
of potentially modifiable health and lifestyle factors
for healthy cognitive ageing through longitudinal,
cohort, and clinical trial designs to elucidate causal
relationships; and (2) focused evaluation of cognitive
impairment among the oldest-old population,
individuals with complex socio-medical conditions
(eg, hard-to-reach groups, those living alone, those
with high comorbidity burden, or limited access to
health and social resources), and residents of LTC
facilities.
Conclusion
The HKMMSOP study provides updated estimates
of NCD prevalence among community-dwelling
adults aged 60 years or above in Hong Kong
during the COVID-19 pandemic period. The
weighted prevalence of major NCD in Hong Kong
was estimated at 9.7%, with the greatest increase
observed among the oldest-old. Given the steep
rise in the proportion of the population reaching
advanced age, the total number of people living
with major NCD is expected to continue increasing over the coming decade. Approximately one in five
adults aged 60 years or above had mild NCD. Public
health education for older adults should focus on
optimising the management of chronic medical and
cerebrovascular diseases, promoting regular physical
exercise, correcting sensory impairments, and
encouraging active engagement in intellectual and
social enrichment activities. Equally important, the
complex needs of older adults residing in residential
care homes should not be overlooked—nearly seven
in ten LTC residents were affected by major NCD.
Author contributions
Concept or design: LCW Lam, WC Chan, ATC Lee, AWT Fung, SL Ma, CPW Cheng, ST Cheng, FHY Lai, BHK Yip, SYS Wong.
Acquisition of data: LCW Lam, WC Chan, ATC Lee, Z Huo, VC Lin.
Analysis or interpretation of data: LCW Lam, WC Chan, ATC Lee, Z Huo, VC Lin.
Drafting of the manuscript: All authors.
Critical revision of the manuscript for important intellectual content: All authors.
Acquisition of data: LCW Lam, WC Chan, ATC Lee, Z Huo, VC Lin.
Analysis or interpretation of data: LCW Lam, WC Chan, ATC Lee, Z Huo, VC Lin.
Drafting of the manuscript: All authors.
Critical revision of the manuscript for important intellectual content: All authors.
All authors had full access to the data, contributed to the study, approved the final version for publication, and take responsibility for its accuracy and integrity.
Conflicts of interest
All authors have disclosed no conflicts of interest.
Acknowledgement
The authors thank the participants, their family members,
and staff of the participating long-term care homes for the
generous support they offered. Special thanks are extended to
the Hospital Authority psychogeriatric teams for encouraging
selected old age homes to participate.
Declaration
Part of this study was presented as oral presentation at
the 2024 International Congress on Neuropsychiatry in
Melbourne, Australia, 27-29 October 2024.
Funding/support
This commissioned study was funded by the Health and
Medical Research Fund of the Hong Kong SAR Government
(Ref No.: MHS-P1 Part 3). The funder had no role in study
design, data collection, analysis, interpretation, or manuscript
preparation.
Ethics approval
This research was approved by the Survey and Behavioural
Research Ethics Committee (Ref No.: SBRE 18-628) and
the Clinical Research Ethics Committee of The Chinese
University of Hong Kong, Hong Kong (Ref No.: CREC NTEC
CUHK 2018-0529). Written consent was obtained from
each participant or their first-degree relative (for those with
profound cognitive impairments or sensory deficits) before
joining the study.
References
1. López-Otín C, Blasco MA, Partridge L, Serrano M, Kroemer G. The hallmarks of aging. Cell 2013;153:1194–217. Crossref
2. Census and Statistics Department, Hong Kong SAR Government. Hong Kong Population Projections 2022-2046. Aug 2023. Available from: https://www.censtatd.gov.hk/en/data/stat_report/product/B1120015/att/B1120015092023XXXXB01.pdf. Accessed 17 Mar 2025.
3. GBD 2019 Dementia Forecasting Collaborators. Estimation of the global prevalence of dementia in 2019 and forecasted prevalence in 2050: an analysis for the Global Burden of Disease Study 2019. Lancet Public Health 2022;7:e105-25. Crossref
4. Lam LC, Tam CW, Lui VW, et al. Prevalence of very mild and mild dementia in community-dwelling older Chinese people in Hong Kong. Int Psychogeriatr 2008;20:135-48. Crossref
5. Jia L, Du Y, Chu L, et al. Prevalence, risk factors, and management of dementia and mild cognitive impairment in adults aged 60 years or older in China: a cross-sectional study. Lancet Public Health 2020;5:e661-71. Crossref
6. Livingston G, Huntley J, Liu KY, et al. Dementia prevention, intervention, and care: 2024 report of the Lancet standing Commission. Lancet 2024;404:572-628. Crossref
7. Brayne C, Davis D. Making Alzheimer’s and dementia research fit for populations. Lancet 2012;380:1441-3. Crossref
8. Livingston G, Huntley J, Sommerlad A, et al. Dementia prevention, intervention, and care: 2020 report of the Lancet Commission. Lancet 2020;396:413-46. Crossref
9. Fung AW, Chan WC, Wong CS, et al. Prevalence of anxiety disorders in community dwelling older adults in Hong Kong. Int Psychogeriatr 2017;29:259-67. Crossref
10. Lam LC, Tam CW, Lui VW, et al. Screening of mild cognitive impairment in Chinese older adults—a multistage validation of the Chinese abbreviated mild cognitive impairment test. Neuroepidemiology 2008;30:6-12. Crossref
11. Wong A, Law LS, Liu W, et al. Montreal Cognitive Assessment: one cutoff never fits all. Stroke 2015;46:3547-50. Crossref
12. Yeung PY, Wong LL, Chan CC, Leung JL, Yung CY. A validation study of the Hong Kong version of Montreal Cognitive Assessment (HK-MoCA) in Chinese older adults in Hong Kong. Hong Kong Med J 2014;20:504-10. Crossref
13. Morris JC, Ernesto C, Schafer K, et al. Clinical Dementia Rating training and reliability in multicenter studies: the Alzheimer’s Disease Cooperative Study experience. Neurology 1997;48:1508-10. Crossref
14. Kaufer DI, Cummings JL, Ketchel P, et al. Validation of the NPI-Q, a brief clinical form of the Neuropsychiatric Inventory. J Neuropsychiatry Clin Neurosci 2000;12:233-9. Crossref
15. Conwell Y, Forbes NT, Cox C, Caine ED. Validation of a measure of physical illness burden at autopsy: the Cumulative Illness Rating Scale. J Am Geriatr Soc 1993;41:38-41. Crossref
16. Malmstrom TK, Morley JE. SARC-F: a simple questionnaire to rapidly diagnose sarcopenia. J Am Med Dir Assoc 2013;14:531-2. Crossref
17. Mok CC, Siu AM, Chan WC, Yeung KM, Pan PC, Li SW. Functional disabilities profile of Chinese elderly people with Alzheimer’s disease—a validation study on the Chinese version of the Disability Assessment for Dementia. Dement Geriatr Cogn Disord 2005;20:112-9. Crossref
18. Lee AT, Richards M, Chan WC, Chiu HF, Lee RS, Lam LC. Lower risk of incident dementia among Chinese older adults having three servings of vegetables and two servings of fruits a day. Age Ageing 2017;46:773-9. Crossref
19. Leung GT, de Jong Gierveld J, Lam LC. Validation of the Chinese translation of the 6-item De Jong Gierveld Loneliness Scale in elderly Chinese. Int Psychogeriatr 2008;20:1262-72. Crossref
20. Leung GT, Fung AW, Tam CW, et al. Examining the association between late-life leisure activity participation and global cognitive decline in community-dwelling elderly Chinese in Hong Kong. Int J Geriatr Psychiatry 2011;26:39-47. Crossref
21. Tsai PS, Wang SY, Wang MY, et al. Psychometric evaluation of the Chinese version of the Pittsburgh Sleep Quality Index (CPSQI) in primary insomnia and control subjects. Qual Life Res 2005;14:1943-52. Crossref
22. American Psychiatric Association, DSM-5 Task Force. Diagnostic and Statistical Manual of Mental Disorders: DSM-5. 5th Edition. American Psychiatric Publishing; 2013. Crossref
23. Census and Statistics Department, Hong Kong SAR Government. Population by sex and age group 2022. Available from: https://www.censtatd.gov.hk/en/web_table.html?id=1A. Accessed 10 Jul 2025.
24. Census and Statistics Department, Hong Kong SAR Government. Thematic Report: Older Persons. Feb 2023. Available from: https://www.censtatd.gov.hk/en/data/stat_report/product/B1120118/att/B11201182021XXXXB0100.pdf. Accessed 23 Dec 2024.
25. Lee AT, Richards M, Chan WC, Chiu HF, Lee RS, Lam LC. Intensity and types of physical exercise in relation to dementia risk reduction in community-living older adults. J Am Med Dir Assoc 2015;16:899.e1-7. Crossref
26. Alzheimer’s Disease International. Dementia in the Asia Pacific Region. Nov 2014. Available from: https://www.alzint.org/resource/dementia-in-the-asia-pacific-region/. Accessed 18 Mar 2026.
27. Census and Statistics Department, Hong Kong SAR Government. Hong Kong 2021 Population Census—Thematic Report. Feb 2023. Available from: https://www.censtatd.gov.hk/en/data/stat_report/product/B1120118/att/B11201182021XXXXB0100.pdf. Accessed 17 Mar 2026.
28. Ministry of Civil Affairs, National Working Committee on Aging. 2024 National Bulletin on the Development of Aging Affairs. Jul 2025. Available from: https://www.mca.gov.cn/n152/n165/c1662004999980006089/part/21508.pdf. Accessed 17 Mar 2026.
29. Statista. Share of population aged 60 and older in China from 1950 to 2020 with forecasts until 2100. Available from: https://www.statista.com/statistics/251529/share-of-persons-aged-60-and-older-in-the-chinese-population/. Accessed 17 Dec 2024.
30. Census and Statistics Department, Hong Kong SAR Government. Hong Kong Population Census—Summary Findings. Available from: https://www.censtatd.gov.hk/en/data/stat_report/product/B1120106/att/B11201062021XXXXB01.pdf. Accessed 17 Dec 2024.
31. Institute for Health Metrics and Evaluation, University of Washington. Global Burden of Disease Results 2021. Available from: https://vizhub.healthdata.org/gbd-results/. Accessed 2 Mar 2025.
32. Kalaria RN, Maestre GE, Arizaga R, et al. Alzheimer’s disease and vascular dementia in developing countries: prevalence, management, and risk factors. Lancet Neurol 2008;7:812-26. Crossref
33. Wong CH, Leung GT, Fung AW, Chan WC, Lam LC. Cognitive predictors for five-year conversion to dementia in community-dwelling Chinese older adults. Int Psychogeriatr 2013;25:1125-34. Crossref
34. Flodgren GM, Berg RC. Primary and secondary prevention interventions for cognitive decline and dementia [internet]. Available from: https://pubmed.ncbi.nlm.nih.gov/29553642/. Accessed 18 Mar 2026.
35. Lam BY, Cai Y, Akinyemi R, et al. The global burden of cerebral small vessel disease in low- and middle-income countries: a systematic review and meta-analysis. Int J Stroke 2023;18:15-27. Crossref
36. Lam LC, Tam CW, Lui VW, et al. Modality of physical exercise and cognitive function in Hong Kong older Chinese community. Int J Geriatr Psychiatry 2009;24:48-53. Crossref
37. Lam LC, Chau RC, Wong BM, et al. A 1-year randomized controlled trial comparing mind body exercise (Tai Chi) with stretching and toning exercise on cognitive function in older Chinese adults at risk of cognitive decline. J Am Med Dir Assoc 2012;13:568.e15-20. Crossref
38. Lee AT, Luo Y, Huo Z, Shi L, Chu WC, Lam LC. Effect of increasing cognitive activity participation on default mode network in older adults with subjective cognitive decline: a randomised controlled trial. EBioMedicine 2024;102:105082. Crossref
39. Huo Z, Yip BH, Lee AT, et al. Healthcare utilization and economic costs of neurocognitive disorders in community-dwelling older Chinese adults: A comparison with 9 Asian economies. J Alzheimers Dis 2025;107:515-28. Crossref