Hong Kong Med J 2026;32:Epub 17 Apr 2026
© Hong Kong Academy of Medicine. CC BY-NC-ND 4.0
ORIGINAL ARTICLE
Has the bacteriology of periprosthetic joint infection after total knee arthroplasty changed over time? A retrospective cohort study of 2171 patients
JR Khoo, MB, BS1; PK Chan, FHKCOS, FHKAM (Orthopaedic Surgery)2; Jeffrey HY Leung, BSc, MSc2; Vincent WK Chan, FHKAM (Orthopaedic Surgery), FRCSEd1; Amy Cheung, FHKCOS, FHKAM (Orthopaedic Surgery)1; Michelle Hilda Luk, FHKAM (Orthopaedic Surgery), FRCSEd1; MH Cheung, FHKCOS, FHKAM (Orthopaedic Surgery)2; Henry Fu, FHKCOS, FHKAM (Orthopaedic Surgery)2; KY Chiu, FHKCOS, FHKAM (Orthopaedic Surgery)2
1 Department of Orthopaedics and Traumatology, Queen Mary Hospital and The University of Hong Kong, Hong Kong SAR, China
2 Department of Orthopaedics and Traumatology, School of Clinical Medicine, Li Ka Shing Faculty of Medicine, The University of Hong Kong, Hong Kong SAR, China
Corresponding author: Dr PK Chan (cpk464@hku.hk)
Abstract
Introduction: Periprosthetic joint infection (PJI) is
an uncommon but serious complication of total knee
arthroplasty (TKA). A previous retrospective cohort
study at our institution reported a PJI incidence of
1.34% between 1993 and 2013. The present study
aimed to determine whether the incidence of PJI
after TKA has changed at our hospital and to evaluate
changes in microbiological patterns between 2014
and 2021.
Methods: In total, 2171 primary TKAs were
performed at Queen Mary Hospital between 1
January 2014 and 31 December 2021. All cases
of PJI were identified using the Musculoskeletal
Infection Society criteria. Patient demographics, PJI
occurrence, and microbiological data were collected
and compared with the previously published findings
from the 1993-2013 PJI cohort.
Results: The incidence of PJI after TKA was 0.64%
between 2014 and 2021, representing a significant
decrease from the incidence of 1.34% observed at
our institution between 1993 and 2013 (P=0.018).
There was no significant difference in the incidence
of early-onset infection (P=0.095). Methicillin-sensitive
Staphylococcus aureus was the most
common causative organism, accounting for 57.1% (n=8) of our cohort and 26.5% (n=9) in the previous cohort.
Conclusion: The incidence of PJI decreased
significantly from 1.34% to 0.64% between the
two study periods, suggesting the effectiveness of
infection-reduction measures implemented at our
institution. Minimal differences were observed in the
microbiological patterns of PJI between the cohorts.
New knowledge added by this study
- Between 2014 and 2021, the incidence of periprosthetic joint infection (PJI) after elective primary total knee arthroplasty (TKA) performed at our institution was 0.64%.
- Methicillin-sensitive Staphylococcus aureus remains the most common causative organism in cases of PJI.
- Antibiotic-resistant microorganisms are less prevalent than expected in cases of PJI.
- Multidisciplinary, protocol-driven optimisation of modifiable risk factors—both preoperatively and perioperatively—directly lowers PJI rates. Hospitals should adopt restrictive transfusion policies and aggressive medical co-morbidity management as standard of care for all patients undergoing TKA.
- Initiating antibiotics only after obtaining appropriate microbiological samples (eg, joint aspiration) significantly improves organism identification. This allows targeted antimicrobial therapy rather than empirical coverage, which is particularly important given the changing bacteriological profile. Clinicians should avoid prescribing empirical antibiotics before sampling to prevent false-negative cultures and subsequent treatment failure.
Introduction
Periprosthetic joint infection (PJI) is an uncommon
but severe complication of total knee arthroplasty
(TKA). The existing literature indicates that approximately 1% to 2% of patients undergoing
primary arthroplasty experience PJI; moreover,
PJI is the leading cause of revision arthroplasty.1 2
Individuals with PJI may experience a substantial decrease in quality of life and must undergo complex
and costly treatments to resolve this complication.3
A previous study at our institution, Queen Mary
Hospital in Hong Kong, examined 2543 patients who
underwent elective primary TKA between 1993 and
2013.4 During that period, the reported incidence
of PJI was 1.34% and the most common causative
organism was methicillin-sensitive Staphylococcus
aureus (MSSA).4 The number of TKAs performed at
our centre is rapidly increasing. In the past 8 years,
clinicians at our institution have performed 85% of
the total number of TKAs between 1993 and 2013,
a span of 20 years. Considering the rapid population
ageing in Hong Kong, we anticipate a continued
increase in the number of TKAs. In recent years,
various measures have been proposed to further
reduce the incidence of PJI, including restrictions on
blood transfusion rates, preoperative optimisation
of modifiable risk factors, and the implementation
of stringent culture techniques to improve microbial
yield.5 6 7 However, the limited availability of local data
makes it difficult to assess the effectiveness of these
techniques in reducing PJI incidence. It is important
to analyse the efficacy of these interventions as part of
ongoing efforts to improve surgical outcomes at our
centre. Furthermore, the increasing consumption
of antibiotics over the past two decades has led to
inevitable changes in the microbiological landscape
of infectious organisms.8
This study had three objectives. First, it aimed
to provide current local data on the incidence of PJI
after elective primary TKA. Second, it sought to
identify changes in the microbiological landscape of
PJI; this information may guide future treatment and
prevention strategies. Third, it aimed to showcase the
efficacy of interventions to reduce PJI incidence and
encourage their adoption beyond our institution.
Considering the measures introduced to reduce
infection at our institution, we hypothesised that
the incidence of PJI after primary TKA decreased
over the past decade. We also hypothesised that
the proportion of methicillin-resistant S aureus
(MRSA)–related PJI increased during this period due
to the increasing global consumption of antibiotics.
Methods
This retrospective cohort study compared the
incidence and bacteriology of PJI after TKA at
our institution. Participants were included if they
underwent primary elective TKA at our institution
between 2014 and 2021, and met the 2011
Musculoskeletal Infection Society (MSIS) criteria for
PJI.9 Exclusion criteria were infection after revision
arthroplasty, knee arthroplasty for malignant joint
conditions, and active bacteraemia. The primary
outcomes of interest were the incidence and
bacteriological patterns of PJI. Secondary outcomes included preoperative patient demographics and time to onset of PJI.
Study population
The Hong Kong Hospital Authority’s Clinical Data
Analysis and Reporting System and the Local Joint
Replacement Registry were utilised to identify all
TKAs performed at our institution between 2014
and 2021. Records were then searched using the
keywords ‘orthopaedic aftercare’ and ‘periprosthetic
joint infection’ to identify potential cases of PJI.
Patients who did not meet the 2011 MSIS criteria for
PJI were excluded; the remaining patients comprised
the study cohort.10 Two senior authors of this study
(PK Chan and KY Chiu) independently screened
the patient database using the 2011 MSIS criteria to
identify suitable patients for further data collection.
Any uncertainties or disagreements were resolved
through discussion.
Using a predefined data extraction form, the
same two senior authors extracted the following
data from the records of all included patients:
intraoperative joint fluid culture results, age, sex,
medical co-morbidities (eg, diabetes mellitus,
rheumatoid arthritis, and immunosuppression), date
of the index operation, surgical technique, operative
time, date of re-operation, and postoperative antibiotic regimen.
A previous retrospective cohort study by Siu
et al4 assessed the incidence and bacteriology of
PJI among patients who underwent TKA at our
institution between 1993 and 2013. From that study,
we extracted data on the incidence and bacteriology
of PJI, patient demographics, and time to onset
of infection for comparison with our cohort. For
both cohorts, we recorded the mean operative
time, number of joint specialists involved, surgical
technique, use of patient-specific instrumentation,
and infection control protocols; these data were
used to identify potential confounders that could
influence the incidence of PJI.
Patients in our cohort were classified according
to the time to infection onset as early, delayed,
or late. Early-onset PJI was defined as infection
occurring within 3 months of the index operation.
These infections commonly arise from intraoperative
contamination by highly virulent microorganisms
and therefore constitute a key focus of intervention.
Delayed-onset PJI was defined as infection occurring
between 3 and 24 months after the index operation.
These infections are also typically acquired during
surgery but involve less virulent microorganisms.
Late-onset PJI was defined as infection occurring
over 24 months after surgery. These infections are
often caused by haematogenous pathogens unrelated
to the index operation.11
In accordance with our institution’s guidelines,
patients were invited to attend follow-up at 2 weeks, 3 months, 6 months, and 12 months postoperatively.
Patients without complications were subsequently
scheduled for annual follow-up. Regarding infection
control, the preoperative, perioperative, and
postoperative protocols for elective primary TKA
remained consistent throughout the study period
in both cohorts. Intravenous antibiotic prophylaxis
(1 g of cefazolin, or vancomycin for patients with
a penicillin allergy) was administered 1 hour prior
to skin incision. Intraoperatively, laminar airflow
and body exhaust systems were utilised. Antibiotic-loaded
cement was not routinely used, and a single
postoperative wound management and rehabilitation
programme was implemented throughout the study
period. Postoperative antibiotics were not routinely
administered.
Statistical analyses
Categorical variables were grouped for analysis;
prevalence was calculated and group differences
were tested with the Chi squared test. Continuous
variables were compared using independent two-tailed
t tests. A P value <0.05 was considered
statistically significant. All statistical analyses were
conducted using SPSS (Windows version 27.0; IBM
Corp, Armonk [NY], United States).
Results
In total, 2543 and 2171 primary TKAs were
performed at our institution between 1993-20134
and 2014-2021, respectively. The incidence of PJI was
0.64% (n=14; 95% confidence interval=0.39-0.89)
between 2014 and 2021, significantly lower than the
1.34% (n=34; 95% confidence interval=0.97-1.71)
recorded between 1993 and 2013 (P=0.018).4
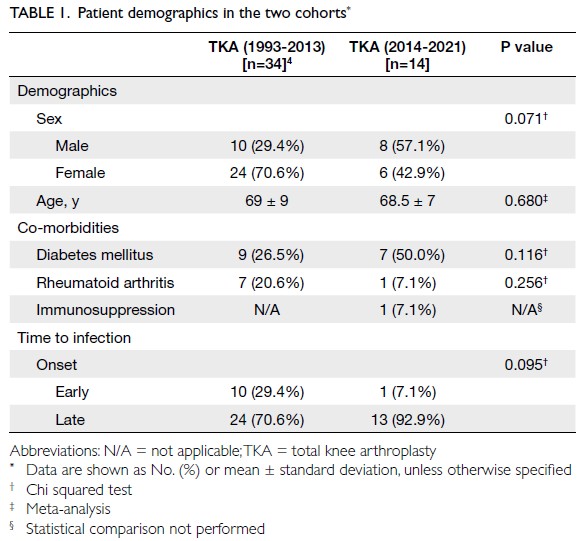
The mean age of the 14 patients with PJI in
our cohort was 68.5 ± 7 years (range, 56-85). Of
these patients, eight were men (57.1%) and six were
women (42.9%). In terms of medical co-morbidities,
seven patients had diabetes mellitus (50.0%), one
had rheumatoid arthritis (7.1%), and one had end-stage
renal disease requiring immunosuppression
(7.1%). The mean follow-up period in our cohort
was 4 years 9 months (interquartile range [IQR], 4
years 0 months to 6 years 11 months). There were
no significant differences in age, sex distribution,
or medical co-morbidities (diabetes mellitus and
rheumatoid arthritis) between the two cohorts. The
cohort demographics are compared in Table 1.4
Confounding factors
We analysed other potential confounding factors
(eg, mean operative time, number of joint specialists
involved, and surgical approach) to minimise their
effects on the primary and secondary outcomes.12
The indications for TKA did not change at our
institution during the two time periods. Our institutional guidelines state that patients with
Kellgren and Lawrence Grade 3 or 4 end-stage knee
osteoarthritis and debilitating symptoms refractory
to nonoperative treatment are candidates for TKA.
The mean operative times for primary elective TKA
were 1 hour 56 minutes during 1993-20134 and
1 hour 33 minutes during 2014-2021. The difference
was not statistically significant (P=0.170). The
number of joint specialists involved increased from
four to six between the two periods. From 1993 to
2019, all TKAs were exclusively performed using the
conventional approach.
After the computed tomography–based
robotic arm–assisted system for total joint
arthroplasty was introduced in 2019,13 surgeons
at our institution could choose between robotic-assisted
TKA and conventional TKA. Currently,
there are no specific indications for either approach;
the choice remains a matter of surgeon preference.14
To our knowledge, no studies have compared PJI
incidence between robotic-assisted and conventional
TKA; future research should explore the infection
rate associated with each procedure. Patients
undergoing robotic-assisted TKA at our institution
follow the same postoperative protocol established
for those undergoing conventional TKA (follow-up
at 2 weeks, 3 months, 6 months, 12 months, and
annually thereafter). Because robotic-assisted TKA
was recently introduced at our institution, the mean
follow-up duration for patients treated with this
approach was short (14 months; IQR, 4.5-26).
Time to infection
Early-onset PJI occurred in 7.1% (n=1) of patients in
the study cohort, arising 60 days after arthroplasty.
In the 1993-2013 cohort, 29.4% (n=10) of patients
experienced early-onset infection at a median of
17 days after arthroplasty (IQR, 9-32).4 However,
the incidence of early-onset PJI did not differ
significantly between the two cohorts (P=0.095)
[Table 1]. Delayed-onset PJI occurred in 28.6% (n=4)
of patients in the study cohort, occurring at a median
of 6 months after arthroplasty (IQR, 5-7). Late-onset
PJI occurred at a median of 3 years after arthroplasty
(IQR, 2 years 1 month to 3 years 7 months). A larger
proportion of patients experienced infection during
the first year after surgery in the 1993-2013 cohort4 compared with the 2014-2021 cohort (59% vs 36%).
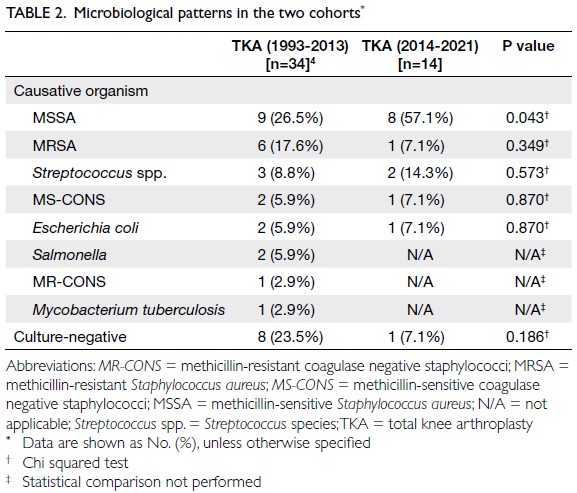
Bacteriology
Methicillin-sensitive S aureus remained the most
common causative organism in cases of PJI between
2014 and 2021, affecting 57.1% (n=8) of patients.
The proportion of PJI cases caused by MSSA was
significantly greater in the 2014-2021 cohort than
in the 1993-2013 cohort, in which 26.5% (n=9) of
patients were infected with MSSA (P=0.043) [Table 2].4
Methicillin-resistant S aureus was the second
most common causative organism in cases of PJI
between 1993 and 2013 (17.6%, n=6)4; Streptococcus spp. (14.3%, n=2) was the second most common
causative organism between 2014 and 2021. The
two cases of streptococcal infection in the 2014-2021 cohort comprised one with Streptococcus dysgalactiae and one with Streptococcus agalactiae.
Other causative organisms in PJI cases within
the 2014-2021 cohort included MRSA (7.1%,
n=1), methicillin-sensitive coagulase-negative
staphylococci (7.1%, n=1), and Escherichia coli (7.1%,
n=1). Methicillin-resistant strains accounted for 40%
of all staphylococcal infections between 1993 and
20134; this proportion was 11.1% between 2014 and 2021. Table 2 compares the microbiological patterns
of PJI between the two cohorts.
There was a non-significant decrease in the
proportion of patients with culture-negative PJI
between the two cohorts, from 23.5% (n=8) between 1993 and 20134 to 7.1% (n=1) between 2014 and 2021 (P=0.186) [Table 2].
Discussion
This study showed that the incidence of PJI after
primary TKA at our institution significantly
decreased. Worldwide, the reported incidence of PJI
after primary elective TKA ranges from 1% to 2%.1 2
Over the years, our institution has implemented
various measures to reduce the incidence of PJI after
TKA, including a preoperative patient optimisation
programme and a restrictive blood management
programme. These measures are summarised in
Table 3.5 6
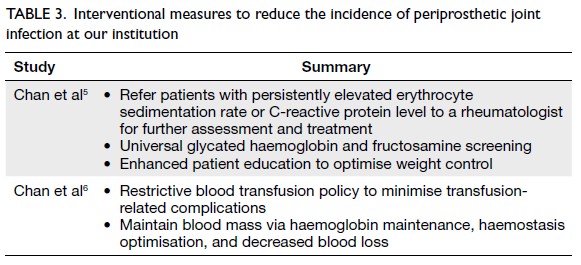
Table 3. Interventional measures to reduce the incidence of periprosthetic joint infection at our institution
Medical risk factors
The association between blood transfusion and
increased perioperative morbidity in patients
undergoing TKA is well documented.15 16 The
American College of Surgeons National Surgical
Quality Improvement Program reported that
patients receiving transfusions experienced
up to a tenfold increase in the risk of adverse
postoperative outcomes.17 Based on these findings,
a more restrictive transfusion approach has been
implemented in the past several years after the
2015 study17 to improve postoperative outcomes. A
retrospective study of 12 590 patients demonstrated
significant decreases in complications, 30-day
readmissions, and hospital length of stay following
implementation of a patient blood management
programme for patients undergoing prosthetic
joint arthroplasty.18 The programme aimed to
reduce transfusion requirements by optimising
red cell mass, minimising blood loss, and defining
appropriate indications for transfusion.18 A patient
blood management programme was introduced
at our institution in 2014; subsequently, the mean
transfusion rate among patients undergoing TKA
decreased from 31.3% in 2013 to 1.9% in 2018.6
Preoperative optimisation
The preoperative optimisation programme at
our institution emphasises the optimisation of
modifiable risk factors for PJI prior to TKA.5
Rheumatological diseases such as rheumatoid
arthritis, juvenile inflammatory arthritis, ankylosing
spondylitis, and psoriatic arthritis are known to
increase the risk of PJI.19 20 A previous review of 2543
TKAs showed that the incidence of PJI was 3.1%
in patients with rheumatoid arthritis, significantly
higher than the 1.2% observed in patients without
rheumatoid arthritis.4 At our institution, patients
with rheumatoid arthritis who exhibit a persistently
elevated erythrocyte sedimentation rate or C-reactive
protein level are referred to a rheumatologist
for further assessment and treatment prior to surgery. Diabetes mellitus is also strongly associated
with an increased risk of PJI. All patients scheduled
for elective TKA at our institution undergo universal
glycated haemoglobin and fructosamine screening,
with referral to an endocrinologist for optimisation
if the glycated haemoglobin level exceeds 7.5%.21
Other modifiable risk factors monitored at our
institution include weight control, vitamin D status,
and nutritional status.22
Antimicrobial resistance
From 2014 to 2021, MSSA was the most common
causative organism in cases of PJI at our institution
(57.1%, n=8). This finding is consistent with the
existing literature, which indicates that S aureus is
the most common causative organism in PJI after
primary joint arthroplasty (19%-29% of cases
worldwide).23 24 25 The unique virulence factors of S
aureus enhance its ability to adhere to implants,
facilitating aggressive biofilm formation and
enabling replication and survival within this
microenvironment.26 27
Owing to the increasing use of antibiotics
in the community, we hypothesised that the
number of antibiotic-resistant causative organisms
would increase in our cohort of patients with
PJI. The SENTRY Antimicrobial Surveillance
Program evaluated 20-year trends in antimicrobial
susceptibility among S aureus isolates across 427
centres in 45 countries.28 The authors reported that
the prevalence of MRSA peaked at 44.2% in 2005-2008, then declined to 42.3% in 2009-2012 and 39.0%
in 2013-2016.28 The incidence of MRSA among PJI
cases in our study was consistent with findings from
other regions. For example, a multicentre study
in New Zealand showed that 9.1% of PJIs were
attributable to MRSA.29
It is well established that early-onset
infection typically occurs during surgery through
intraoperative contamination, whereas late-onset PJI
commonly arises from haematogenous spread.10 We
observed a decrease in the proportion of early-onset
infection between the two cohorts; however, this
difference was not statistically significant (P=0.095).
Additional measures should be implemented to
further reduce the incidence of PJI after primary
TKA at our institution.
Culture-negative periprosthetic joint
infection
In recent years, the incidence of culture-negative PJI
has increased among patients undergoing total joint
arthroplasty.30 This increase has been hypothesised
to result from a higher prevalence of low-virulence
organisms, premature antibiotic treatment, and
failure to use enriched culture media.31 32 An
inability to identify causative organisms in cases of
PJI represents a serious problem for surgeons and infection control teams because of the uncertainties
associated with antimicrobial selection. To reduce
the incidence of culture-negative PJI, our institution
implemented recommendations published by Tan et
al,7 including extending the incubation period, using
blood culture bottles and flasks, and collecting an
adequate number of separate intraoperative tissue
samples from patients with suspected PJI.7 The
incidence of culture-negative PJI at our institution
declined; however, this decline was not statistically
significant (P=0.186).
Limitations
This study had several limitations. First, it included
patients treated at a single academic centre in Hong
Kong; therefore, the findings may not accurately
reflect changing trends in PJI incidence and
bacteriology across the region. Further multicentre
studies are warranted to better understand these
trends. Second, the duration of patient recruitment
differed between the present and former studies (7 years vs 20 years). Third, the number of patients varied
between the two cohorts (2543 vs 2171). Despite this
difference, we proceeded with the 7-year recruitment
period considering the clinical value of evaluating
recent PJI incidence and bacteriology in Hong
Kong. Considerable effort was made to standardise
patient characteristics and baseline co-morbidities
to ensure comparability between cohorts. Fourth,
given the relatively short mean follow-up duration
(4 years 9 months), some cases of late-onset PJI
may have occurred after completion of follow-up.
Fifth, inconsistencies in record-keeping over the
past decade prevented analysis of all documented
risk factors for PJI; this limitation was unavoidable
because of the retrospective study design. Sixth,
the limited number of PJI cases hindered further
subgroup analyses (ie, assessment and comparison
of PJI incidence between conventional and robotic-assisted
approaches). A similar study with a larger
cohort may therefore be beneficial. Despite these
limitations, the present study represents the
largest series of PJI cases in Hong Kong to compare
bacteriological patterns across two time periods.
We believe that the findings have important clinical
implications for PJI management in local hospitals.
Conclusion
This is the first study in Hong Kong to assess changes
in the incidence and microbiological patterns of PJI
after TKA across two time periods. Our findings
have substantial clinical implications, as they
demonstrate the effectiveness of interventional
measures implemented at our institution in reducing
the incidence of PJI, the rate of culture-negative PJI,
and the number of early-onset cases. Prevention
of PJI improves patient outcomes and reduces the economic burden on the healthcare system. Larger,
multicentre, prospective studies are required to
further elucidate bacteriological trends in PJI in
Hong Kong.
Author contributions
Concept and design: All authors.
Acquisition of data: JR Khoo.
Analysis or interpretation of data: JR Khoo, PK Chan.
Drafting of the manuscript: JR Khoo.
Critical revision the manuscript for important intellectual content: All authors.
Acquisition of data: JR Khoo.
Analysis or interpretation of data: JR Khoo, PK Chan.
Drafting of the manuscript: JR Khoo.
Critical revision the manuscript for important intellectual content: All authors.
All authors had full access to the data, contributed to the study, approved the final version for publication, and take responsibility for its accuracy and integrity.
Conflicts of interest
All authors have disclosed no conflicts of interest.
Declaration
This manuscript was presented as an oral presentation at the 42nd Annual Congress of the Hong Kong Orthopaedic
Association (5-6 November 2022, Hong Kong).
Funding/support
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethics approval
This research was approved by the Institutional Review Board
of The University of Hong Kong/Hospital Authority Hong
Kong West Cluster, Hong Kong (Ref No.: UW 25-585). The
requirement for informed patient consent was waived by the
Committee due to the retrospective nature of the research.
References
1. Pulido L, Ghanem E, Joshi A, Purtill JJ, Parvizi J. Periprosthetic joint infection: the incidence, timing, and predisposing factors. Clin Orthop Relat Res 2008;466:1710-5.
Crossref
2. Zimmerli W, Trampuz A, Ochsner PE. Prosthetic-joint infections. N Engl J Med 2004;351:1645-54.
Crossref
3. Premkumar A, Kolin DA, Farley KX, et al. Projected economic burden of periprosthetic joint infection of the hip and knee in the United States. J Arthroplasty 2021;36:148-49.e3.
Crossref
4. Siu KT, Ng FY, Chan PK, Fu H, Yan CH, Chiu KY. Bacteriology and risk factors associated with periprosthetic joint infection after primary total knee arthroplasty: retrospective study of 2543 cases. Hong Kong Med J 2018;24:152-7.
Crossref
5. Chan VW, Chan PK, Fu H, et al. Preoperative optimization to prevent periprosthetic joint infection in at-risk patients. J Orthop Surg (Hong Kong) 2020;28:2309499020947207.
Crossref
6. Chan PK, Hwang YY, Cheung A, et al. Blood transfusions in total knee arthroplasty: a retrospective analysis of a multimodal patient blood management programme. Hong Kong Med J 2020;26:201-7.
Crossref
7. Tan TL, Kheir MM, Shohat N, et al. Culture-negative periprosthetic joint infection: an update on what to expect. JB JS Open Access 2018;3:e0060.
Crossref
8. Roberts SC, Zembower TR. Global increases in antibiotic consumption: a concerning trend for WHO targets. Lancet Infect Dis 2021;21:10-1.
Crossref
9. Workgroup Convened by the Musculoskeletal Infection Society. New definition for periprosthetic joint infection. J Arthroplasty 2011;26:1136-8.
Crossref
10. Parvizi J, Zmistowski B, Berbari EF, et al. New definition for periprosthetic joint infection: from the Workgroup of the Musculoskeletal Infection Society. Clin Orthop Relat Res 2011;469:2992-4.
Crossref
11. Tande AJ, Patel R. Prosthetic joint infection. Clin Microbiol Rev 2014;27:302-45.
Crossref
12. Wang Q, Goswami K, Shohat N, Aalirezaie A, Manrique J, Parvizi J. Longer operative time results in a higher rate of subsequent periprosthetic joint infection in patients undergoing primary joint arthroplasty. J Arthroplasty 2019;34:947-53.
Crossref
13. The University of Hong Kong. HKUMed introduces the latest robotic arm assisted joint replacement technology for enhancing surgical precision [press release]. 2020 May 28. Available from: https://www.hku.hk/press/press-releases/detail/21111.html#:~:text=Robotic%20Arm%20Assisted%20Joint%20Replacement%20Surgery,enable%20accurate%20patient%2Dspecific%20planning . Accessed 1 Apr 2026.
14. Deckey DG, Rosenow CS, Verhey JT, et al. Robotic-assisted total knee arthroplasty improves accuracy and precision compared to conventional techniques. Bone Joint J 2021;103-B(6 Suppl A):74-80.
Crossref
15. Everhart JS, Sojka JH, Mayerson JL, Glassman AH, Scharschmidt TJ. Perioperative allogeneic red blood-cell transfusion associated with surgical site infection after total hip and knee arthroplasty. J Bone Joint Surg Am 2018;100:288-94.
Crossref
16. Lu Q, Peng H, Zhou GJ, Yin D. Perioperative blood management strategies for total knee arthroplasty. Orthop Surg 2018;10:8-16.
Crossref
17. Ferraris VA, Hochstetler M, Martin JT, Mahan A, Saha SP. Blood transfusion and adverse surgical outcomes: the good and the bad. Surgery 2015;158:608-17.
Crossref
18. Loftus TJ, Spratling L, Stone BA, Xiao L, Jacofsky DJ. A patient blood management program in prosthetic joint arthroplasty decreases blood use and improves outcomes. J Arthroplasty 2016;31:11-4.
Crossref
19. Kunutsor SK, Whitehouse MR, Blom AW, Beswick AD; INFORM Team. Patient-related risk factors for periprosthetic joint infection after total joint arthroplasty: a systematic review and meta-analysis. PLoS One 2016;11:e0150866.
Crossref
20. Morrison TA, Figgie M, Miller AO, Goodman SM. Periprosthetic joint infection in patients with inflammatory joint disease: a review of risk factors and current approaches to diagnosis and management. HSS J 2013;9:183-94.
Crossref
21. Duensing I, Anderson MB, Meeks HD, Curtin K, Gililland JM. Patients with type-1 diabetes are at greater risk of periprosthetic joint infection: a population-based, retrospective, cohort study. J Bone Joint Surg Am 2019;101:1860-7.
Crossref
22. Kong L, Cao J, Zhang Y, Ding W, Shen Y. Risk factors for periprosthetic joint infection following primary total hip or knee arthroplasty: a meta-analysis. Int Wound J 2017;14:529-36.
Crossref
23. Triffault-Fillit C, Ferry T, Laurent F, et al. Microbiologic epidemiology depending on time to occurrence of prosthetic joint infection: a prospective cohort study. Clin Microbiol Infect 2019;25:353-8.
Crossref
24. Zeller V, Kerroumi Y, Meyssonnier V, et al. Analysis of postoperative and hematogenous prosthetic joint-infection microbiological patterns in a large cohort. J Infect 2018;76:328-34.
Crossref
25. Benito N, Franco M, Ribera A, et al. Time trends in the aetiology of prosthetic joint infections: a multicentre cohort study. Clin Microbiol Infect 2016;22:732.e1-8.
Crossref
26. Kherabi Y, Zeller V, Kerroumi Y, et al. Streptococcal and Staphylococcus aureus prosthetic joint infections: are they really different? BMC Infect Dis 2022;22:555.
Crossref
27. Sendi P, Rohrbach M, Graber P, Frei R, Ochsner PE, Zimmerli W. Staphylococcus aureus small colony variants in prosthetic joint infection. Clin Infect Dis 2006;43:961-7.
Crossref
28. Diekema DJ, Pfaller MA, Shortridge D, Zervos M, Jones RN. Twenty-year trends in antimicrobial susceptibilities among Staphylococcus aureus from the SENTRY Antimicrobial Surveillance Program. Open Forum Infect Dis 2019;6(Suppl 1):S47-53.
Crossref
29. Ravi S, Zhu M, Luey C, Young SW. Antibiotic resistance in early periprosthetic joint infection. ANZ J Surg 2016;86:1014-8.
Crossref
30. Bejon P, Berendt A, Atkins BL, et al. Two-stage revision for prosthetic joint infection: predictors of outcome and the role of reimplantation microbiology. J Antimicrob Chemother 2010;65:569-75.
Crossref
31. Berbari EF, Marculescu C, Sia I, et al. Culture-negative prosthetic joint infection. Clin Infect Dis 2007;45:1113-9.
Crossref
32. Malekzadeh D, Osmon DR, Lahr BD, Hanssen AD, Berbari EF. Prior use of antimicrobial therapy is a risk factor for culture-negative prosthetic joint infection. Clin Orthop Relat Res 2010;468:2039-45.
Crossref