Hong Kong Med J 2023 Aug;29(4):324–9 | Epub 10 Jul 2023
© Hong Kong Academy of Medicine. CC BY-NC-ND 4.0
ORIGINAL ARTICLE
Disease-related complications in patients with metastatic hormone-sensitive prostate cancer
CF Ng, FRCSEd (Urol), FHKAM (Surgery)1; Christy WH Mak, MB, ChB1; Samson YS Chan, MB, ChB1; ML Li, MNurs2; CH Leung, MPH1; Jeremy YC Teoh, MB, BS1; Peter KF Chiu, MB, ChB1; Peggy SK Chu, MB, BS2
1 Department of Surgery, SH Ho Urology Centre, The Chinese University of Hong Kong, Hong Kong SAR, China
2 Department of Surgery, Tuen Mun Hospital, Hong Kong SAR, China
Corresponding author: Prof CF Ng (ngcf@surgery.cuhk.edu.hk)
Abstract
Introduction: Prostate-specific antigen-based
screening for prostate cancer reportedly does not
improve cancer-specific survival. However, there
remain concerns about the increasing incidence
of advanced disease at initial presentation. Here,
we investigated the incidences and types of
complications that occur during the course of
disease in patients with metastatic hormone-sensitive
prostate cancer (mHSPC).
Methods: This study included 100 consecutive patients who were diagnosed with mHSPC at five
hospitals from January 2016 to August 2017. Analyses
were conducted using patient data extracted from
a prospectively collected database, along with
information about complications and readmission
obtained from electronic medical records.
Results: The median patient age was 74 years and
the median serum prostate-specific antigen level at
diagnosis was 202.5 ng/mL. Ninety-nine patients
received androgen deprivation therapy; 17 of these
patients also received chemotherapy. During a mean
follow-up period of 32.9 months, 41 patients reported
bone pain; of these patients, 21 developed pathologic
fractures and eight had cord compression. Twenty-eight
patients developed retention of urine; of these
patients, 10 (36%) required surgery and 11 (39%)
required long-term urethral catheter use. Among 15 patients who developed ureteral obstruction,
four (27%) required ureteral stenting and four (27%)
required long-term nephrostomy drainage. Other
complications included anaemia (41%) and deep
vein thrombosis (4%). Fifty-nine (59%) patients had
≥1 unplanned hospital admission during the course
of disease; 16% of such patients had >5 episodes of
readmission.
Conclusion: Among patients with mHSPC, 70% experienced disease-related complications and
unplanned hospital admissions, which substantially
burdened both patients and the healthcare system.
New knowledge added by this study
- Advanced prostate cancer was associated with serious disease-related complications, which required surgical interventions and unplanned hospital admissions.
- The role of prostate-specific antigen-based screening in prostate cancer should be reconsidered.
- Early prostate cancer detection may help reduce disease-related comorbidities.
- Advances in diagnostic tools and the use of active surveillance may help to minimise the harms associated with diagnostic procedures and overtreatment of early disease.
Introduction
Prostate cancer (PCa) is the second most common
cancer in men worldwide, and its incidence is
rapidly increasing in Hong Kong.1 Despite increased
awareness of PCa, many patients in Hong Kong
are diagnosed with advanced disease involving
metastasis, which does not respond to curative
treatment. Previously, there was considerable interest in using serum prostate-specific antigen
(PSA) for screening and early detection of PCa,
with the hope that this approach would improve
treatment outcomes. However, a Cochrane review
showed that screening was associated with more
frequent detection of localised disease, but there
was no cancer-specific survival benefit; patients
may even be harmed by complications associated with diagnostic and therapeutic procedures.2
Thus, the use of PSA-based screening has declined
in the United States in the past decade.3 Notably,
a concomitant epidemiologic shift from early to
advanced disease (ie, reverse stage migration) has
occurred, leading to serious concerns within the
urological community.4
The slow progression and protracted clinical
course of PCa are well-known.5 Patients with metastatic disease can survive for several years
before death, which may also be caused by other
medical conditions.6 However, during the course of disease, patients may experience complications
related to PCa, including skeletal-related events and
urinary tract complications. These complications
can threaten a patient’s quality of life and lead to
increased medical expenses. To our knowledge,
minimal information is available regarding the
course of disease (particularly complications) in
patients with advanced PCa.
Here, we assessed the incidences of
complications and unplanned hospital admissions
among patients with metastatic hormone-sensitive
PCa (mHSPC). This information may provide useful
insights regarding the disease course and treatment
needs of patients with mHSPC. It may also provide a
more comprehensive understanding of the potential
benefits of PSA-based screening in the management
of patients with PCa.
Methods
The Asian Prostate Cancer (A-CaP) Study, a
prospective multi-nation study designed to
investigate real-world clinical management of PCa
in Asia, began in January 2016. Patients with a
diagnosis of PCa were recruited into the study.7 8
Clinical information was prospectively collected,
including baseline patient and disease parameters,
treatment received, and clinical progress. Thus far,
>30 000 patients from 14 Asian countries have been
recruited (based on an unpublished annual meeting
report of A-CaP meeting held on 24 November 2020).
In Hong Kong, five hospitals (Alice Ho Miu
Ling Nethersole Hospital, North District Hospital,
Pok Oi Hospital, Prince of Wales Hospital, and Tuen
Mun Hospital) formed the Hong Kong Prostate
Cancer (HK-CaP) study group as part of the A-CaP
project. Since 2016, all patients who presented to
these hospitals (as an outpatient or inpatient) and
received a diagnosis of PCa were recruited into the
Hong Kong cohort, which currently includes >1000
cases of PCa (across all stages). In this study, we
identified the first 100 consecutive patients with
mHSPC (with no additional inclusion or exclusion
criteria), then extracted their data from the HK-CaP
database. Analyses were conducted using the
extracted data, along with clinical information that
had been retrospectively retrieved from electronic
medical records.
Continuous variables are presented as medians
with interquartile ranges, while categorical variables
are presented as frequencies and percentages. All
statistical analyses were performed using R version
3.6.2 (R Foundation for Statistical Computing,
Vienna, Austria). Two-sided P values <0.05 were
considered statistically significant.
Results
Study population
From January 2016 to August 2017, 100 consecutive
patients with mHSPC were included in this study.
The median age was 74 years (range, 50-93) [Table].
Overall, 17% of patients had no history of chronic
disease. In contrast, 55%, 25%, 10%, and 63% of
patients had pre-existing hypertension, diabetes,
cerebrovascular disease, and history of smoking,
respectively. The median serum PSA level at
diagnosis was 202.5 ng/mL (range, 0.9-6260), and
83% of patients had abnormal findings on digital
rectal examination at initial presentation. Thirteen
patients (13%) had a family history of PCa. Most
patients presented with symptoms, including lower
urinary tract symptoms (62%), haematuria (9%), and
constitutional symptoms (22%). Only 7% of patients
had incidental findings of an abnormal serum PSA
level during wellness screening.
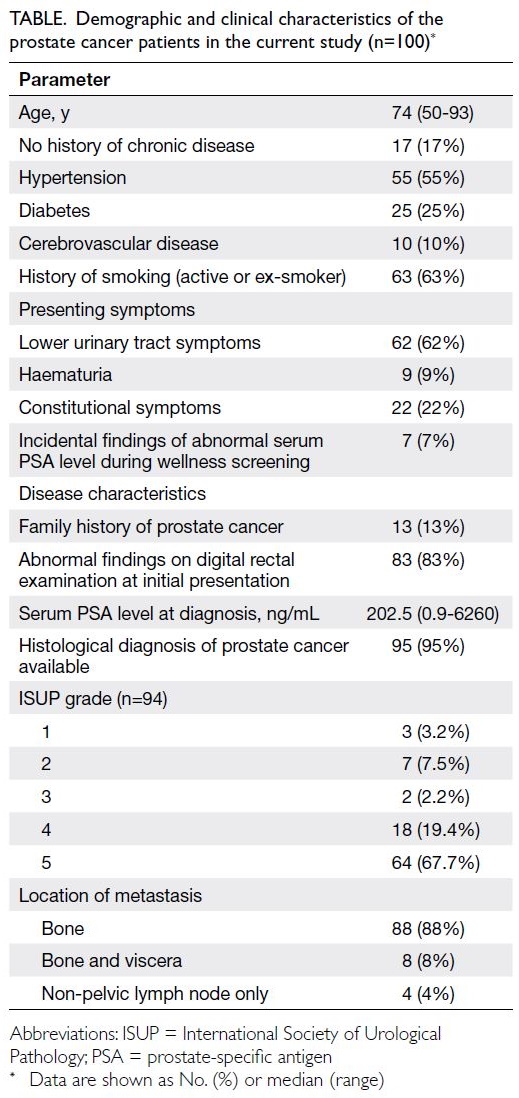
Table. Demographic and clinical characteristics of the prostate cancer patients in the current study (n=100)
Most patients (95%) had histologically proven
PCa; the remaining 5% of patients had a clinical
diagnosis of PCa based on a high serum PSA level
(219-2779 ng/mL), with or without abnormal
findings on digital rectal examination. Among
patients with a histological diagnosis (n=94), the
proportions with International Society of Urological
Pathology grades 1 to 5 were 3.2%, 7.5%, 2.2%, 19.4%,
and 67.7%, respectively. Most patients had bone
metastases (96%); among them, eight patients also
had visceral metastases. The remaining four patients
were diagnosed with non-pelvic lymph node
metastases (M1a) at initial presentation (Table).
Treatment received
With the exception of one patient who selected
watchful waiting, all patients received androgen
deprivation therapy. Initial androgen deprivation
therapies included luteinising hormone-releasing
hormone antagonists (46 patients, 46%), luteinising
hormone-releasing hormone agonists (with
short-term antiandrogen treatment during flares)
[28 patients, 28%], and bilateral orchidectomy
(25 patients, 25%). Seventeen patients also received
upfront chemotherapy for advanced disease; no
patient received upfront abiraterone.
The mean follow-up period was 32.9 months
(range, 0.3-54.2). During follow-up, 59 patients
developed metastatic castration-resistant prostate
cancer (mCRPC). Among these patients, older-generation
antiandrogens, docetaxel, abiraterone,
enzalutamide, and prednisolone alone were used
by 26 (44.1%), nine (15.3%), 17 (28.8%), 18 (30.5%),
and five (8.5%) patients, respectively. Thirty-two
patients (32%) also received palliative radiotherapy
for symptom control. Seven (11.9%) patients with
mCRPC used denosumab for bone protection.
The median overall survival time was 3.7 years; 33
(67.3%) patients died of PCa and 16 (32.7%) patients
died of other causes, and none of these causes were
cardiovascular events (Fig 1).
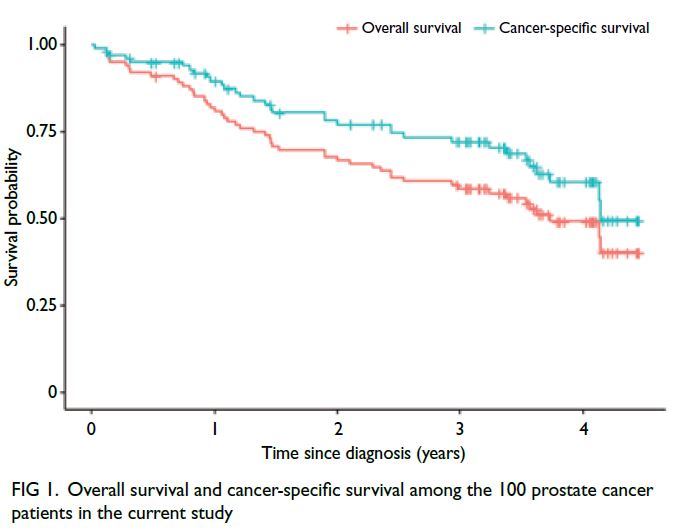
Figure 1. Overall survival and cancer-specific survival among the 100 prostate cancer patients in the current study
Complications
Only 30 patients (30%) had no disease-related
complications. Among the observed complications,
skeletal-related events were most common: 41
patients reported bone pain during follow-up.
Of these 41 patients, 35 (85%) required regular
analgesics, and 12 (29%) required opioid analgesics
for pain control. Approximately half of the 41
patients (ie, 20 patients, 49%) required palliative
radiotherapy for bony metastases. Moreover, 21
patients developed pathologic fractures, and eight
patients had cord compression.
The second most common complication was
retention of urine secondary to prostatic obstruction
(28 patients, 28%). Among these 28 patients, only
seven (25%) were able to discontinue urethral catheter use. Of the remaining patients, 10 (36%) required endoscopic prostatic surgery and 11 (39%)
required long-term urethral catheter use.
Among 15 patients who developed ureteral
obstruction, four (27%) required ureteral stenting
and four (27%) required long-term nephrostomy
drainage. The remaining seven (47%) patients
received conservative management. During follow-up,
17 patients developed gross haematuria.
Forty-one patients (41%) developed anaemia
(haemoglobin level <10 g/dL); 22 of these patients
(53.7%) required transfusion. Furthermore, only
four of the 41 patients received chemotherapy to
manage mCRPC. Therefore, most cases of anaemia
were presumably the direct result of PCa. Other
complications included deep vein thrombosis (4%), psychiatric problems (adjustment disorder or depression) [4%], and suicidal ideation (1%).
Fifty-nine (59%) patients experienced ≥1
unplanned hospital admissions during the course of
disease. The proportions of patients with 1-5, 6-10,
and >10 unplanned hospital admission episodes
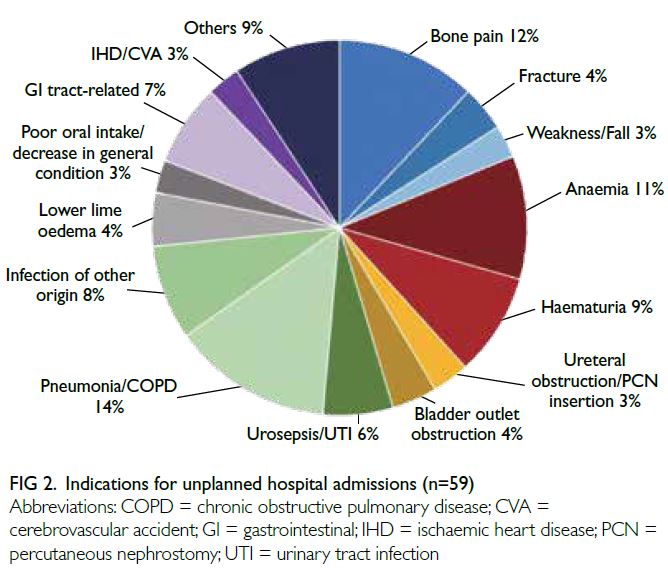
were 43%, 10%, and 6%, respectively. The indications
for these admissions included skeletal-related
events (bone pain, fracture, and fall, 19%), urinary
complications (haematuria, ureteric obstruction,
and bladder outcome obstruction, 16%), and sepsis (urosepsis, pneumonia, and infection of other origin,
28%) [Fig 2]. At least half of the admissions were
presumably the direct result of PCa, such as bone
pain and urinary complications.
Discussion
In this prospective observational study, over a mean
follow-up period of approximately 32 months,
PCa-related complications occurred in 70% of 100
patients with newly diagnosed mHSPC; around 60%
of these patients had unplanned hospital admissions
during the course of disease. Slightly less than half of
the patients died during this study period. More than
two-thirds of the patients died of PCa. Also, many of
the patients experienced PCa-related complications
had received various treatments for their disease
and complications. These real-world data provide
insights concerning the natural course of disease
in patients with advanced PCa; they also suggest
a need to reconsider management approaches for
such patients. Additionally, these data may help
to evaluate the potential benefits of PSA-based
screening for PCa.
The primary purpose of disease screening
involves identifying a disease in its early or
asymptomatic stages, which can allow more
effective treatment and support better outcomes.
Consequently, disease-related mortality and
complications can be minimised, while improving
patient quality of life.9 Early intervention may also
help to reduce medical expenses through disease
treatment at an earlier stage, rather than a later
and more complex stage. Prostate cancer fulfils
some of the criteria for disease screening: it is a
common cancer, displays a latent disease stage,
and has acceptable diagnostic tests and effective
treatments.10
Controversies related to prostate-specific antigen–based prostate cancer screening
However, PSA-based PCa screening is among the
most controversial topics in urology. In a review
based on data from five randomised trials of PCa
screening, no cancer-specific survival benefit
was identified; moderate harm was caused by
diagnostic procedures.2 Moreover, overdiagnosis
and overtreatment were common; they could cause
treatment-related harm. Therefore, Chou et al11
recommended against PSA-based screening. This
recommendation led to a decline in the use of
PSA testing during the past decade.3 As expected,
the overall incidence of PCa, particularly low-risk
disease, has decreased in recent years.12
Unfortunately, there has been a concomitant
increase in diagnoses of advanced and higher-grade
disease (ie, reverse stage migration).4 13 Furthermore,
the European Randomized Study of Screening for
Prostate Cancer revealed a 30% reduction in the relative risk of metastatic disease in the screened
population, compared with the non-screened
population.14 Therefore, PSA-based screening may
at least reduce the number of patients who present
with metastatic disease.
Potential benefits of early cancer detection
Discussions of PSA-based screening have mainly
focused on survival benefits (ie, decreases in overall
and cancer-specific or all-cause mortality), as well as
the harms associated with screening procedures and
overtreatment of low-risk disease.2 However, there
has been minimal consideration of the potential
advantages of screening in terms of preventing
disease-related complications, as well as the
negative effects of advanced disease on quality of life.
Moreover, there has been little discussion regarding
the potential financial implications of managing
advanced PCa and its complications.
Rather than investigating the value of PSA-based
screening, the present study was conducted
to fill the gap in knowledge regarding the course
of disease in patients who present with mHSPC. In
our cohort, 70% of patients developed PCa-related
complications (eg, bone, urinary tract, and anaemia)
during the course of disease. We found that nearly
60% of patients had unplanned hospital admissions
for various complications; this proportion was much
greater than the observed readmission rate of 6.5%
(6 of 93 patients) for localised PCa over a period of
>5 years in Hong Kong.15 Therefore, early diagnosis
may be the only way to minimise the incidence of
PCa-related complications in patients with mHSPC.
Treatment-related complications
In addition to PCa-related complications, cancer
treatment can cause adverse effects in patients with
mHSPC. Typical androgen deprivation therapy is
notorious for causing cardiovascular and metabolic
complications.16 17 18 Treatments specifically for
mCRPC, such as chemotherapy or next-generation
androgen receptor targeting agents, are also
associated with adverse effects such as febrile
neutropenia, gastric distress, hypertension, and
cardiac events.19 Additionally, the direct and indirect
costs of treatment place additional financial burdens
on patients and their families (ie, ‘financial toxicity’),
impose a psychological burden, and adversely
affect the quality of life of patients.20 Therefore,
earlier diagnosis of PCa may help patients to avoid
progression to advanced or metastatic disease,
thereby reducing suffering associated with advanced
disease and its treatment-related complications.
Limitations
This study had some limitations. First, bone-protecting
agents, which are relatively expensive, are not commonly used in Hong Kong. The absence
of such agents may have led to a higher rate of
skeletal-related events in our patients. Second, only
17 patients received upfront chemotherapy and no
patients received upfront next-generation androgen
receptor–targeting agents. Thus, we could not assess
whether the use of these newer treatment approaches
could minimise disease-related complications. Third,
we did not collect data regarding the quality of life of
patients, which would help to clarify the effects of
disease-related complications on patients and their
families.
In recent years, there have been many advances
in PCa diagnosis and treatment. The use of new
markers for PCa, such as the Prostate Health Index,21
urinary exosomes,22 and multiparametric magnetic resonance imaging,23 has considerably improved
diagnostic accuracy and reduced the need for
prostate biopsy (ie, to rule out false-positives based
on elevated PSA levels). The use of the transperineal
route for prostate biopsy has also minimised
prostate biopsy–related complications.24 In addition,
active surveillance for low-risk PCa has mitigated
possible harms associated with overtreatment.25
In combination, these new advances and better
knowledge of the disease course will improve
support for PCa screening, thereby minimising
disease-related suffering.
Conclusion
In this observational study, 70% of patients with
metastatic PCa at initial presentation had various
PCa-related complications and many unplanned
hospital admissions during the course of disease.
Although there remains controversy concerning
whether PSA-based PCa screening is beneficial for
cancer-specific survival, the recent observation of
reverse stage migration in PCa related to decreased
PSA testing is problematic for PCa management.
Advanced PCa may be associated with significant
disease-related complications; it can also place
an increased burden on the healthcare system by
contributing to more unplanned hospital admissions.
Thus, there is a need for more comprehensive
assessment of the value of PSA-based PCa screening
in terms of preventing disease-related morbidity
and mortality. Advances in diagnostic tools and
the use of active surveillance may help reduce the
harms associated with diagnostic procedures and
overtreatment of early disease.
Author contributions
Concept or design: CF Ng.
Acquisition of data: CWH Mak, SYS Chan, ML Li, CH Leung.
Analysis or interpretation of data: CWH Mak, CH Leung.
Drafting of the manuscript: CF Ng, CWH Mak.
Critical revision of the manuscript for important intellectual content: JYC Teoh, PKF Chiu, PSK Chu.
Acquisition of data: CWH Mak, SYS Chan, ML Li, CH Leung.
Analysis or interpretation of data: CWH Mak, CH Leung.
Drafting of the manuscript: CF Ng, CWH Mak.
Critical revision of the manuscript for important intellectual content: JYC Teoh, PKF Chiu, PSK Chu.
All authors had full access to the data, contributed to the study, approved the final version for publication, and take responsibility for its accuracy and integrity.
Conflicts of interest
As editors of the journal, CF Ng and JYC Teoh were not
involved in the peer review process. Other authors have disclosed no conflicts of interest.
Declaration
This research has been presented as oral presentation in the 25th Annual Scientific Meeting of Hong Kong Urological Association held on 25 October 2020 in Hong Kong SAR, China.
Funding/support
The research received support from J-CaP (Japan Study Group of Prostate Cancer) and Takeda Pharmaceutical Company Limited. The funder had no role in study design, data collection/analysis/interpretation, or manuscript preparation.
Ethics approval
The study protocol was approved by the Joint Chinese
University of Hong Kong–New Territories East Cluster
Clinical Research Ethics Committee (Ref No.: 2014.251) and
registered in ClinicalTrials.gov (Identifier: NCT03344835).
Informed patient consent has been waived by the Committee
because of the observational nature of the study.
References
1. Teoh JY, Hirai HW, Ho JM, Chan FC, Tsoi KK, Ng CF. Global incidence of prostate cancer in developing and
developed countries with changing age structures. PLoS
One 2019;14:e0221775. Crossref
2. Ilic D, Neuberger MM, Djulbegovic M, Dahm P. Screening for prostate cancer. Cochrane Database Syst Rev 2013;(1):CD004720. Crossref
3. Fleshner K, Carlsson SV, Roobol MJ. The effect of the USPSTF PSA screening recommendation on prostate cancer incidence patterns in the USA. Nat Rev Urol 2017;14:26-37. Crossref
4. Reese AC, Wessel SR, Fisher SG, Mydlo JH. Evidence of prostate cancer “reverse stage migration” toward more advanced disease at diagnosis: data from the Pennsylvania Cancer Registry. Urol Oncol 2016;34:335.e21-8. Crossref
5. Popiolek M, Rider JR, Andrén O, et al. Natural history of early, localized prostate cancer: a final report from three decades of follow-up. Eur Urol 2013;63:428-35. Crossref
6. Tangen CM, Faulkner JR, Crawford ED, et al. Ten-year survival in patients with metastatic prostate cancer. Clin Prostate Cancer 2003;2:41-5. Crossref
7. Akaza H, Hirao Y, Kim CS, et al. Asia prostate cancer study (A-CaP Study) launch symposium. Prostate Int 2016;4:88-96. Crossref
8. Lojanapiwat B, Lee JY, Gang Z, et al. Report of the third Asian Prostate Cancer study meeting. Prostate Int 2019;7:60-7. Crossref
9. The Society of Radiographers. NHS screening
programmes–purpose of the programmes. Available from: https://www.sor.org/getmedia/160c519f-7d43-4138-95f4-84e955bc2857/nhs_population_screening_2.pdf. Accessed 10 Jun 2023.
10. Wilson JM, Jungner G, World Health Organization. Public Health Papers. Principles and practice of screening for
disease. World Health Organization, 1968. Available from:
https://apps.who.int/iris/handle/10665/37650. Accessed 10 Jun 2023.
11. Chou R, Croswell JM, Dana T, et al. Screening for prostate cancer: a review of the evidence for the U.S. Preventive
Services Task Force. Ann Intern Med 2011;155:762-71. Crossref
12. Jemal A, Culp MB, Ma J, Islami F, Fedewa SA. Prostate cancer incidence 5 years after US Preventive Services Task
Force Recommendations against screening. J Natl Cancer
Inst 2021;113:64-71. Crossref
13. Hu JC, Nguyen P, Mao J, et al. Increase in prostate cancer distant metastases at diagnosis in the United States. JAMA Oncol 2017;3:705-7. Crossref
14. Schröder FH, Hugosson J, Carlsson S, et al. Screening for
prostate cancer decreases the risk of developing metastatic
disease: findings from the European Randomized Study
of Screening for Prostate Cancer (ERSPC). Eur Urol
2012;62:745-52. Crossref
15. Ng CF, Kong KY, Li CY, et al. Patient-reported outcomes after surgery or radiotherapy for localised prostate cancer:
a retrospective study. Hong Kong Med J 2020;26:95-101. Crossref
16. Hu JR, Duncan MS, Morgans AK, et al. Cardiovascular
effects of androgen deprivation therapy in prostate cancer:
contemporary meta-analyses. Arterioscler Thromb Vasc
Biol 2020;40:e55-64. Crossref
17. Ng CF, Chiu PK, Yee CH, Lau BS, Leung SC, Teoh JY.
Effect of androgen deprivation therapy on cardiovascular
function in Chinese patients with advanced prostate
cancer: a prospective cohort study. Sci Rep 2020;10:18060. Crossref
18. Shore ND, Saad F, Cookson MS, et al. Oral relugolix for androgen-deprivation therapy in advanced prostate cancer.
N Engl J Med 2020;382:2187-96. Crossref
19. Tonyali S, Haberal HB, Sogutdelen E. Toxicity, adverse events, and quality of life associated with the treatment of
metastatic castration-resistant prostate cancer. Curr Urol
2017;10:169-73. Crossref
20. Rotter J, Spencer JC, Wheeler SB. Financial toxicity in advanced and metastatic cancer: overburdened and
underprepared. J Oncol Pract 2019;15:e300-7. Crossref
21. Chiu PK, Ng CF, Semjonow A, et al. A multicentre
evaluation of the role of the Prostate Health Index (PHI)
in regions with differing prevalence of prostate cancer:
adjustment of PHI reference ranges is needed for European
and Asian settings. Eur Urol 2019;75:558-61. Crossref
22. Wang WW, Sorokin I, Aleksic I, et al. Expression of small noncoding RNAs in urinary exosomes classifies
prostate cancer into indolent and aggressive disease. J Urol
2020;204:466-75. Crossref
23. Kasivisvanathan V, Rannikko AS, Borghi M, et al. MRI-targeted or standard biopsy for prostate-cancer diagnosis. N Engl J Med 2018;378:1767-77. Crossref
24. Chiu PK, Lo KL, Teoh JY, et al. Sectoral cancer detection and tolerability of freehand transperineal prostate biopsy
under local anaesthesia. Prostate Cancer Prostatic Dis
2021;24:431-8. Crossref
25. Kinsella N, Helleman J, Bruinsma S, et al. Active surveillance for prostate cancer: a systematic review of
contemporary worldwide practices. Transl Androl Urol
2018;7:83-97. Crossref