© Hong Kong Academy of Medicine. CC BY-NC-ND 4.0
CASE REPORT
Extraosseous myeloma of liver mimicking
multifocal hepatocellular carcinoma where a
distinction has to be made: two case reports
HM Kwok, MB, BS, FRCR#; Eugene Sean Lo, MB, BS#; T Wong, FRCR, FHKAM (Radiology); Heather HC Lee, MB, BS/BSC, FRCR; HT Chau, MB, BS; FH Ng, FRCR, FHKAM (Radiology); WH Luk, FRCR, FHKAM (Radiology); Johnny KF Ma, FRCR, FHKAM (Radiology)
Department of Radiology, Princess Margaret Hospital, Hong Kong
# Equal contribution
Corresponding author: Dr HM Kwok (khm778@ha.org.hk)
Case report
Case 1
A 67-year-old man with kappa light chain multiple
myeloma (MM) had a baseline negative skeletal
survey and had undergone dual-tracer positron
emission tomography–computed tomography (CT)
with fluorine-18 fluorodeoxyglucose and carbon-11
acetate. An initial biochemical response to combined
chemotherapy with bortezomib, thalidomide
and dexamethasone later plateaued; therefore, he
was switched to second-line chemotherapy with
ixazomib, lenalidomide and dexamethasone. He
then developed progressively deranged liver function and new-onset pancytopenia with fever during the
first cycle. Ultrasound revealed multiple bi-lobar
hepatic hypoechoic lesions, some with a central
echogenic focus surrounded by hypoechoic rim
(target appearance), suggesting possible hepatic
candidiasis. Nonetheless there was progressive
worsening of liver function and recurrent fever
despite intravenous antifungal therapy. Urgent
multiphasic CT revealed a small arterial enhancing
nodule in hepatic segment V with washout. A newly
developed lytic sacral lesion was suspected to be
myeloma involvement. The liver CT 3 weeks later
showed an interval increase in size and number of
these liver lesions with similar enhancement pattern. Multifocal hepatocellular carcinoma (HCC) was one
of the prime differential diagnoses (Fig 1). Hepatitis B
and C tests were negative. Tumour markers including
alpha-fetoprotein were normal. Due to the rapid
interval lesion enlargement, absence of risk factor for
HCC, and the need to exclude possible opportunistic
fungal infection, ultrasound-guided liver biopsy was
performed and confirmed myeloma involvement.
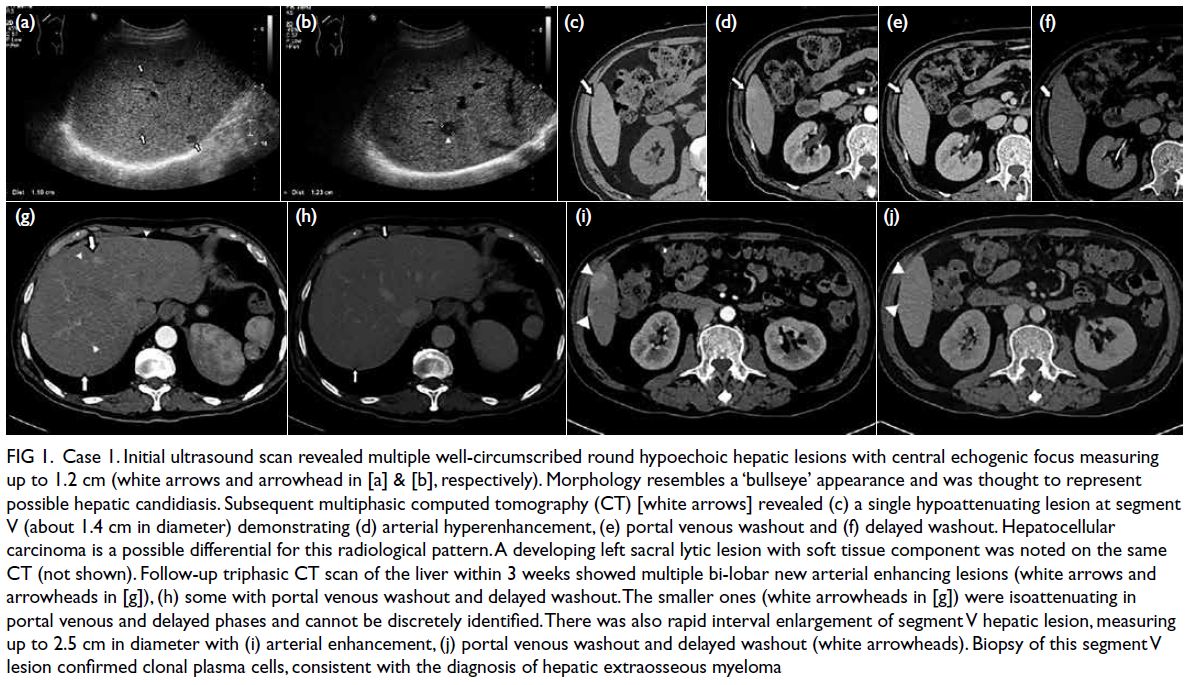
Figure 1. Case 1. Initial ultrasound scan revealed multiple well-circumscribed round hypoechoic hepatic lesions with central echogenic focus measuring up to 1.2 cm (white arrows and arrowhead in [a] & [b], respectively). Morphology resembles a ‘bullseye’ appearance and was thought to represent possible hepatic candidiasis. Subsequent multiphasic computed tomography (CT) [white arrows] revealed (c) a single hypoattenuating lesion at segment V (about 1.4 cm in diameter) demonstrating (d) arterial hyperenhancement, (e) portal venous washout and (f) delayed washout. Hepatocellular carcinoma is a possible differential for this radiological pattern. A developing left sacral lytic lesion with soft tissue component was noted on the same CT (not shown). Follow-up triphasic CT scan of the liver within 3 weeks showed multiple bi-lobar new arterial enhancing lesions (white arrows and arrowheads in [g]), (h) some with portal venous washout and delayed washout. The smaller ones (white arrowheads in [g]) were isoattenuating in portal venous and delayed phases and cannot be discretely identified. There was also rapid interval enlargement of segment V hepatic lesion, measuring up to 2.5 cm in diameter with (i) arterial enhancement, (j) portal venous washout and delayed washout (white arrowheads). Biopsy of this segment V lesion confirmed clonal plasma cells, consistent with the diagnosis of hepatic extraosseous myeloma
Case 2
A 51-year-old woman with lambda light chain
MM diagnosed in 2011 was in remission following
treatment with bortezomib, thalidomide and
dexamethasone. Autologous peripheral blood
stem cell transplantation was performed but she
developed disease relapse 18 months later, salvaged
by combined bortezomib-melphalan-prednisone.
She presented within a year with a second relapse and
a 1-week history of fever, increasing diffuse bone pain
and abdominal distention. Mild hepatosplenomegaly
was noted on physical examination. Pancytopenia
was evident (haemoglobin 7.6 g/dL, platelet count
17×109/L, white blood cell count 1.7×109/L). Blood
culture and hepatitis markers were negative.
Ultrasound revealed a hypoechoic lesion at the
right hepatic lobe, bi-lobar hepatic hyperechoic lesions with hypoechoic rim and mild ascites.
In the presence of her high swinging fever, liver
abscesses were suspected. Hepatosplenomegaly was
confirmed on CT performed 1 day later. In addition,
multiple hypodense liver lesions, most of which were
subcapsular, were observed. They showed arterial
contrast enhancement and became hypo-enhancing
in the portovenous phase. No rim-enhancing lesions
were present to suggest abscess formation. There were
innumerable lytic lesions in bone, including partial
collapse of T11 and L1 and a lytic lesion at the right
transverse process of T10 with enhancing soft tissue
mass (Fig 2). Overall features suggested progressive
disease with presumably myelomatous involvement
of liver and bone. She was prescribed two cycles
of lenalidomide, bortezomib, and dexamethasone
followed by four more cycles of lenalidomide and
dexamethasone. Follow-up CT showed interval
resolution of the previous noted bi-lobar liver
lesions and sub-centimetre hypo-enhancing focus
representing post-treatment change or residual
disease, supporting the presumption of liver
extraosseous myeloma (EM). Repeat bone marrow
examination revealed hypercellular marrow with
residual plasma cell myeloma. She then underwent
hematopoietic stem cell transplantation.
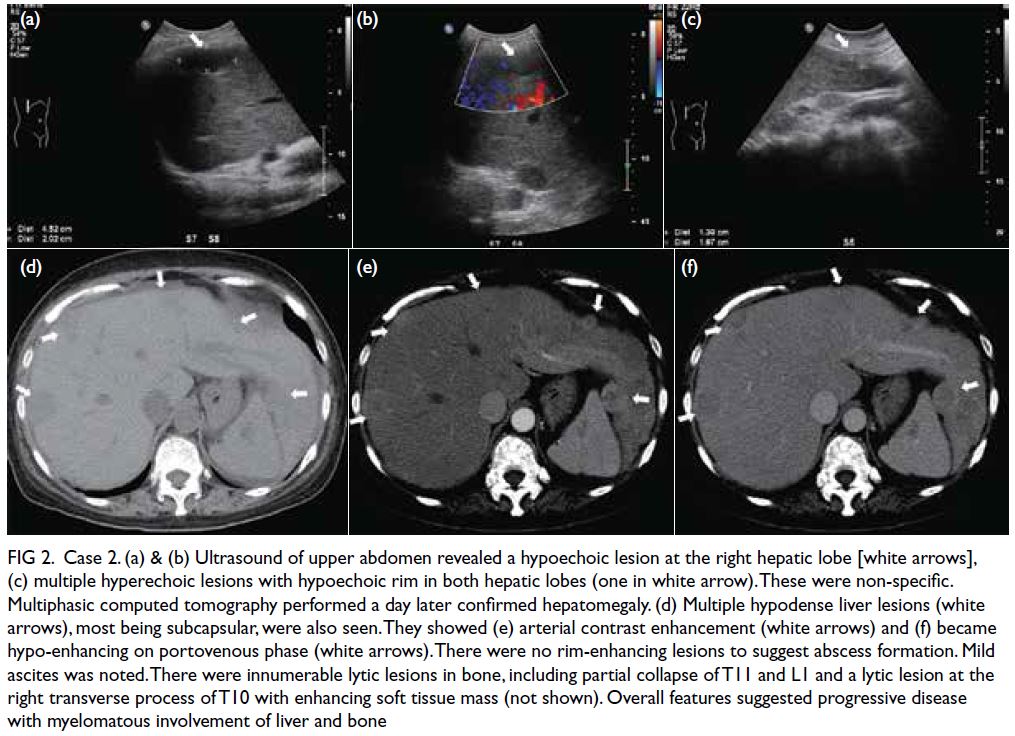
Figure 2. Case 2. (a) & (b) Ultrasound of upper abdomen revealed a hypoechoic lesion at the right hepatic lobe [white arrows], (c) multiple hyperechoic lesions with hypoechoic rim in both hepatic lobes (one in white arrow). These were non-specific. Multiphasic computed tomography performed a day later confirmed hepatomegaly. (d) Multiple hypodense liver lesions (white arrows), most being subcapsular, were also seen. They showed (e) arterial contrast enhancement (white arrows) and (f) became hypo-enhancing on portovenous phase (white arrows). There were no rim-enhancing lesions to suggest abscess formation. Mild ascites was noted. There were innumerable lytic lesions in bone, including partial collapse of T11 and L1 and a lytic lesion at the right transverse process of T10 with enhancing soft tissue mass (not shown). Overall features suggested progressive disease with myelomatous involvement of liver and bone
Discussion
Extraosseous myeloma is an uncommon form of MM
associated with poorer prognosis and survival.1 2 It is
caused by migration of malignant plasma cells from
the bone marrow microenvironment. The presence of
extraosseous involvement of MM is not uncommon;
it has been previously reported in more than 63%
of patients in an autopsy series, with 28 to 30%
having liver involvement.1 The reticuloendothelial
system (liver, spleen and lymph nodes) is the most
commonly affected extraosseous site.2 Although well
documented in the pathology literature, this clinical
entity remains under-recognised and underreported
in radiology.
We report two cases of multifocal EM of
the liver in two Chinese patients from a tertiary
hospital in Hong Kong, mimicking multifocal HCC
on multiphasic CT. To the best of our knowledge,
this pattern has not been reported previously. First,
we aim to increase radiologist awareness of the
hypervascular multinodular pattern of liver EM.
Second, HCC is common in Southeast Asia including
Hong Kong and remains an imaging diagnosis with
no histological confirmation required prior to
treatment. There are overlapping imaging features
of both extraosseous MM in liver and HCC. Hence,
biopsy is needed for differentiation.
Imaging findings of EM are highly variable and
non-specific. The two most common presentations
are the more common diffuse form with hepatomegaly
in the absence of a focal lesion due to diffuse liver
parenchymal infiltration and the focal nodular form
with hypodense non-calcified nodule and minimal
enhancement. On ultrasound, focal patterns of
involvement can be hypoechoic, hyperechoic, mixed
or target (isoechoic nodule with hypoechoic rim).
On CT, focal lesions are generally described as
hypoattenuating with minimal enhancement and
no calcification. On magnetic resonance imaging,
focal lesions may be hyper- or hypo-intense on T1-weighted images and hyperintense on T2-weighted
images with minimal gadolinium enhancement.2 3
Scarce literature has documented hypervascular
enhancement patterns with washout on multiphasic
CT or magnetic resonance imaging, and only few
case reports have reported only a solitary focal
mass.4 5 6 The multinodular form with hypervascular
enhancement pattern has not been reported before.
Currently there remains a lack of knowledge about
distinction of EM of liver from other hypervascular
liver tumours due to its rarity. Arterial phase imaging
is vital for lesion detection since some of the lesions
may be too small and too vaguely hypo-enhancing
to be detected during portovenous or delayed
phases. The differential diagnoses with multiple
hypervascular liver masses commonly include
multifocal HCC and hypervascular metastases. Its significance is underestimated, especially in areas
where HCC is endemic, such as Southeast Asia.
Clinicians and even radiologists may misdiagnose
these lesions as HCC, which is an imaging diagnosis,
and specific oncological treatment will be given
without histological confirmation of the lesion
leading to mismanagement. It is important to
bear in mind the possibility of myeloma of liver in
patients with known myeloma who present with
hypervascular mass on CT. We advocate a diagnostic
approach with emphasis on the use of multiphasic
cross-sectional studies including CT for detection,
and risk stratification (by alpha-foetal protein, and
hepatitis status). If these appear atypical of HCC or
EM involvement of liver, a timely biopsy to confirm
the diagnosis is recommended to avoid misdiagnosis
and subsequent mismanagement.
There are other points in the diagnostic
challenge posed by EM of the liver that influence
clinical management.
First, the variable sonographic appearance
of multinodular hepatic lesions, including target
appearance mimicking hepatic candidiasis, and
hypoechoic lesions raising a suspicion of pyogenic
abscesses, may lead to unnecessary antifungal or
antibacterial treatment.
Second, only one single large lesion was initially seen in our first case on multiphasic CT. This was
in concordance with multiple previous studies that
reported cases of EM of liver where lesions are more
conspicuous on ultrasound than on CT.3 Regarding
the hepatic lesions on CT from our cases, they were
most conspicuous on the arterial phase, while the
smaller ones may be isoattenuating or minimally
hypo-enhancing on portovenous or delayed phases.
In addition, most lesions had a subcapsular location
in the liver, an important area to review. Knowing that
this entity may be underdiagnosed, further studies are
needed to determine the most sensitive initial staging
modality to look for liver involvement. Based on our
cases, both ultrasound and multiphasic CT (including
arterial, portovenous, and 5-minute delayed) phases
play an important role in initial screening, subsequent
characterisation, and in guiding biopsy.
Conclusion
Extraosseous myeloma of the liver is a rare and under-recognised
entity associated with poorer prognosis
and survival. Imaging features are non-specific but
can mimic multifocal HCC on multiphase CT. We
advocate the use of multiphasic CT (including arterial
phase) for detection. The presence of hypervascular
liver masses in patients with known MM should alert
radiologists to this diagnosis. Definitive diagnosis
should be by tissue biopsy if there is a mismatch
between clinical risk factors and imaging, especially
in areas endemic for HCC.
Author contributions
Concept or design: HM Kwok, ES Lo.
Acquisition of data: HM Kwok, ES Lo.
Analysis or interpretation of data: All authors.
Drafting of the manuscript: HM Kwok, ES Lo.
Critical revision of the manuscript for important intellectual content: HM Kwok, ES Lo.
Acquisition of data: HM Kwok, ES Lo.
Analysis or interpretation of data: All authors.
Drafting of the manuscript: HM Kwok, ES Lo.
Critical revision of the manuscript for important intellectual content: HM Kwok, ES Lo.
All authors had full access to the data, contributed to the study, approved the final version for publication, and take responsibility for its accuracy and integrity.
Conflicts of interest
All authors have disclosed no conflicts of interest.
Acknowledgement
We would like to express our gratitude to the haematology and oncology physicians of Princess Margaret Hospital,
Hong Kong, for their professional patient care and invaluable
contribution to the understanding of a novel disease.
Declaration
Case 1 of the study was accepted as oral presentation in the 19th Asian Oceanian Congress of Radiology 2021, Malaysia.
Funding/support
This study received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethics approval
This study was approved by the Kowloon West Cluster Research Ethics Committee [Ref No.: KW/EX-21-054 (157-19)]. Patients were treated in accordance with the Declaration
of Helsinki, with informed consent provided for treatment,
procedures, and publication.
References
1. Oshima K, Kanda Y, Nannya Y, et al. Clinical and pathologic findings in 52 consecutively autopsied cases with multiple myeloma. Am J Hematol 2001;67:1-5. Crossref
2. Moulopoulos LA, Granfield CA, Dimopoulos MA, Kim EE, Alexanian R, Libshitz HI. Extraosseous multiple myeloma: imaging features. AJR Am J Roentgenol 1993;161:1083-7. Crossref
3. Philips S, Menias C, Vikram R, Sunnapwar A, Prasad SR.
Abdominal manifestations of extraosseous myeloma:
cross-sectional imaging spectrum. J Comput Assist
Tomogr 2012;36:207-12. Crossref
4. Cho R, Myers DT, Onwubiko IN, Williams TR. Extraosseous
multiple myeloma: imaging spectrum in the abdomen and
pelvis. Abdom Radiol (NY) 2021;46:1194-209. Crossref
5. Marcon M, Cereser L, Girometti R, Cataldi P, Volpetti S,
Bazzocchi M. Liver involvement by multiple myeloma
presenting as hypervascular focal lesions in a patient with
chronic hepatitis B infection. BJR Case Rep 2016;2:20150013. Crossref
6. Tan CH, Wang M, Fu WJ, Vikram R. Nodular
extramedullary multiple myeloma: hepatic involvement
presenting as hypervascular lesions on CT. Ann Acad Med
Singap 2011;40:329-31. Crossref

