© Hong Kong Academy of Medicine. CC BY-NC-ND 4.0
CASE REPORT
Gynaecological transurethral resection of the
prostate syndrome–induced acute pulmonary
oedema treated with high-dose nitro-glycerine:
a case report
SH Huang, MD1; SW Chang, MD2; AY Wang, MD3,4,5
1 Department of Surgery, Taipei Medical University Hospital, Taipei City, Taiwan
2 Division of Acute Care Surgery and Traumatology, Department of Surgery, Taipei Medical University Hospital, Taipei City, Taiwan
3 Department of Emergency Medicine, School of Medicine, College of Medicine, Taipei Medical University, Taipei City, Taiwan
4 Graduate Institute of Injury Prevention and Control, College of Public Health, Taipei Medical University, Taipei City, Taiwan
5 Department of Critical Care Medicine, Taipei Medical University Hospital, Taipei City, Taiwan
Corresponding author: Dr AY Wang (anyimilk@gmail.com)
Case report
A previously healthy 48-year-old woman developed
acute respiratory distress during operative
hysteroscopy for submucosal myomectomy. The
preoperative chest X-ray was normal (Fig a).
During surgery, facial oedema and acute dyspnoea
developed following intrauterine irrigation with
19 100 mL normal saline. Analysis of arterial blood
showed mixed metabolic acidosis and respiratory
acidosis with pH 6.989, PaCO2 57.8 mm Hg, and a
bicarbonate concentration of 14.0 mmol/L (Table).
Desaturation developed progressively and her SpO2
was 85% with O2 mask at a flow rate of 10 L/min. The
PaO2/FiO2 ratio was 121.5 mm Hg.
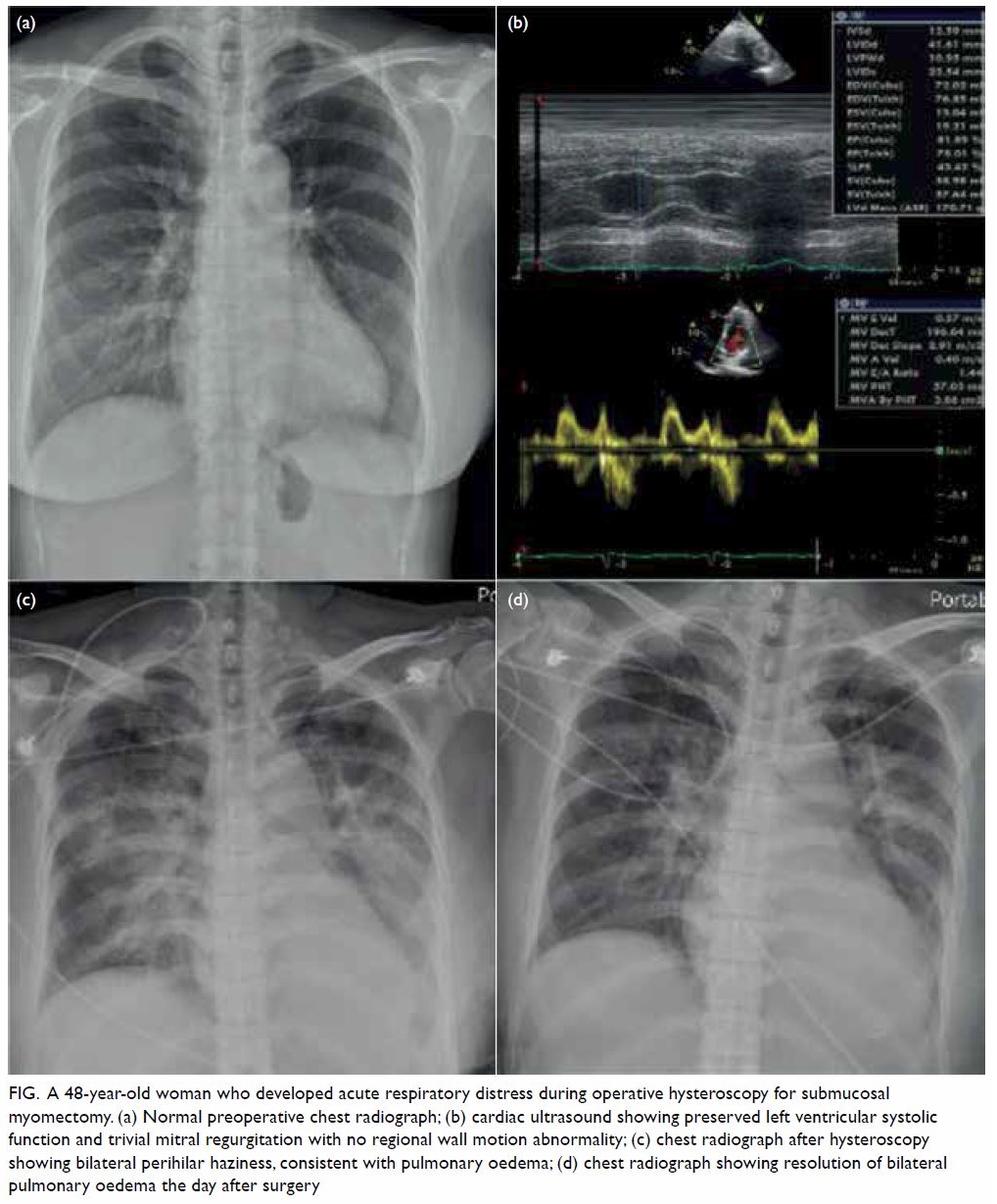
Figure. A 48-year-old woman who developed acute respiratory distress during operative hysteroscopy for submucosal myomectomy. (a) Normal preoperative chest radiograph; (b) cardiac ultrasound showing preserved left ventricular systolic function and trivial mitral regurgitation with no regional wall motion abnormality; (c) chest radiograph after hysteroscopy showing bilateral perihilar haziness, consistent with pulmonary oedema; (d) chest radiograph showing resolution of bilateral pulmonary oedema the day after surgery
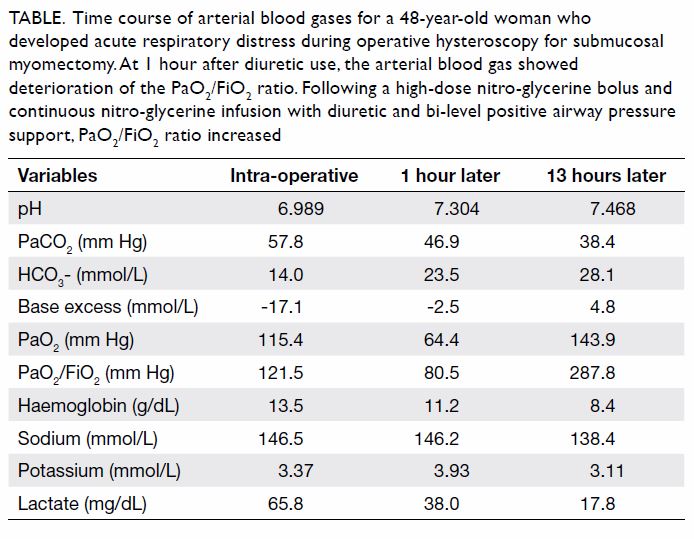
Table. Time course of arterial blood gases for a 48-year-old woman who developed acute respiratory distress during operative hysteroscopy for submucosal myomectomy. At 1 hour after diuretic use, the arterial blood gas showed deterioration of the PaO2/FiO2 ratio. Following a high-dose nitro-glycerine bolus and continuous nitro-glycerine infusion with diuretic and bi-level positive airway pressure support, PaO2/FiO2 ratio increased
On arrival in the intensive care unit, the patient
had a blood pressure of 160/90 mm Hg, heart rate
72 bpm, respiratory rate 30 breaths per minute, and SpO2 84%. Chest auscultation revealed bilateral
crackles and point-of-care ultrasonography showed a
bilateral diffuse B-line pattern consistent with acute
pulmonary oedema. Cardiac ultrasound revealed
preserved left ventricular systolic function (75.01%)
[Fig b]. There was neither regional wall motion
abnormality nor aortic stenosis. Non-invasive
positive pressure ventilation with bi-level positive
airway pressure was commenced to assist ventilation.
Subsequent chest X-ray showed acute pulmonary
oedema (Fig c) and intravenous diuretic (furosemide,
40 mg) was administered; nevertheless her PaO2/FiO2
ratio dropped to 80.5 mm Hg (Table). The overall
presentation was compatible with gynaecological
transurethral resection of the prostate (TURP)
syndrome–induced acute pulmonary oedema.
Intravenous high-dose 1 mg nitro-glycerine
(NTG) was administered followed by 1 mg NTG
bolus 5 minutes later and continuous infusion at a
rate of 1 mg/h. Transient hypotension was noted after
the high-dose NTG bolus. The patient’s respiratory
distress and oxygenation improved gradually and
arterial blood gas analysis revealed improvement
with pH 7.304, PaCO2 46.9 mm Hg, PaO2 64.4 mm Hg,
and lactate 38 mg/dL (Table). The next day, chest
X-ray revealed resolution of bilateral pulmonary
congestion (Fig d). The patient was maintained in a
negative fluid balance and fitted with a nasal cannula
that was well tolerated. She was transferred to the
general ward the next day and discharged from
hospital on the third day after surgery.
Hysteroscopic myomectomy is a surgical treatment
for submucosal myoma. During hysteroscopy,
continuous fluid irrigation is required to distend
the uterine cavity for optimal visualisation of the
operative field. However, when intrauterine pressure
exceeds mean arterial pressure, rapid intravasation via the venous sinuses may occur and absorption of
excessive irrigation fluid may cause hypervolaemia
and noncardiogenic pulmonary oedema. This
phenomenon is termed operative hysteroscopy
intravascular absorption syndrome with pathogenesis
similar to that of TURP syndrome; as such, operative
hysteroscopy intravascular absorption syndrome is
also known as gynaecological TURP syndrome.1
Irrigation with electrolyte-free fluid in
monopolar operative hysteroscopy may result in
dilutional hyponatraemia and hypo-osmolality.
Treatment of severe hyponatraemia-related
neurological symptoms such as convulsion or
cerebral oedema requires an infusion of 3%
hypertonic saline solution. Use of a glycine-based
solution can result in hyperammonaemia since ammonia is a by-product of glycine once absorbed
and metabolised in the liver. In bipolar operative
hysteroscopy, 0.9% normal saline is the commonly
used irrigant, so there is no significant procedure-related
hyponatraemia. Nonetheless in our patient,
severe metabolic acidosis developed due to rapid
absorption of chloride causing smaller plasma strong
ion difference, leading to an increase in hydrogen
ion, and therefore acidosis. There are no definitive
diagnostic criteria for gynaecological TURP
syndrome; the signs are varied and non-specific.
Clinical symptoms following a hysteroscopic
procedure in patients should be evaluated carefully
to exclude or diagnose gynaecological TURP
syndrome.
An intravenous NTG infusion is one of
the treatments for acute pulmonary oedema.
Nitro-glycerine produces nitric oxide that
increases the formation of cyclic guanosine
3’,-5’-monophosphate.2 The increased intracellular
cyclic guanosine 3’,-5’-monophosphate inhibits
the influx of calcium into cells thereby decreasing
intracellular calcium levels and causing vascular
smooth muscle relaxation. This vasodilator effect on
veins causes a decreased venous return, helps reduce
cardiac preload and reduces pulmonary capillary
hydrostatic pressure. The vasodilator effect on
arteries in turn reduces systemic vascular resistance,
helps reduce afterload and increases cardiac output.
The main effect of NTG is on fluid redistribution of
blood volume away from the lungs rather than on
fluid removal per se.
To treat acute heart failure, the initial dose
of NTG is 10 to 20 μg/min titrated to a total dose
up to 200 μg/min until the desired haemodynamic
effect is obtained.3 The main side-effects of NTG
are hypotension and headache. The major concern
of NTG therapy is resistance and tolerance under
continuous infusion. In a previous study, intravenous
bolus of high-dose NTG (2 mg) with repeated
administration every 3 minutes, up to a total of
10 doses in treatment of severe decompensated
heart failure, was associated with lower frequency
of endotracheal intubation and intensive care unit
admissions as well as few adverse events.4 The
contra-indications to high-dose NTG bolus are
severe aortic stenosis, hypertrophic obstructive
cardiomyopathy, hypotension, and concurrent use
of phosphodiesterase type 5 inhibitors.
Other strategies to treat acute pulmonary
oedema include supplemental oxygen and/or
ventilatory support, morphine, diuretics (eg, loop diuretic or thiazide), fluid restriction, inotropic agents
in cases of co-morbid hypotension or hypoperfusion,
and treatment of any underlying aetiologies.5
Non-invasive positive pressure ventilation improves
pulmonary compliance and reduces atelectasis. This
can reduce the work of breathing and decrease the
need for endotracheal intubation.
The effect of NTG is rapid with an immediate
peak effect and short half-life of 3 to 5 minutes. It
offers greater efficacy, safety, and a faster onset of
action than diuretics or morphine administered to
cause rapid improvement of pulmonary congestion.
In our case, the patient did not experience significant
improvement after diuretics so an intravenous bolus
of high-dose NTG was administered along with
bi-level positive airway pressure support. As a
result, while awaiting the fluid removal effect of
diuretic therapy, high-dose NTG helped improve the
respiratory distress and avoid an immediate need for
intubation.
Our case findings suggest that high-dose
NTG can be used to treat gynaecological TURP
syndrome–induced acute pulmonary oedema and
obviate the need for endotracheal intubation. As
far as we know, this is the first case report of the
application of high-dose NTG to treat gynaecological
TURP syndrome. The success in this case may offer
new insight for recommended treatment and further
research.
References
1. Jackson S, Lampe G. Operative hysteroscopy intravascular
absorption syndrome. West J Med 1995;162:53-4.
2. Ignarro LJ, Lippton H, Edwards JC, et al. Mechanism of
vascular smooth muscle relaxation by organic nitrates,
nitrites, nitroprusside and nitric oxide: evidence for the
involvement of S-nitrosothiols as active intermediates. J
Pharmacol Exp Ther 1981;218:739-49.
3. Ponikowski P, Voors AA, Anker SD, et al. 2016 ESC
Guidelines for the diagnosis and treatment of acute and
chronic heart failure: The Task Force for the diagnosis
and treatment of acute and chronic heart failure of the
European Society of Cardiology (ESC). Developed with
the special contribution of the Heart Failure Association
(HFA) of the ESC. Eur J Heart Fail 2016;18:891-975. Crossref
4. Levy P, Compton S, Welch R, et al. Treatment of severe
decompensated heart failure with high-dose intravenous
nitroglycerin: a feasibility and outcome analysis. Ann
Emerg Med 2007;50:144-52. Crossref
5. Wilson SS, Kwiatkowski GM, Millis SR, Purakal JD,
Mahajan AP, Levy PD. Use of nitroglycerin by bolus
prevents intensive care unit admission in patients with
acute hypertensive heart failure. Am J Emerg Med
2017;35:126-31. Crossref

