Hong Kong Med J 2018 Apr;24(2):152–7 | Epub 29 Mar 2018
DOI: 10.12809/hkmj176885
© Hong Kong Academy of Medicine. CC BY-NC-ND 4.0
ORIGINAL ARTICLE
Bacteriology and risk factors associated with
periprosthetic joint infection after primary total knee arthroplasty:
retrospective study of 2543 cases
KT Siu1; FY Ng2; PK Chan1;
Henry CH Fu1; CH Yan3; KY Chiu3
1 Department of Orthopaedics and
Traumatology, Queen Mary Hospital, Pokfulam, Hong Kong
2 Private practice, Hong Kong
3 Department of Orthopaedics and
Traumatology, The University of Hong Kong, Pokfulam, Hong Kong
Corresponding author: Prof KY Chiu (pkychiu@hkucc.hku.hk)
Abstract
Introduction: Periprosthetic
joint infection after total knee arthroplasty is a serious complication.
This study aimed to identify risk factors and bacteriological features
associated with periprosthetic joint infection after primary total knee
arthroplasty performed at a teaching hospital.
Methods: We reviewed 2543
elective primary total knee arthroplasties performed at our institution
from 1993 to 2013. Data were collected from the Hong Kong Hospital
Authority’s Clinical Data Analysis and Reporting System, the Infection
Control Team, and the joint replacement division registry. The
association between potential risk factors and periprosthetic joint
infection was examined by univariable analysis and multivariable
logistic regression. Univariable analyses were also performed to examine
the association between potential risk factors and bacteriology and
between potential risk factors, including bacteriology, and early-onset
infection.
Results: The incidence of
periprosthetic joint infection in our series was 1.34% (n=34). The
incidence of early-onset infection was 0.39% (n=24). Of the
periprosthetic joint infections, 29.4% were early-onset infections. In
both univariable and multivariable analyses, only rheumatoid arthritis
was a significant predictor of periprosthetic joint infection.
Methicillin-sensitive Staphylococcus aureus was the most common
causative organism. We did not identify any significant association
between potential risk factors and bacteriology. Periprosthetic joint
infection caused by skin flora was positively associated with
early-onset infection but the association was not statistically
significant.
Conclusion: The incidence of
periprosthetic joint infection after elective primary total knee
arthroplasty performed at our institution from 1993 to 2013 was 1.34%.
Rheumatoid arthritis was a significant risk factor for periprosthetic
joint infection.
New knowledge added by this study
- The incidence of periprosthetic joint infection after elective primary total knee arthroplasty performed at our institution from 1993 to 2013 was 1.34%.
- Rheumatoid arthritis was the only significant risk factor identified in the series.
- Early-onset infection may be associated with infection with skin flora. Therefore, in early-onset periprosthetic joint infection with negative cultures, an empirical antibiotic regimen should preferably provide adequate coverage against skin flora organisms.
Introduction
Periprosthetic joint infection (PJI) is an uncommon
but serious complication after total knee arthroplasty (TKA). Treatment is
often challenging and has a major impact on the patient. Multiple
operations are often required and patients may suffer from a long period
of disability. Moreover, PJI incurs considerable health care costs.1 2 3 Therefore, multiple strategies including antibiotic
prophylaxis, body exhaust systems, and laminar airflow systems have been
developed to reduce the incidence of PJI. Studies have also identified
modifiable risk factors for PJI after elective total joint replacement,4 5
6 7
8 9
10 11
12 13
14 with the aim of further
reducing the incidence of PJI. However, local data on the risk factors and
bacteriological features associated with PJI are still lacking.
This study had several aims. First, it aimed to
provide the most up-to-date local data on incidence of and risk factors
for PJI, including age, sex, presence of diabetes, presence of rheumatoid
arthritis, and one-stage bilateral TKA. Second, this study aimed to
provide an update on the bacteriology of PJI after elective primary TKA
and to examine the association between potential risk factors and
bacteriology. Third, we attempted to determine which risk factors,
including bacteriology, were more likely to be associated with early-onset
infection after elective primary TKA.
It is hoped that risk factors can be optimised or
modified to prevent infection after TKA. Furthermore, an improved
understanding of local bacteriological patterns and their relationship
with various risk factors can help guide antimicrobial therapy.
Methods
We reviewed 2543 elective primary TKAs performed at
the Queen Mary Hospital, Hong Kong, from 1993 to 2013. Data were collected
by an infection control nurse of the Department of Microbiology who was
blinded to the study objectives. The cohort data were collected from the
Hong Kong Hospital Authority’s Clinical Data Analysis and Reporting
System, the Infection Control Team, and the hospital’s joint replacement
division registry. The keywords used in the data search were
‘periprosthetic joint infection’, ‘total knee arthroplasty’, and ‘surgical
site infection’. Revision arthroplasties and knee arthroplasties for
malignant conditions were excluded from the study. In patients with a
history of native joint infection, elective primary TKA was performed only
after eradication of the infection. Patients with active bacteraemia were
also precluded from elective primary TKA until they were infection-free.
There were no cases of severe immunosuppression. In relation to infection
control, the majority of perioperative protocols for primary TKA were the
same throughout the study period. Preoperatively, intravenous antibiotic
prophylaxis (1 g of cefazolin) was given within 1 h before skin incision.
In patients with penicillin allergy, other antibiotics were prescribed as
appropriate. Intra-operatively, laminar airflow and body exhaust systems
were used. There was no routine use of antibiotic-loaded cement or
postoperative antibiotics. Postoperative wound management was the same
throughout the study period.
Cohort characteristics, occurrence of PJI, and
bacteriological data were retrieved. Bacterial type was defined as
infection with skin flora or non-skin flora. Skin flora included
methicillin-susceptible Staphylococcus aureus (MSSA),
methicillin-resistant S aureus (MRSA), methicillin-susceptible
coagulase-negative staphylococci (MSCNS), and methicillin-resistant
coagulase-negative staphylococci (MRCNS). Other organisms were considered
non-skin flora.
The following potential risk factors for PJI were
analysed: age, sex, presence of diabetes, presence of rheumatoid
arthritis, and one-stage bilateral TKA. They were examined by univariable
analyses and then multivariable logistic regression to identify potential
predictors of PJI, while controlling for confounders. We also studied the
association of those potential risk factors with bacteriology and with the
timing of infection onset; culture-negative PJI was excluded from these
analyses. According to a working party convened by the Musculoskeletal
Infection Society in 2014,15 PJI
that occurs within 90 days of the index operation is considered
early-onset infection, whereas PJI that occurs later is considered
late-onset infection.
Both univariable and multivariable logistic
regression in this study used the simultaneous entry method, with
covariates of age (as a continuous variable) and sex, diabetes, rheumatoid
arthritis, and one-stage bilateral TKA (as dichotomous variables).
Outcomes are presented as odds ratios (ORs) with 95% confidence intervals
(CIs). The regression model and data fitting were assessed using the
Hosmer–Lemeshow goodness-of-fit test, and diabetes and one-stage bilateral
TKA were excluded from the final model because of poor goodness-of-fit.
For associations between potential risk factors and bacteriology and
between potential risk factors and early onset of infection, only
univariable analyses were used owing to small numbers of events.
Categorical variables were compared with the chi-square test, whereas age
was compared with the independent t test (two-tailed).
Significance was assumed if P<0.05. All statistical analyses were
conducted using SPSS version 22.0 (IBM Corporation, Armonk [NY], United
States). The study was conducted in accordance with the principles
outlined in the Declaration of Helsinki.
Results
The incidence of PJI in our series was 1.34%
(n=34). The incidence of early-onset infection was 0.39% (n=10) and that
of late-onset infection was 0.94% (n=24). Among the cases PJI, 29.4% were
early-onset infection. Early-onset infection occurred within a median of
17 days after arthroplasty (interquartile range, 9-32 days). Late-onset
infection occurred within a median of 1 year and 8 months after
arthroplasty (interquartile range, 7 months to 2 years and 11 months).
Fifty-nine percent of infections occurred in the first year of surgery,
whereas 74% occurred in the first 2 years.
The mean (standard deviation) age was 69 (9) years,
with a range from 21 to 91 years; age followed a normal distribution.
Overall, PJI developed in 10 males (1.9%) and 24 females (1.2%). In the
one-stage bilateral TKA group, PJI occurred in 13 knees (1.2%). For the
single-side TKA group, 21 knees (1.4%) developed PJI. Nine patients with
diabetes (1.9%) and 25 patients without diabetes (1.2%) developed PJI. The
highest rate of PJI, at 3.1%, was found in patients with rheumatoid
arthritis, compared with 1.2% in patients without rheumatoid arthritis.
The descriptive data are summarised in Table 1.
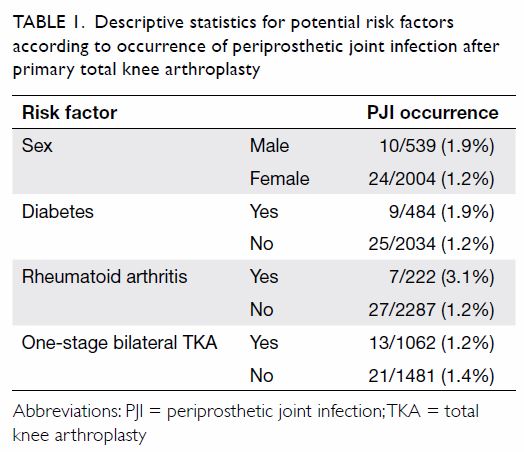
Table 1. Descriptive statistics for potential risk factors according to occurrence of periprosthetic joint infection after primary total knee arthroplasty
The most frequent causative organism was MSSA
(26.5%, n=9), followed by MRSA (17.6%, n=6), Streptococcus spp
(8.8%, n=3), MSCNS (5.9%, n=2), Escherichia coli (5.9%, n=2), Salmonella
(5.9%, n=2), MRCNS (2.9%, n=1) and Mycobacterium tuberculosis
(2.9%, n=1), The three cases of streptococcal infection comprised two Streptococcus
dysgalactiae infections and one Streptococcus agalactiae
infection. Culture-negative PJI comprised 23.5% of cases (n=8).
Methicillin-resistant strains constituted 39% of all staphylococcal
organisms. There was no significant association between the potential risk
factors and skin flora infection (Table 2).
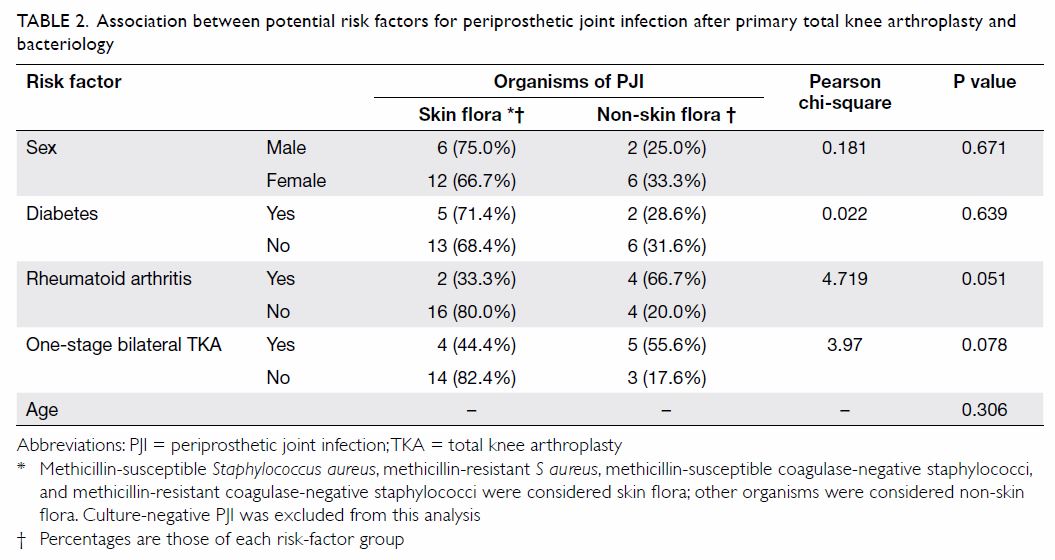
Table 2. Association between potential risk factors for periprosthetic joint infection after primary total knee arthroplasty and bacteriology
Rheumatoid arthritis was a significant risk factor
of PJI in the univariable analysis, with an OR of 2.67 (95% CI, 1.15-6.20;
P=0.02), as well as in the multivariable analysis, with an OR of 3.12 (CI,
1.29-7.56; P=0.01) [Table 3]. Being male (OR=1.9; P=0.11 in the
multivariable analysis) and having diabetes (OR=1.54; P=0.27 in the
univariable analysis) were not significantly associated with PJI.
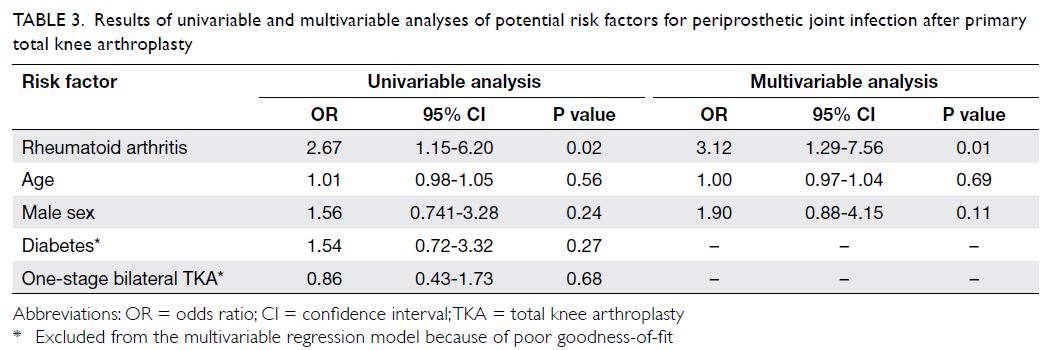
Table 3. Results of univariable and multivariable analyses of potential risk factors for periprosthetic joint infection after primary total knee arthroplasty
Age (P=0.655), sex (P=0.961), diabetes (P=0.462),
and rheumatoid arthritis (P=0.315) were not associated with early-onset
infection (Table 4). Infection caused by skin flora was
associated with early-onset infection (P=0.099), but the association was
not statistically significant.
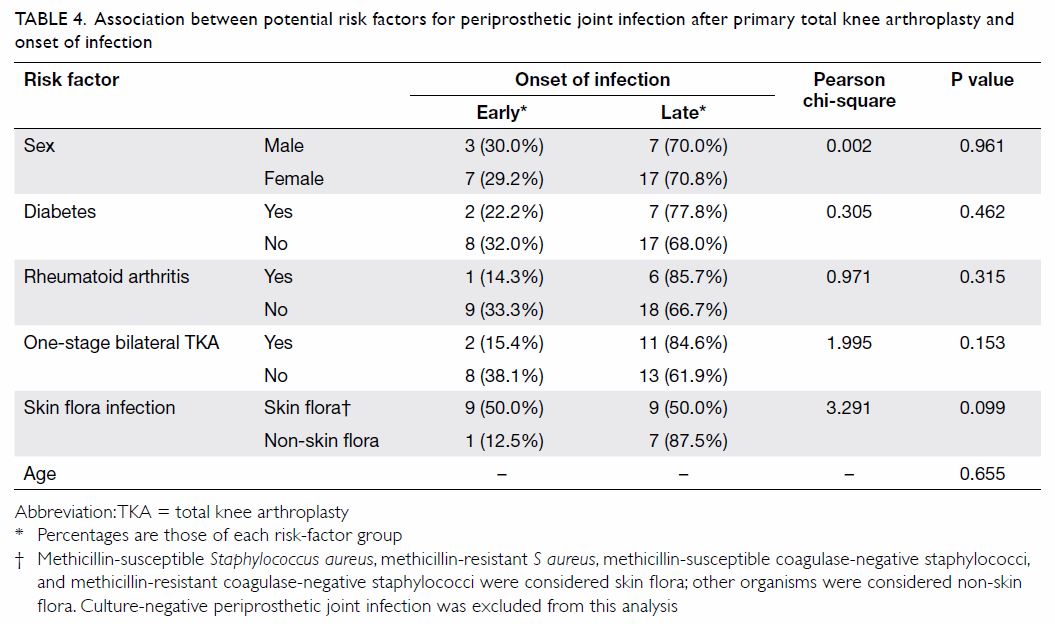
Table 4. Association between potential risk factors for periprosthetic joint infection after primary total knee arthroplasty and onset of infection
Discussion
In this study, the incidence of PJI after primary
TKA was 1.34% and the incidence of early-onset infection was 0.39%. The
majority of PJIs (70%) were late-onset infections. The reported incidence
of PJI after primary TKA ranges from 1.1% to 2.18%.16 17 18 Pulido et al16
reported the incidence of PJI after TKA to be 1.1%, of which 27% were
diagnosed during the first 30 days after arthroplasty, and a majority of
65% were diagnosed in the first year after surgery. In our study, the
average time to diagnosis was 431 days after the index surgery (range,
11-1699 days).
Rheumatoid arthritis was a significant risk factor
for PJI after primary TKA. This finding is in keeping with the current
literature.6 8 11 Although
various authors have found male sex to be a risk factor for PJI,4 19 20 the association was not significant in this study.
The OR of 1.9 may be of clinical importance but not significant as a
result of the small number of PJIs and inadequate statistical power. The
correlation between age and PJI has been a matter of controversy, with
some reports mentioning young age as a risk factor for PJI4 21 and some
otherwise.22 In our study, age was
not associated with PJI occurrence. For one-stage bilateral TKA, age has
been a controversial risk factor for PJI. Some studies16 23 have
suggested that one-stage bilateral TKA is associated with an increased
risk of superficial and deep infection. Hussain et al24 nonetheless reported a similar infection rate between
one- and two-stage bilateral TKA. Our study did not find an association
between one-stage bilateral TKA and PJI occurrence.
The local bacteriological pattern for PJI was
comparable to that reported in the literature.4
16 In our study, skin flora and
gram-positive bacteria were the most commonly isolated organisms, followed
by gram-negative bacteria such as Escherichia coli and Salmonella.
Coagulase-negative staphylococci were the most common causative organism
in one study.4 In contrast, in our
series, S aureus was the most common causative organism,
particularly methicillin-sensitive strains. Methicillin-resistant strains
were less common in our series, constituting 39% of all staphylococcal
organisms.
Other authors have reported that male sex is a risk
factor for PJI, which may be related to a sex difference in immune
response to pathogenic bacteria. Studies6 have shown that males (compared
with females) have a significantly higher likelihood of being a persistent
S aureus carrier. However, our study did not support male sex as a
risk factor for infection with skin flora. With regard to onset of
infection, PJI caused by skin flora was positively associated with
early-onset infection, although the association did not reach statistical
significance (P=0.099). Direct inoculation and spread from contiguous foci
of infection are more common in early-onset infection caused by wound
complications and local soft-tissue conditions. In contrast, distant foci
of infection, such as in bacteraemia, play a more important role in
late-onset infection. Therefore, in early-onset periprosthetic joint
infection with negative cultures, an empirical antibiotic regimen may
provide adequate coverage against skin flora organisms.
Fan et al20
reported 479 TKAs and rates of 1.9% for superficial wound infection, 0.2%
for early deep infection (n=1), and 0.6% for late deep infection (n=2).
Methicillin-sensitive S aureus and coagulase-negative
staphylococci were causative organisms. Lee et al25
reviewed 1133 primary TKAs and found a 0.71% incidence of PJI. The most
common causative organisms in descending order were methicillin-sensitive
S aureus, coagulase-negative staphylococci, methicillin-resistant S
aureus, and Pseudomonas aeruginosa. This finding is in
keeping with our data. Among risk factors identified by Lee et al25 were young age, diabetes, anaemia, thyroid disease,
heart disease, lung disease, and long operating time. However, the
researchers identified limitations of having only a small number of
patients with infection (n=8) and insufficient power for analysis. In
addition, multivariable analysis should have been performed to account for
the effect of confounders among the multiple risk factors. They also
reported the limitation that the mean follow-up duration was only 2 years.
A short follow-up period may underestimate the occurrence of late-onset
infection.
Our study has several limitations. The number of
PJI-positive cases was small and thus subgroup analysis was limited. This
study included subjects treated at a single centre in Hong Kong;
multicentre studies may improve the representativeness of local data. In
addition, perioperative management for elective TKA has evolved over the
past 20 years, including the introduction of an MRSA-screening programme
in 2011. In the screening programme, a nasal swab is taken from all
elective joint-replacement patients. Patients with a positive result are
prescribed 5 days of decolonisation therapy including a daily
chlorhexidine bath. Furthermore, intravenous vancomycin is now
administered for prophylaxis instead of cefazolin.26
There are many potential risk factors for PJI
documented in the literature. Nonetheless, only a limited number were
included in this study, most of which are not be modifiable. Thus, it may
not provide the necessary guidance for preoperative optimisation.
Furthermore, the exclusion of some potential risk factors may have led to
inadequate control for potential confounding factors. Inclusion of more
risk factors with better characterisation is needed to provide a more
comprehensive understanding and to better account for the confounding
effect of other variables.
Conclusion
The incidence of PJI after elective primary TKA in
our institution over two decades from 1993 to 2013 was 1.34%. Rheumatoid
arthritis was a significant risk factor for PJI in this series. In the
early-onset infection group, PJI was caused by skin flora, but this was
not statistically significant. It is hoped that this study has updated the
local data for PJI after primary TKA and serves as a model for future
related studies.
Acknowledgements
We thank colleagues from the Department of
Orthopaedics and Traumatology and the Infection Control Team at the Queen
Mary Hospital for their assistance in data collection, and those who
advised on this project to make its publication possible.
Declaration
The authors have no conflicts of interest to
disclose.
References
1. Kurtz SM, Lau E, Watson H, Schmier JK,
Parvizi J. Economic burden of periprosthetic joint infection in the United
States. J Arthroplasty 2012;27(8 Suppl):61-5.e1. Crossref
2. Lamarsalle L, Hunt B, Schauf M,
Szwarcensztein K, Valentine WJ. Evaluating the clinical and economic
burden of healthcare-associated infections during hospitalization for
surgery in France. Epidemiol Infect 2013;141:2473-82. Crossref
3. Nero DC, Lipp MJ, Callahan MA. The
financial impact of hospital-acquired conditions. J Health Care Finance
2012;38:40-9.
4. Crowe B, Payne A, Evangelista PJ, et al.
Risk factors for infection following total knee arthroplasty: a series of
3836 cases from one institution. J Arthroplasty 2015;30:2275-8. Crossref
5. Meller MM, Toossi N, Johanson NA,
Gonzalez MH, Son MS, Lau EC. Risk and cost of 90-day complications in
morbidly and superobese patients after total knee arthroplasty. J
Arthroplasty 2016;31:2091-8. Crossref
6. Zmistowski B, Alijanipour P. Risk
factors for periprosthetic joint infection. In: Springer BD, Parvizi J,
editors. Periprosthetic Joint Infection of the Hip and Knee. New York:
Springer; 2014: 15-40. Crossref
7. Jämsen E, Huhtala H, Puolakka T,
Moilanen T. Risk factors for infection after knee arthroplasty. A
register-based analysis of 43,149 cases. J Bone Joint Surg Am
2009;91:38-47. Crossref
8. Wilson MG, Kelley K, Thornhill TS.
Infection as a complication of total knee-replacement arthroplasty. Risk
factors and treatment in sixty-seven cases. J Bone Joint Surg Am
1990;72:878-83. Crossref
9. Namba RS, Inacio MC, Paxton EW. Risk
factors associated with deep surgical site infections after primary total
knee arthroplasty: an analysis of 56,216 knees. J Bone Joint Surg Am
2013;95:775-82. Crossref
10. Pruzansky JS, Bronson MJ, Grelsamer
RP, Strauss E, Moucha CS. Prevalence of modifiable surgical site infection
risk factors in hip and knee joint arthroplasty patients at an urban
academic hospital. J Arthroplasty 2014;29:272-6. Crossref
11. Chesney D, Sales J, Elton R, Brenkel
IJ. Infection after knee arthroplasty: a prospective study of 1509 cases.
J Arthroplasty 2008;23:355-9. Crossref
12. Moucha CS, Clyburn T, Evans RP,
Prokuski L. Modifiable risk factors for surgical site infection. J Bone
Joint Surg Am 2011;93:398-404.
13. Peersman G, Laskin R, Davis J,
Peterson M. Infection in total knee replacement: a retrospective review of
6489 total knee replacements. Clin Orthop Relat Res 2001;392:15-23. Crossref
14. Rasouli MR, Restrepo C, Maltenfort MG,
Purtill JJ, Parvizi J. Risk factors for surgical site infection following
total joint arthroplasty. J Bone Joint Surg Am 2014;96:e158. Crossref
15. Parvizi J, Gehrke T; International
Consensus Group on Periprosthetic Joint Infection. Definition of
periprosthetic joint infection. J Arthroplasty 2014;29:1331. Crossref
16. Pulido L, Ghanem E, Joshi A, Purtill
JJ, Parvizi J. Periprosthetic joint infection: the incidence, timing, and
predisposing factors. Clin Orthop Relat Res 2008;466:1710-5. Crossref
17. Tsaras G, Osmon DR, Mabry T, et al.
Incidence, secular trends, and outcomes of prosthetic joint infection: a
population based study, Olmsted County, Minnesota, 1969-2007. Infect
Control Hosp Epidemiol 2012;33:1207-12. Crossref
18. Tande AJ, Patel R. Prosthetic joint
infection. Clin Microbiol Rev 2014;27:302-45. Crossref
19. Herwaldt LA, Cullen JJ, French P, et
al. Preoperative risk factors for nasal carriage of Staphylococcus
aureus. Infect Control Hosp Epidemiol 2004;25:481-4. Crossref
20. Fan JC, Hung HH, Fung KY. Infection in
primary total knee replacement. Hong Kong Med J 2008;14:40-5.
21. Meehan JP, Danielsen B, Kim SH, Jamali
AA, White RH. Younger age is associated with a higher risk of early
periprosthetic joint infection and aseptic mechanical failure after total
knee arthroplasty. J Bone Joint Surg Am 2014;96:529-35. Crossref
22. Berbari EF, Osmon DR, Lahr B, et al.
The Mayo prosthetic joint infection risk score: implication for surgical
site infection reporting and risk stratification. Infect Control Hosp
Epidemiol 2012;33:774-81. Crossref
23. Luscombe JC, Theivendran K, Abudu A,
Carter SR. The relative safety of one-stage bilateral total knee
arthroplasty. Int Orthop 2009;33:101-4. Crossref
24. Hussain N, Chien T, Hussain F, et al.
Simultaneous versus staged bilateral total knee arthroplasty: a
meta-analysis evaluating mortality, peri-operative complications and
infection rates. HSS J 2013;9:50-9. Crossref
25. Lee QJ, Mak WP, Wong YC. Risk factors
for periprosthetic joint infection in total knee arthroplasty. J Orthop
Surg (Hong Kong) 2015;23:282-6. Crossref
26. Cheng VC, Tai JW, Wong ZS, et al.
Transmission of methicillin-resistant Staphylococcus aureus in the long
term care facilities in Hong Kong. BMC Infect Dis 2013;13:205. Crossref

