Hong Kong Med J 2017 Aug;23(4):381–6 | Epub 7 Jul 2017
DOI: 10.12809/hkmj166064
© Hong Kong Academy of Medicine. CC BY-NC-ND 4.0
ORIGINAL ARTICLE CME
Neonatal outcomes of preterm or very-low-birth-weight infants over a decade from Queen
Mary Hospital, Hong Kong: comparison with the
Vermont Oxford Network
YY Chee, FHKAM (Paediatrics); Mabel SC Wong, FHKAM (Paediatrics), FHKCPaed; Rosanna MS Wong, FHKAM (Paediatrics); KY Wong, FHKAM (Paediatrics)
Department of Paediatrics and Adolescent Medicine, Queen Mary Hospital,
Pokfulam, Hong Kong
Corresponding author: Dr YY Chee (yychee@hotmail.com)
Abstract
Introduction: There is a paucity of local data on
neonatal outcomes of preterm/very-low-birth-weight
infants in Hong Kong. This study aimed to
evaluate the survival rate on discharge and morbidity
of preterm/very-low-birth-weight infants (≤29+6
weeks and/or birth weight <1500 g) over a decade
at Queen Mary Hospital in Hong Kong, so as to
provide centre-specific data for prenatal counselling
and to benchmark these results against the Vermont
Oxford Network.
Methods: Standardised perinatal/neonatal data were
collected for infants with gestational age of 23+0
to 29+6 weeks and/or birth weight of <1500 g who were born
at Queen Mary Hospital between 1 January 2005 and
31 December 2014. These data were compared with
all neonatal centres in the Vermont Oxford Network
in 2013. The Chi squared test was used to compare
the categorical Queen Mary Hospital data with that
of Vermont Oxford Network. A two-tailed P value of
<0.05 was considered statistically significant.
Results: The overall survival rate on discharge from
Queen Mary Hospital for 449 infants was significantly higher than
that of the Vermont Oxford Network (87% versus
80%; P=0.0006). The morbidity-free survival at
Queen Mary Hospital (40%) was comparable with
the Vermont Oxford Network (44%). At Queen Mary
Hospital, 86% of infants had respiratory distress
syndrome, 40% bronchopulmonary dysplasia, 44%
patent ductus arteriosus, 7% severe intraventricular
haemorrhage, 5% necrotising enterocolitis, 10% severe retinopathy of prematurity, 10% late-onset sepsis, and
84% growth failure on discharge. Rates of respiratory
distress syndrome, intraventricular haemorrhage,
necrotising enterocolitis, and severe retinopathy of
prematurity were similar in the two populations.
At Queen Mary Hospital, significantly more infants
had bronchopulmonary dysplasia (P=0.011), patent
ductus arteriosus (P=0.015), and growth failure
(P=0.0001) compared with the Vermont Oxford
Network. In contrast, rate of late-onset sepsis was
significantly lower at Queen Mary Hospital than the
Vermont Oxford Network (P=0.0002).
Conclusions: Mortality rate and most of the
morbidity rates of our centre compare favourably
with international standards, but rates of
bronchopulmonary dysplasia and growth failure
are of concern. A regular benchmarking process
is crucial to audit any change in clinical outcomes
after implementation of a local quality improvement
project.
New knowledge added by this study
- Mortality rate and most of the morbidity rates at our centre compare favourably with international standards.
- Significantly more preterm/very-low-birth-weight infants had bronchopulmonary dysplasia and growth failure at our centre compared with the Vermont Oxford Network. Identification of these areas for improvement could facilitate the development of a quality improvement project.
- Local survival and morbidity rates provided by this study can be used for prenatal counselling about preterm delivery.
- The baseline data (growth failure rate upon discharge) obtained in the present study can be used to audit a future quality improvement project in the local community (standardised nutritional pathway for very-low-birth-weight infants).
Introduction
There has been a substantial improvement in the
outcomes for preterm infants over the past few
decades. This improvement reflects the advances made in antenatal, perinatal, and neonatal care. It is
crucial to audit and benchmark local data on clinical
outcomes with international standards.1 One of the
largest neonatal databases, the Vermont Oxford Network (VON), has been collecting and maintaining
data on very-low-birth-weight (VLBW) infants and
infants who fulfil other eligibility requirements from
all over the world since 1989 (website: https://public.vtoxford.org).2 Queen Mary Hospital (QMH) in
Hong Kong has been participating in the VON since
1998. Today, QMH is one of almost 1000 centres all
over the world participating in the network.3
There is a paucity of data reporting neonatal
outcomes for preterm/VLBW local infants. These
data are valuable in providing centre- and gestational
age (GA)–specific information that can be used
for parental counselling about high-risk infants,
and facilitate decision making by neonatologists,
obstetricians, and parents.
The primary aim of this study was to evaluate
the survival rate on discharge and morbidity in
preterm/VLBW infants (born between 23+0 and
29+6 weeks and/or birth weight of <1500 g) at
a tertiary perinatal centre in Hong Kong over a
decade. We also compared the neonatal outcomes
with the VON database, aiming to identify key areas
for quality improvement in the local community.
Methods
Study population and clinical outcomes
The study was conducted at QMH, Hong Kong,
which is a tertiary referral perinatal centre with an
annual delivery rate of approximately 3500 to 4500.
The hospital provides care for preterm infants who
are predominantly of Chinese ethnicity. This study was done in accordance with the principles outlined in the Declaration of Helsinki.
Perinatal/neonatal data of all infants born
alive at QMH with GA of 23+0 to 29+6 weeks and/or birth weight of <1500 g between 1 January 2005
and 31 December 2014 were collected. These infants
were part of the VON database. Infants born outside
of QMH were not included in this study. Data
were collected retrospectively from the Clinical
Management System and Clinical Information
System and entered into the VON database by
members of the QMH neonatal team.
Definitions for maternal and infant
characteristics were provided in the manual
“Nightingale Data Definitions” by the VON. In this
study, GA was determined as the best obstetric
estimate using ultrasonography and/or date of the
last menstrual period. If no antenatal data were
available, GA was estimated by postnatal neonatal
assessment. Intrauterine growth restriction, defined
as birth weight of <10th percentile for gender and
GA, was determined using growth charts published
by Fenton and Kim.4 Maternal obstetric data
included data on antenatal steroid use, presence of
chorioamnionitis, and mode of delivery. Antenatal
steroid therapy was considered to be given if it was
provided to the mother during pregnancy at any time
prior to delivery. Chorioamnionitis was diagnosed
on histopathological findings.
The survival rate was defined as neonates
who survived to the time of discharge. Surfactant
was used as early rescue therapy for infants with
respiratory distress syndrome (RDS), defined by
the presence of clinical and radiological features
within the first 24 hours of life. Conventional
mechanical ventilation use at any time was defined
as intermittent positive pressure ventilation through
an endotracheal tube with a conventional ventilator
at any time after leaving the delivery room. Rescue
postnatal steroids were used beyond 2 weeks of age
at our centre to facilitate extubation of ventilator-dependent
neonates with oxygen requirement
of >40% and significant radiological features of
persistent lung disease. Data on infants discharged
with oxygen were also collected.
Early-onset sepsis was defined as blood or
cerebrospinal fluid culture–positive bacterial sepsis
occurring within 72 hours of life. Haemodynamically
significant patent ductus arteriosus (PDA) on two-dimensional
echocardiography was treated with
intravenous ibuprofen (only data from 2010 to 2014
were collected as ibuprofen was used as medical treatment for PDA at QMH since 2010) or surgical
ligation if medical treatment was unsuccessful or
contra-indicated. Severe retinopathy of prematurity
was defined as stage III or above according to
international classification.5
The major morbidities included severe
neurological injury (defined as grade 3 or
4 intraventricular haemorrhage [IVH],
or periventricular leukomalacia [PVL]),
bronchopulmonary dysplasia (BPD; defined as
supplemental oxygen use at a postmenstrual age
[PMA] of 36 weeks), pneumothorax (defined as
extrapleural air diagnosed by chest radiograph or
needle aspiration), necrotising enterocolitis (NEC;
defined as stage ≥2 of Bell’s criteria), and late-onset
infection (defined as bacterial or fungal infection
after day 3 of life).
Data from QMH were compared with those of
all neonatal centres in the VON database in a single
year in 2013, the latest data available at the time of
the study.
Statistical analysis
The Chi squared test was used to compare the
categorical QMH data with that of VON. All P
values were two-tailed, and a P value of <0.05 was
considered statistically significant.
Results
Study group
A total of 449 infants with GA of 23+0 to 29+6
weeks and/or birth weight of 345 g to 1890 g who
were born at QMH between 1 January 2005 and 31
December 2014 were included in this study. This
study compared survival rate and morbidity of these
infants from QMH against 38 754 infants in the VON
database in 2013. A significantly greater proportion
of infants was delivered at 23 weeks of gestation in
the VON group compared with the QMH group,
which is contrary to the two groups at 27 and 29
weeks of gestation (P=0.0002). The proportion of
infants born at 24 to 26 weeks and 28 weeks was
similar for both groups (Table 1).
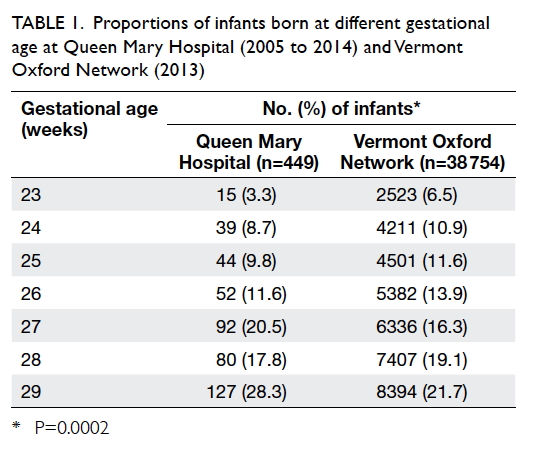
Table 1. Proportions of infants born at different gestational age at Queen Mary Hospital (2005 to 2014) and Vermont Oxford Network (2013)
Antenatal, maternal, and neonatal demographics
Overall, 31.0% of the cohort subjects at QMH were
multiple births (no difference between the QMH
and VON group; P=0.061). Fewer infants in the
QMH group were delivered by caesarean section
compared with the VON group (58.6% vs 68.2%;
P=0.0001). Antenatal steroids were given to 90.4%
of mothers in QMH; rate of prenatal steroid use
increased with increasing GA, from 73% at 23 weeks,
to 82%-89% between 24 and 25 weeks and 90%-96%
between 26 and 29 weeks. There was no significant
difference in prenatal steroid use between the two groups (P=0.065). Chorioamnionitis was confirmed
by placental histology in 24.3% of mothers at QMH,
which was significantly more than that in the VON
group (17.9%; P=0.0006).
Infants at QMH were predominantly born to Asian
(mainly Chinese) mothers whereas the majority of
the VON group were of White origin (P<0.001). In
QMH, 11.8% of the neonates were born small for
GA, similar to the VON group (9.2%, P=0.07) [Table 2].
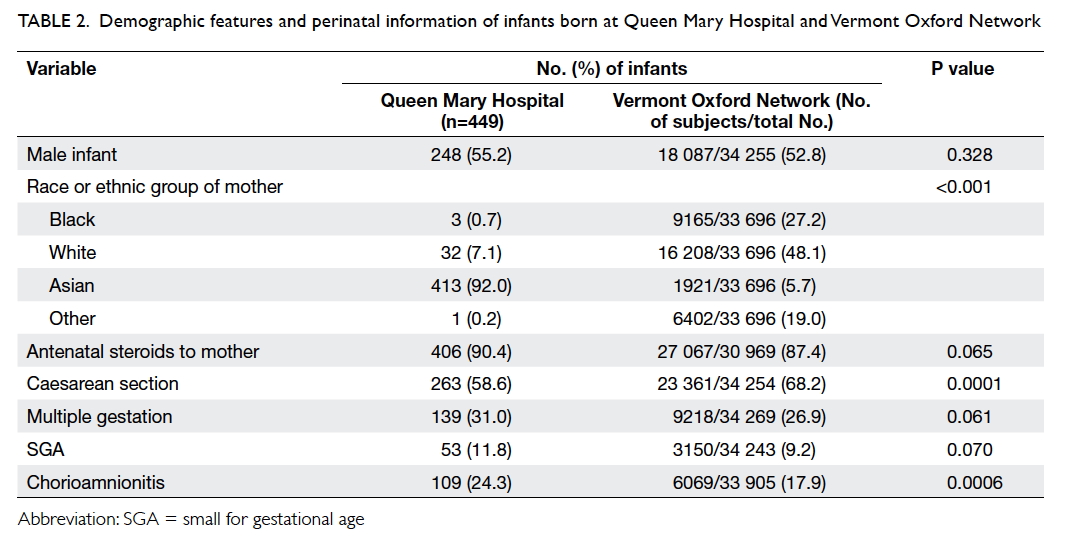
Table 2. Demographic features and perinatal information of infants born at Queen Mary Hospital and Vermont Oxford Network
In QMH, 78% of the infants were being
intubated for delivery room resuscitation. At 23
weeks, all infants underwent intubation in the
delivery room. Around half of the infants delivered
at 29 weeks and 20% at 28 weeks did not need
intubation in the delivery room, whereas 83%-97%
of infants born between 24 and 27 weeks needed
intubation at birth. Compared with infants with GA
of ≥26 weeks, the proportion of infants with GA
of <26 weeks with Apgar score of ≤3 at 1 minute was
higher. Only 16% of the infants were able to achieve
a target core temperature of 36.5°C to 37.5°C upon
admission to the neonatal intensive care unit (vs 51%
in the VON group; P<0.0001).
Survival rate on discharge and major
morbidities
In QMH, 87% of the 449 infants survived to discharge.
Rates of survival increased with increasing GA, from
27% at 23 weeks to 96% at 29 weeks. Overall survival
rate of the QMH group was significantly higher than
that in the VON group (87% vs 80%; P=0.0006).
In QMH, morbidity-free survival rate increased
from 0% at 23 weeks to 65% at 29 weeks (Table 3).
Survival without major morbidity was similar when
comparing the QMH and VON data (40% vs 44%;
P=0.105) [Table 3].
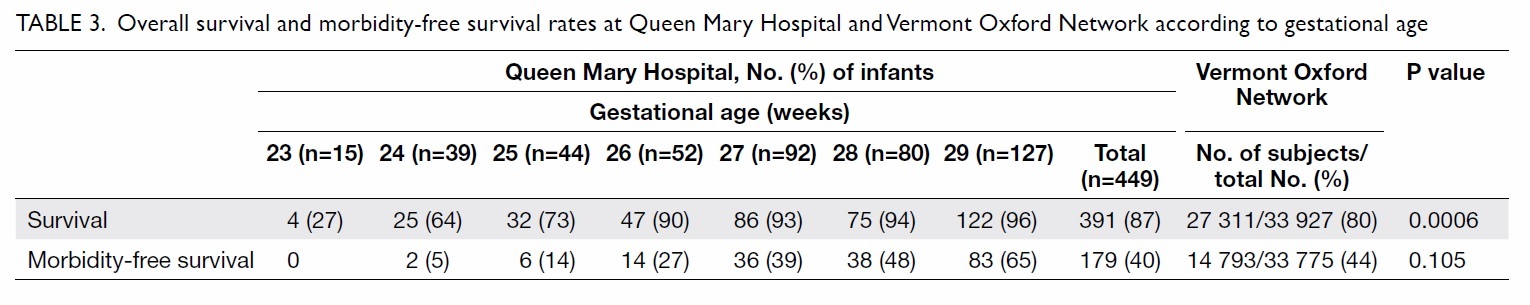
Table 3. Overall survival and morbidity-free survival rates at Queen Mary Hospital and Vermont Oxford Network according to gestational age
In QMH, 86% of infants experienced RDS and
83% needed surfactant therapy. Rate of mechanical
ventilation at any time decreased from 100% at 23 weeks to 69% at 29 weeks. There was no significant
difference in the incidence of RDS between QMH
and VON groups (P=0.808). Significantly more
infants in the QMH, however, were given surfactant
therapy for RDS and put on mechanical ventilation,
compared with VON (83% vs 74%; P=0.0001). The
overall frequency of pneumothorax was similar in
both groups (P=0.57).
Use of postnatal steroids for BPD was lower in
QMH compared with VON (3% vs 14%; P=0.0001)
although the QMH rate for BPD was higher (40%
vs 34%; P=0.011) and rate of home oxygen use was
lower (10% vs 18%; P=0.0001) [Table 4].
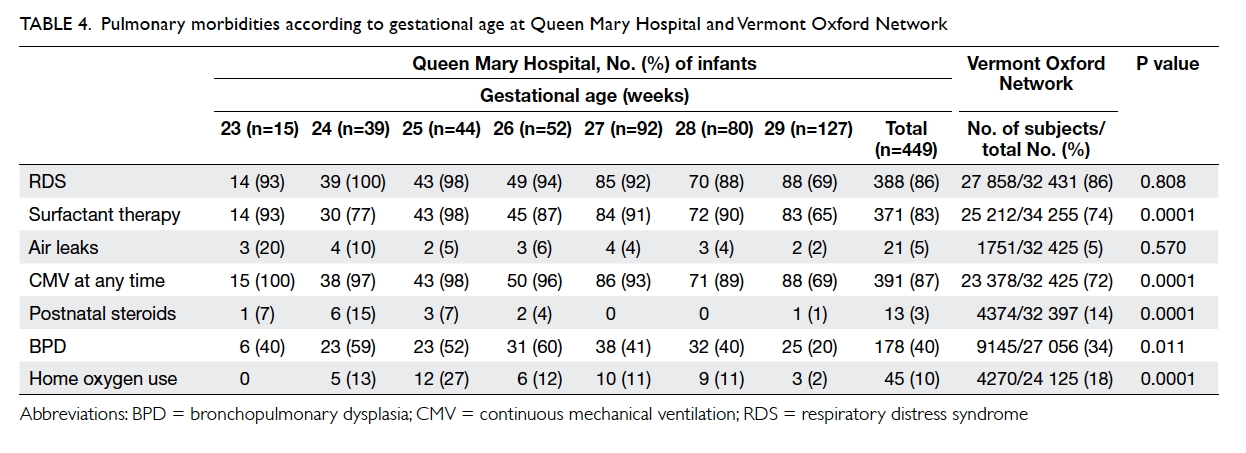
Table 4. Pulmonary morbidities according to gestational age at Queen Mary Hospital and Vermont Oxford Network
In QMH, rates of early- and late-onset sepsis
were 3% and 10%, respectively. No significant
difference was noted in the incidence of early-onset sepsis between QMH and VON (P=0.792) although
QMH had a significantly lower rate of late-onset
sepsis (10% vs 16%; P=0.0002) [Table 5].
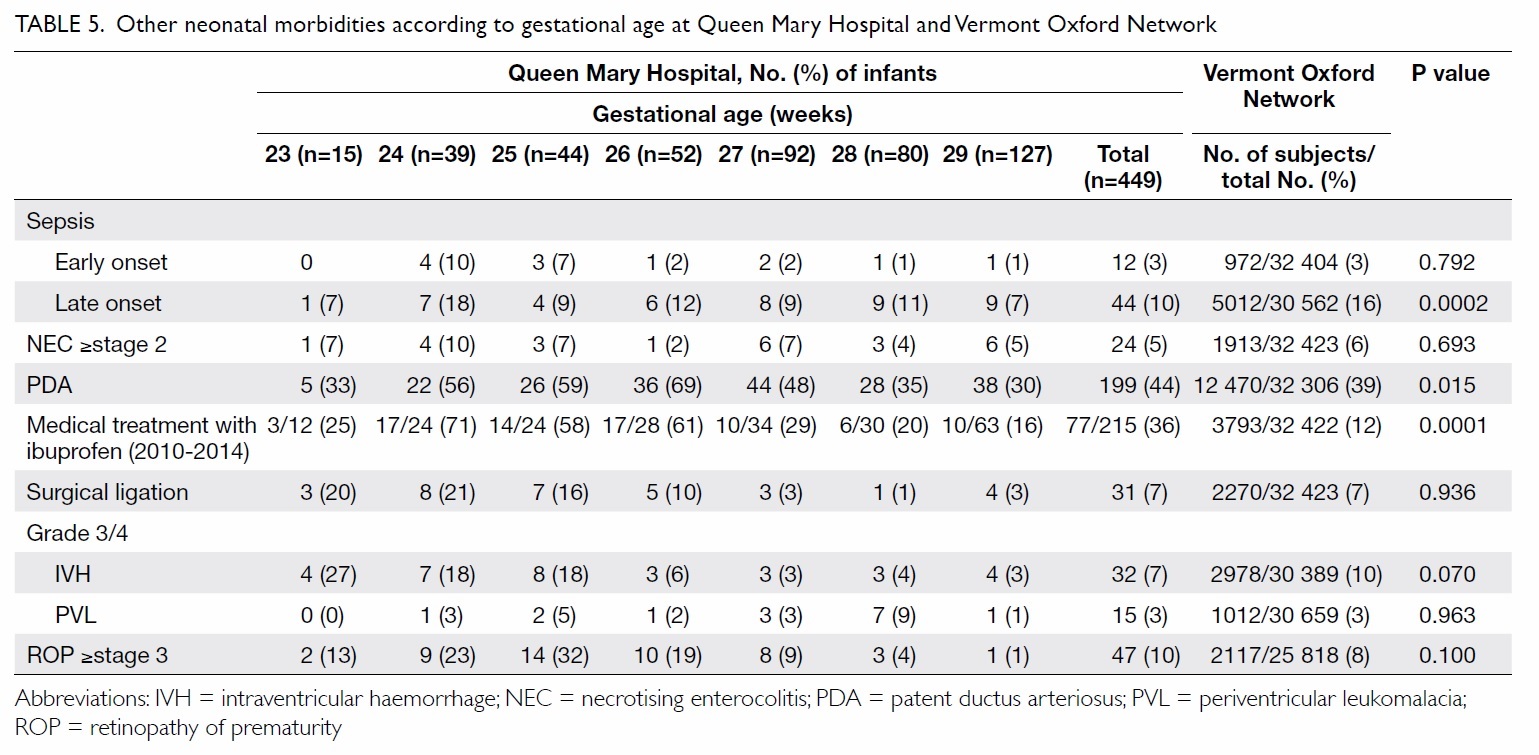
Table 5. Other neonatal morbidities according to gestational age at Queen Mary Hospital and Vermont Oxford Network
In QMH, NEC developed in 5% of infants
(≥stage 2) and 10% of infants were diagnosed as
having severe retinopathy of prematurity (≥stage
3). The overall frequencies of NEC and severe
retinopathy of prematurity were similar in the QMH
and VON groups (P=0.693 and P=0.100, respectively)
[Table 5].
In QMH, PDA was diagnosed in 44% of
infants, of whom 36% were treated with ibuprofen
(in 2010-2014), and 7% with surgical closure. For
management of PDA, QMH had higher rates of
ibuprofen treatment than the VON group (36% vs
12%; P=0.0001), whereas the rates of PDA ligation
were similar (P=0.936) [Table 5].
In QMH, 7% of sonograms indicated severe
IVH (grade 3 or 4); PVL was observed in 3% of
infants. There were no significant differences in the
incidence of severe IVH or PVL between QMH and
VON groups (P=0.070 and P=0.963, respectively;
Table 5).
In QMH, the length of hospital stay among
survivors decreased with increasing GA, from 19
weeks at GA 23 weeks to 8 weeks at GA of 29 weeks.
Similarly, PMA at discharge decreased from 42
weeks for surviving infants born at GA of 23 weeks,
to 41 weeks at GA of 24 weeks, 39 to 40 weeks at GA
of 25 to 27 weeks, 38 weeks at GA of 28 weeks, and
37 weeks at GA of 29 weeks.
Growth failure (body weight <10th centile for
age and sex) was evident in 84% of infants at QMH
(vs VON 42%; P=0.0001) upon discharge.
Discussion
This study reports the mortality and morbidity in
the neonatal intensive care unit in QMH over a
10-year period (2005-2014). As the only intensive
care centre from China participating in the VON,
QMH data provide an important source of local
epidemiological information. Data recorded at
QMH were compared with the entire VON database.
This allows the benchmarking of our neonatal
care and clinical outcomes for preterm infants
internationally. Local centre–specific information
about preterm infants based on GA facilitates
parental counselling about high-risk infants and
aids decision making.
Our study revealed that the survival rate
on discharge of preterm/VLBW infants (≤29+6
weeks and/or birth weight of <1500 g) from QMH
was higher than that from VON, with comparable
morbidity-free survival. The higher survival rate
on discharge from QMH may be partly explained
by the higher proportion of 23-week GA infants
in the VON (3.3% vs 6.5%; P=0.0002). Advances
in perinatal and neonatal care have contributed to
improved survival among preterm infants. The goal
in the care of these babies should be to improve
intact survival without morbidities. Among the
key morbidities (severe IVH, PVL, BPD, NEC,
pneumothorax, any late infection), BPD was the only
clinical entity with a significantly higher incidence at
QMH compared with VON (40% vs 34%; P=0.011).
Reducing the risk of BPD will have a great impact
on morbidity-free survival in our population.6 Of
note, BPD is a condition with multifactorial causes
and preterm infants are predisposed to lung injury including ventilator-induced lung injury, infection,
and inflammation.7 8 9 The higher rate of BPD in QMH
compared with VON could be related to the higher
prevalence of chorioamnionitis, a known risk factor
that inhibits alveolar development.10 As we were
ventilating more infants than VON (as evidenced
by the higher rate of surfactant use for RDS and
use of conventional mechanical ventilation at any
time), the risk of ventilator-associated lung injury
was increased. Haemodynamically significant PDA
was present in a greater proportion of infants at
QMH compared with VON (44% vs 39%; P=0.015);
PDA increases pulmonary blood flow and causes
interstitial oedema. The mechanical ventilator
setting and oxygen requirement increase as a result
and provide a foundation for BPD. The risk of BPD
is also influenced by growth restriction. Growth
failure upon discharge was present in 84% of our
infants. With poor nutrition in these infants, normal
lung growth, maturation, and repair are inhibited.
Postnatal steroid was used less frequently in QMH
compared with VON (3% vs 14%), and may explain
in part the decreased incidence of BPD in VON.
In order to reduce the incidence of BPD in
our population, modifiable risk factors need to be
reduced. In recent years, our unit has been managing
RDS more with continuous positive airway pressure
support with subsequent selective surfactant
administration—that is, INSURE (INtubation,
SURfactant administration, then Extubation)—in
order to avoid unnecessary or prolonged ventilation,
thereby reducing ventilator-associated lung injury
and BPD.11 12 Targeted oxygen saturation has also
been used in our centre to minimise oxygen toxicity
associated with BPD.13 14
We had a significantly higher proportion of
infants who were small for GA upon discharge
compared with the VON group (84% vs 42%;
P=0.0001). Apart from affecting lung growth and
maturation, postnatal growth failure is associated
with poor long-term neurocognitive outcome.15 16
One possible explanation for growth failure in our
population is the lack of guidelines about preterm
infant nutrition (eg timing of feeding initiation and
milk volume advancement, prescription of total
parenteral nutrition etc). Without such guidelines,
a preterm infant’s caloric intake may be suboptimal
with consequent compromise of growth. In order
to improve the growth of our preterm infants, a
standardised nutritional pathway for the VLBW
infants has been in use since 2015. Its effect has yet
to be evaluated.
A limitation of our study is the lack of adjustment
for potential confounding factors. The two groups,
QMH and VON, were not directly comparable, for
instance, the higher survival rate of preterm infants
in our centre could be partly affected by the lower
proportion of GA of 23 weeks in our study population.
Conclusions
The majority of neonatal outcomes for preterm/VLBW infants at QMH were comparable with VON, with the exception of BPD and growth
failure upon discharge. Regular auditing and
benchmarking of clinical outcomes will help ensure
quality improvement with implementation of new
interventions and projects in our unit.
Declaration
All authors have disclosed no conflicts of interest.
References
1. Horbar JD, Plsek PE, Leahy K; NIC/Q 2000. NIC/Q 2000:
establishing habits for improvement in neonatal intensive
care units. Pediatrics 2003;111(4 Pt 2):e397-410.
2. Horbar JD. The Vermont-Oxford Neonatal Network:
integrating research and clinical practice to improve the
quality of medical care. Semin Perinatol 1995;19:124-31. Crossref
3. Annual report for infants born
in 2014. Center 320. Vermont Oxford Network; 2015.
4. Fenton TR, Kim JH. A systematic review and meta-analysis
to revise the Fenton growth chart for preterm infants.
BMC Pediatr 2013;13:59. Crossref
5. An international classification of retinopathy of prematurity.
The Committee for the Classification of Retinopathy of
Prematurity. Arch Ophthalmol 1984;102:1130-4. Crossref
6. Greenough A, Ahmed N. Perinatal prevention of
bronchopulmonary dysplasia. J Perinat Med 2013;41:119-26. Crossref
7. Jobe AH, Ikegami M. Mechanisms initiating lung injury in
the preterm. Early Hum Dev 1998;53:81-94. Crossref
8. Kallapur SG, Jobe AH. Contribution of inflammation
to lung injury and development. Arch Dis Child Fetal
Neonatal Ed 2006;91:F132-5. Crossref
9. Kinsella JP, Greenough A, Abman SH. Bronchopulmonary
dysplasia. Lancet 2006;367:1421-31. Crossref
10. Thomas W, Speer CP. Chorioamnionitis is essential in the
evolution of bronchopulmonary dysplasia—the case in
favour. Paediatr Respir Rev 2014;15:49-52. Crossref
11. Morley CJ, Davis PG, Doyle LW, et el. Nasal CPAP or
intubation at birth for very preterm infants. N Engl J Med
2008;358:700-8. Crossref
12. Committee on Fetus and Newborn; American Academy of
Pediatrics. Respiratory support in preterm infants at birth.
Pediatrics 2014;133:171-4. Crossref
13. Supplemental Therapeutic Oxygen for Prethreshold
Retinopathy of Prematurity (STOP-ROP), a randomized,
controlled trial. I: primary outcomes. Pediatrics
2000;105:295-310. Crossref
14. Askie LM, Henderson-Smart DJ, Irwig L, Simpson JM.
Oxygen-saturation targets and outcomes in extremely
preterm infants. N Engl J Med 2003;349:959-67. Crossref
15. Ehrenkranz RA, Dusick AM, Vohr BR, Wright LL,
Wrage LA, Poole WK. Growth in the neonatal intensive
care unit influences neurodevelopmental and growth
outcomes of extremely low birth weight infants. Pediatrics
2006;117:1253-61. Crossref
16. Franz AR, Pohlandt F, Bode H, et al. Intrauterine,
early neonatal, and postdischarge growth and
neurodevelopmental outcome at 5.4 years in extremely
preterm infants after intensive neonatal nutritional
support. Pediatrics 2009;123:e101-9. Crossref

