Hong Kong Med J 2016 Oct;22(5):454–63 | Epub 12 Aug 2016
DOI: 10.12809/hkmj154806
© Hong Kong Academy of Medicine. CC BY-NC-ND 4.0
ORIGINAL ARTICLE
Population-based survey of the prevalence of lower urinary tract symptoms in adolescents with and without psychotropic substance abuse
YH Tam, FHKAM (Surgery)1;
CF Ng, FHKAM (Surgery)2;
YS Wong, FHKAM (Surgery)1;
Kristine KY Pang, FHKAM (Surgery)1;
YL Hong, MSc1;
WM Lee, MSc2;
PT Lai, BN2
1 Division of Paediatric Surgery and Paediatric Urology, Department of Surgery, Prince of Wales Hospital, The Chinese University of Hong Kong, Shatin, Hong Kong
2 Division of Urology, Department of Surgery, Prince of Wales Hospital, The Chinese University of Hong Kong, Shatin, Hong Kong
Corresponding author: Dr YH Tam (pyhtam@surgery.cuhk.edu.hk)
Abstract
Objective: To investigate the prevalence of lower
urinary tract symptoms in adolescents and the
effects of psychotropic substance use.
Methods: This was a population-based cross-sectional
survey using a validated questionnaire
in students from 45 secondary schools in Hong
Kong randomly selected over the period of January
2012 to January 2014. A total of 11 938 secondary
school students (response rate, 74.6%) completed
and returned a questionnaire that was eligible for
analysis. Individual lower urinary tract symptoms
and history of psychotropic substance abuse were
documented.
Results: In this study, 11 617 non-substance abusers
were regarded as control subjects and 321 (2.7%)
were psychotropic substance users. Among the
control subjects, 2106 (18.5%) had experienced at
least one lower urinary tract symptom with urinary
frequency being the most prevalent symptom (10.2%).
Females had more daytime urinary incontinence (P<0.001)
and males had more voiding symptoms (P=0.01). Prevalence of lower urinary tract symptoms
increased with age from 13.9% to 25.8% towards
young adulthood and age of ≥18 years (P<0.001). Among the substance users, ketamine
was most commonly abused. Substance users had
significantly more lower urinary tract symptoms
than control subjects (P<0.001). In multivariate
analysis, increasing age and psychotropic substance
abuse increased the odds for lower urinary tract
symptoms. Non-ketamine substance users and
ketamine users were respectively 2.8-fold (95%
confidence interval, 2.0-3.9) and 6.2-fold (4.1-9.1)
more likely than control subjects to develop lower
urinary tract symptoms. Females (odds ratio=9.9;
95% confidence interval, 5.4-18.2) were more likely
to develop lower urinary tract symptoms than males
(4.2; 2.5-7.1) when ketamine was abused.
Conclusions: Lower urinary tract symptoms are
prevalent in the general adolescent population. It is
important to obtain an accurate history regarding
psychotropic substance use when treating teenagers
with lower urinary tract symptoms.
New knowledge added by this study
- Prevalence of lower urinary tract symptoms (LUTS) increases consistently from onset of adolescence towards adulthood. Psychotropic substance abuse, particularly ketamine, is associated with an increased risk of developing LUTS in adolescents. Girls are more susceptible than boys if ketamine is abused.
- It is important to obtain an accurate history regarding psychotropic substance use when treating teenagers with LUTS.
Introduction
Lower urinary tract symptoms (LUTSs) are
prevalent worldwide. An estimated 45.2% of the 2008
worldwide population aged ≥20 years are affected by
at least one LUTS.1 Large-scale population-based
survey has reported that LUTS prevalence increases
with advancing age up to 60% at the age of 60 years.2
Evaluation and treatment of LUTS for the general
population have incurred significant costs to the
health care system. In children, the association
of LUTS with urinary tract infection, persistent
vesicoureteric reflux, renal scarring, and constipation
have drawn substantial attention over the years.3 4 Among various LUTSs, urinary incontinence (UI)
has been most extensively investigated in children
with the reported prevalence varying from 1.8% to
20%.5 Previous studies of the prevalence of individual
LUTS using the International Children’s Continence
Society (ICCS) definitions6 have focused primarily
on pre-adolescent children in primary schools.7 8 9 10
To date, no large-scale studies have investigated the
prevalence of LUTSs in adolescents.
Psychotropic substance use among adolescents
is a growing concern worldwide and creates
psychosocial, security, and health care issues. In
recent years, ketamine abuse has been found to
cause severe LUTSs and Hong Kong is one of the
earliest countries/regions to report the newly established
clinical entity of ketamine-associated uropathy.11 12 13
Ketamine is the most popular psychotropic
substance being abused by people aged <21 years
in our society.14 The aim of the present study was
to investigate the prevalence of LUTSs in our
adolescents and the differences between those with
and without psychotropic substance use.
Methods
Study design, sample size estimation, and participant recruitment
This was a cross-sectional questionnaire survey
that recruited adolescents from secondary schools
serving Hong Kong local residents during the
period of January 2012 to January 2014. There were
almost 500 secondary schools in Hong Kong serving
approximately 470 000 adolescents in 2009/10. Based
on the data of children and young adults available in
the literature,2 9 we assumed the prevalence of LUTSs among our adolescents to be 20%. A study sample
of 6050 participants would be required to allow an
error of ±1%. Government sources suggested 2.3%
of our secondary school students used psychotropic
substance in 2011/12.15 We assumed the prevalence
of LUTSs among those secondary students using
psychotropic substance was 15% higher than in
normal subjects. In order to detect a difference with
a type 1 error of 0.05 and a power of 0.8, a sample
size of 4500 participants would be required. Based
on the above two assumptions and a predicted
response rate of 50% to 60%, we determined that a
potential target of not less than 10 000 participants
would be required.
In the selection of schools we included all
government, aided, and Direct Subsidy Scheme
schools. Private international schools and special
schools were excluded. Co-educational, boys’, and
girls’ schools were included. The list of secondary
schools was provided by the Education Bureau and
schools were grouped into 18 geographical districts.
As the prevalence of psychotropic substance use
might vary significantly between schools, we
arbitrarily determined to recruit participants from
not less than 8% to 10% of the secondary schools in
order to reduce the sampling bias.
The random selection process started with
drawing a district followed by a school within the
selected district. Based on a rough estimation of
population distribution, we intended to select schools
from Hong Kong Island (HKI), Kowloon (Kln), and
New Territories (NT) in an approximate ratio of
1:2:3. We invited the selected schools to participate
in the study. If the invitation was declined, the next
school following the drawing sequence would be
contacted. The above procedure was repeated until
the target sampling size was reached. Finally, 45 out
of 121 schools were selected and approached, and
agreed to participate in the study (HKI, n=7; Kln,
n=13; NT, n= 25) giving a potential target of 16 000
participants.
The grades/classes of students participating
in the survey from each school were not randomly
selected but were determined after discussion and
mutual agreement with the school management.
In order to avoid the possible bias of intentional
selection or exclusion of a particular class of
students, school management was invited to express
their preferences about which grade/grades of
students would participate provided that all students
of the selected grade/grades participated. Although
we tried to avoid over-representation of a particular
grade of students by making some suggestions to the
school management, their preferences were always
respected and accepted. Of the 45 participating
schools, we recruited two or three grades of Form
1-3 students, two or three grades of Form 4-6
students, and all the students in 18, 10, and 8 schools, respectively. In the remaining nine schools,
we recruited only one grade of their students.
Study measures
The measuring tool was an anonymous self-reported
questionnaire accompanied by an information sheet.
In both the information sheet and the questionnaire,
we stated clearly that participation in the study was
voluntary and consent to participate was presumed
on receipt of a completed questionnaire that was
returned in the envelope provided. Individuals who
did not consent to participate were told to disregard
the questionnaire. The questionnaire consisted of
three parts: demographic data on gender and age,
LUTS assessment, and history of psychotropic
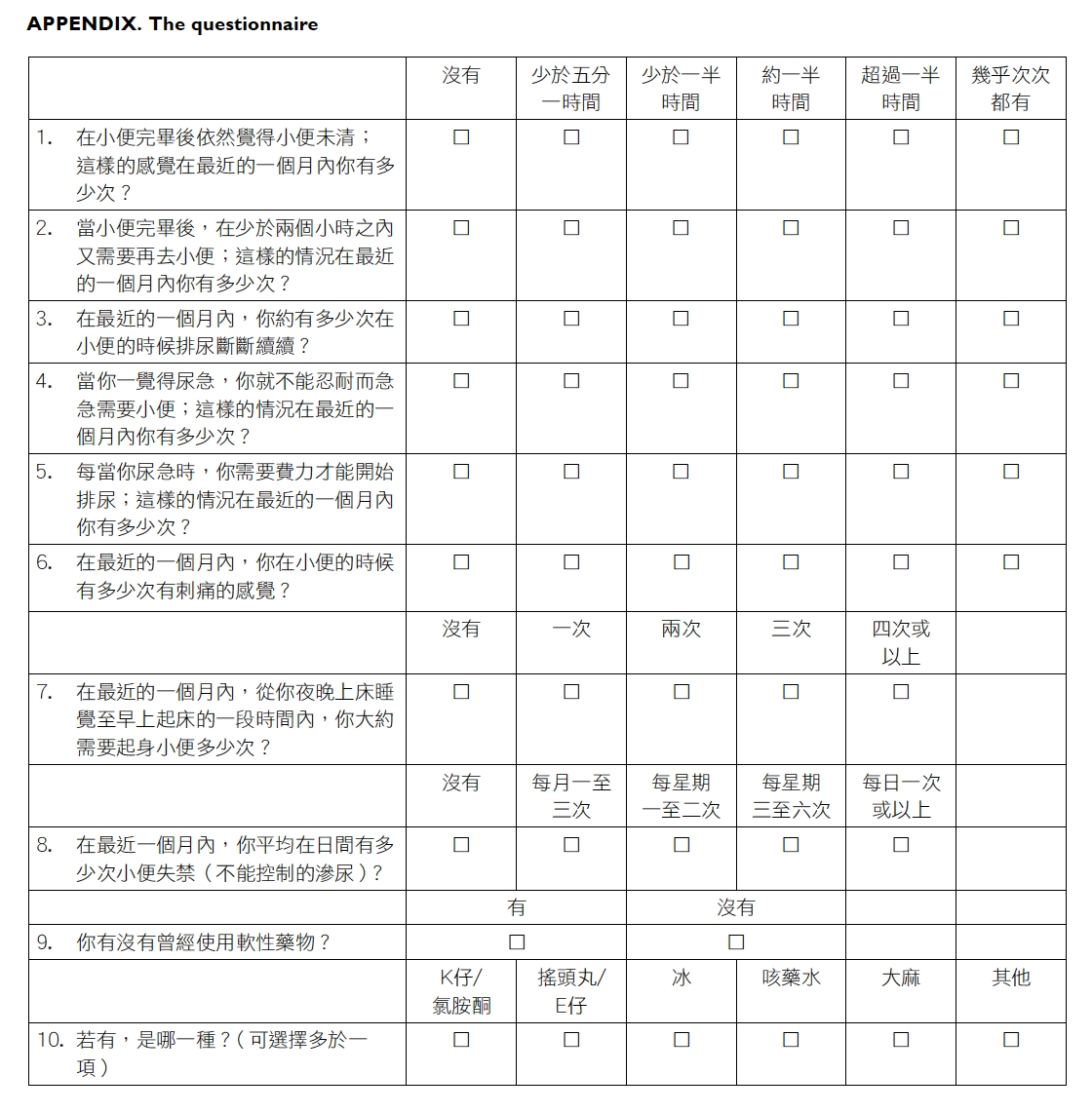
substance use (Appendix).
Age was divided into four categories: <13,
13-15, 16-17, and ≥18 years. Participants were asked to
respond to an 8-item LUTS assessment that included
storage symptoms (urinary frequency, urgency,
nocturia, and daytime UI), voiding symptoms
(intermittent stream, straining, and dysuria), and
post-micturition symptom (incomplete emptying).
The recall period was the last 4 weeks. The LUTS
questions were adapted from the Hong Kong
Chinese version of International Prostate Symptom
Score questionnaire that has been validated to
assess LUTSs in our local adult population.16 We
believed that the level of comprehension of most of
our adolescent participants in secondary education
was close to that of an average adult. The response
options for most of the LUTSs were on a 6-point
Likert scale: “never”, “seldom (<20% of the time)”,
“sometimes (20-50% of the time)”, “often (50% of the
time)”, “always (>50% of the time)”, and “almost every
time”. Any LUTS with frequency threshold of ‘≥20%
of the time’ was defined as being present in the study
subject. Daytime UI and nocturia were assessed on
a different 5-point Likert scale according to their
frequency. Daytime UI and nocturia were defined
as present if the study subject had ≥1 to 3 times per
month and ≥2 times per night, respectively.2 17
Responses to questions on psychotropic
substance use were dichotomised as either “yes” or
“no”. Those with positive responses were directed
to questions on the type of substance being abused,
which included ketamine, ecstasy, methamphetamine,
cough mixture, marijuana, and others. Participants
were allowed to indicate more than one substance.
According to the response to questions on
psychotropic substance use, the participants were
classified as control subjects or psychotropic
substance users. The psychotropic substance users
were further subdivided into either ketamine users
or non-ketamine users.
Statistical analysis
The responses to each LUTS were dichotomised
as “present” versus “absent” and prevalence rate
for each LUTS was expressed in percentage with
95% confidence interval (CI). Missing data were
excluded for analysis. Chi squared and trend tests
were performed in univariate analysis to compare
prevalence differences between groups divided
by gender, age, and psychotropic substance use.
Using the outcome of “at least one LUTS”, which
was dichotomised into “yes” or “no”, a binary logistic
regression model using enter method was set up
to investigate risk factors including gender, age,
and psychotropic substance use. Odds ratio (OR)
of “at least one LUTS” was estimated with 95% CI
for the potential risk factors. A P value of <0.05 was
considered to be significant.
The study protocol was approved by the Joint
CUHK-NTEC Clinical Research Ethics Committee.
Results
A total of 16 000 questionnaires were sent to schools
and 11 938 were returned (estimated response rate,
74.6%) that were eligible for analysis in the study.
The response rate was estimated since the number
of questionnaires delivered to each school was
not necessarily equal to the number of students
of that school who received the questionnaire.
The conduction of the survey at schools was not
supervised. We were uncertain if students absent
from school would receive our questionnaire. The
number of questionnaires requested by each school
was always rounded off to the nearest 10 and not
necessarily equal to the actual number of students
in the selected classes. It seems logical to assume
that the actual number of students who received our
questionnaires was less than 16 000 and the actual
response rate might be higher. There were similar
numbers of males (n=6040) and females (n=5819)
among the participants who responded to the
question on gender. Among the 11 938 participants,
11 617 did not report use of any psychotropic
substances and were defined as control subjects;
321 (2.7%) participants reported to have used one or
more types of psychotropic substance were defined
as substance users.
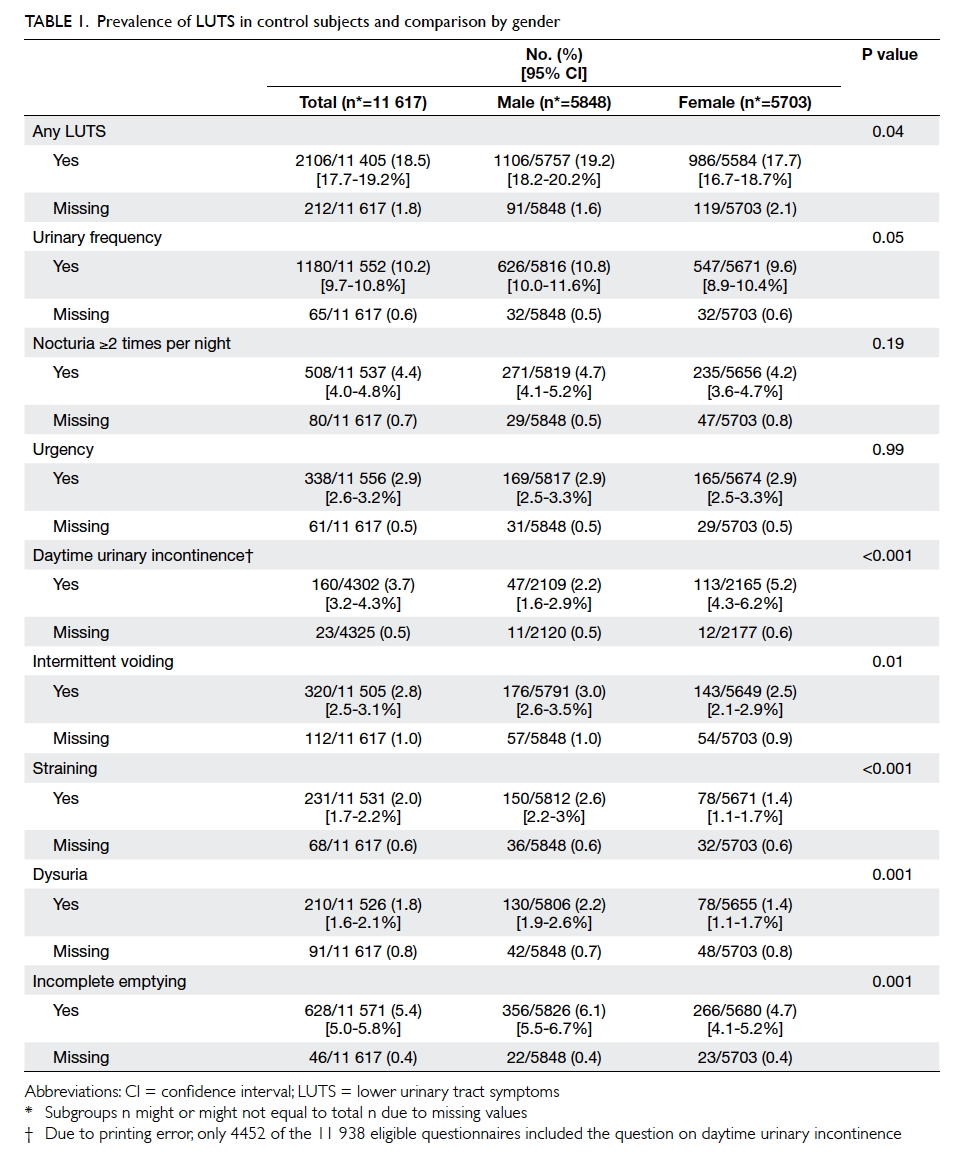
Of 11 617 control subjects, 2106 (18.5%;
including only the valid subjects) had experienced at
least one LUTS with the symptom frequency of ‘≥20%
of the time’ in the last 4 weeks (Table 1). The most
prevalent LUTSs were urinary frequency (10.2%),
incomplete emptying (5.4%), and nocturia ≥2 times
per night (4.4%). Daytime UI ≥1 to 3 times
per month was reported by 3.7% of control subjects.
Females had more daytime UI than males (5.2% vs
2.2%; P<0.001), while males had significantly more
voiding symptoms and incomplete emptying. There
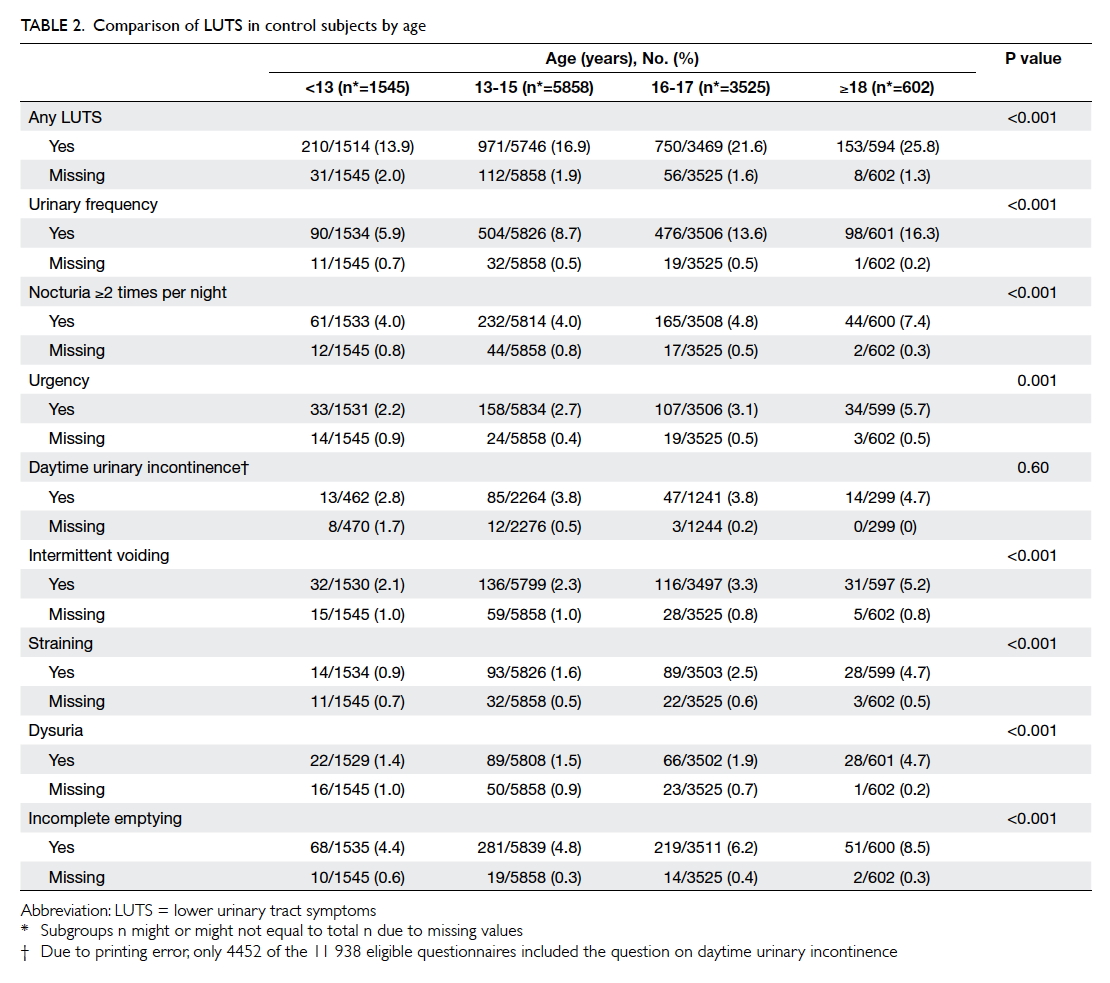
was significant increase in the prevalence of all LUTSs except for
daytime UI across the age-groups from <13 years to
the young adulthood age-group of ≥18 years (Table 2).
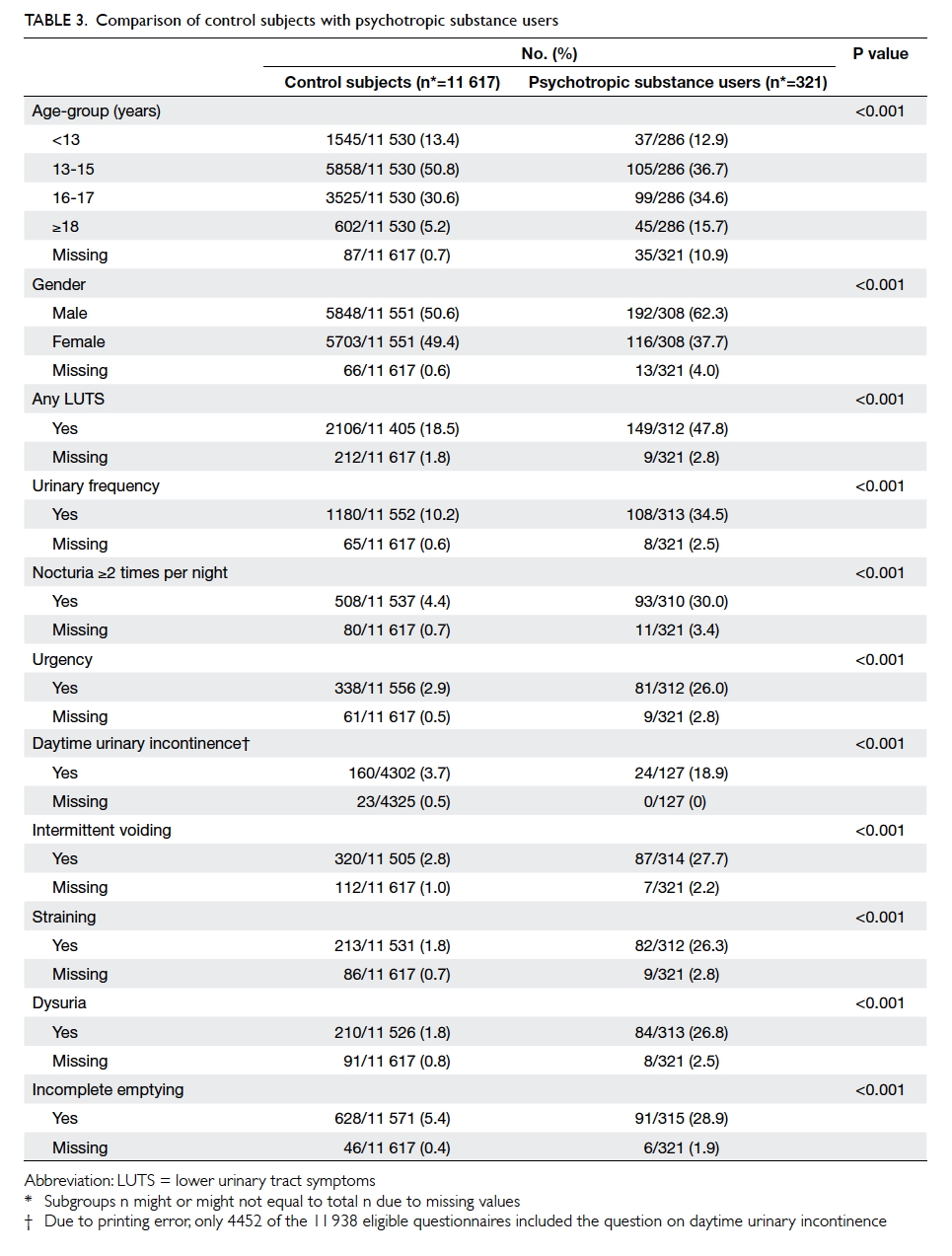
Compared with control subjects, the
psychotropic substance users experienced
significantly more LUTSs in all areas (Table 3). Of
the 321 substance abusers, 305 responded to the
question about types of psychotropic substance
abused. Ketamine was the most commonly abused
substance (n=139; 45.6%), followed by cough
mixture (n=96; 31.5%), ecstasy (n=77; 25.2%),
methamphetamine (n=76; 24.9%), and marijuana
(n=70; 23.0%). Of ketamine users, 60.7% had at
least one LUTS. Comparing the ketamine users
with other non-ketamine substance users, the
former experienced significantly more
LUTSs in all areas except for daytime UI, though for which
a higher prevalence was still observed. Female ketamine users appeared to be more affected by LUTS than males
(Table 4).
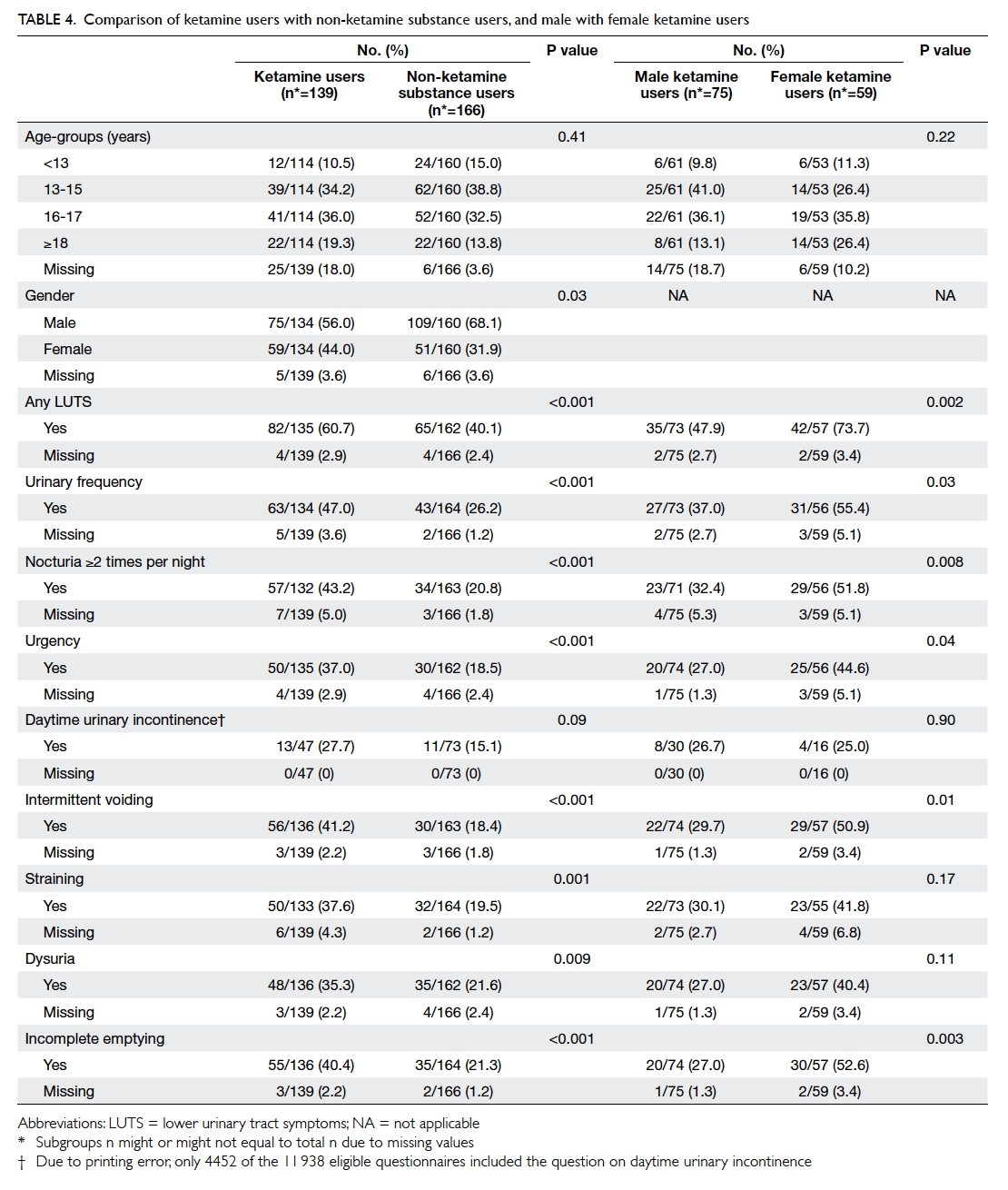
Table 4. Comparison of ketamine users with non-ketamine substance users, and male with female ketamine users
In multivariate analysis, increasing age and
psychotropic substance use were found to increase
the odds for experiencing at least one LUTS. With
reference to age of <13 years, the ORs of experiencing
at least one LUTS at age 13-15, 16-17, and ≥18 years
were 1.3 (95% CI, 1.1-1.5), 1.7 (95% CI, 1.4-2.0), and
2.1 (95% CI, 1.7-2.7), respectively. With reference to
the control subjects, the ORs of experiencing at least
one LUTS were 2.8 (95% CI, 2.0-3.9) for those who
used substances other than ketamine, and 6.2 (95%
CI, 4.1-9.1) for those who used ketamine. When
assessing the two genders separately in multivariate
analysis, female ketamine users were 9.9-fold (95%
CI, 5.4-18.2) and male ketamine users were 4.2-fold
(95% CI, 2.5-7.1) more likely than their non-abuser
counterparts to develop LUTSs.
Discussion
Large-scale population-based surveys of LUTS
prevalence have been conducted in adults.2 17 Recently a few paediatric studies using the ICCS
definitions have reported LUTS prevalence in
children varying from 9.3% to 46.4%.7 8 9 The wide
variation in prevalence can be attributed to the
differences in the study population, questions
used to assess LUTS, and the criteria to define
the presence of symptoms. Vaz et al8 reported a
prevalence of 21.8% in 739 Brazilian children aged
6 to 12 years while Yüksel et al7 found 9.3% of their
4016 Turkish children aged 6 to 15 years had LUTSs.
In both studies, the investigators used validated
scoring systems for a combination of LUTSs being
assessed and pre-determined cut-off points in the
total scores to define the presence or absence of
LUTS.7 8 In contrast, Chung et al9 investigated 16 516
Korean children aged 5 to 13 years by measuring the
presence of individual LUTS and reported the highest
prevalence of 46.4% experiencing at least one LUTS.
The high prevalence rate in the Korean study can be
partly explained by their methodology wherein the
responses to the LUTS questions were dichotomised
into “yes” or “no” and a positive symptom was
defined without considering its frequency.9
To the best of our knowledge, the present
study is the first large-scale prevalence study focused
on adolescents. We used a similar methodology to
other major adult studies to measure each LUTS
individually and define its presence by a frequency
threshold of ‘≥20% of the time’.2 17 18 19 We agree with
others that using a scoring system to define LUTS in
a prevalence study may not reflect the true impact
of individual LUTS as it is possible that a highly
prevalent symptom may happen alone and the
summed score may not reach the threshold.17
In our adolescents without any substance
abuse, 18.5% experienced at least one LUTS. Our
finding suggests that LUTS prevalence in adolescents
appears to be lower than that in young adults.
Previous studies including two conducted in Chinese
populations have reported that 17% to 42% of men
and women aged 18 to 39 years experience at least
one LUTS.2 18 19 Notably, LUTS prevalence increased
with age during adolescence from 13.9% in those <13
years to 25.8% in those aged ≥18 years in this study.
In children, the prevalence of LUTS peaks at age 5
to 7 years and then declines with increasing age up
to 13 to 14 years.7 8 9 10 20 The decline in prevalence has been attributed to the maturation of urinary bladder
function along with the growth and development of
children. Our study is the first to provide evidence
that LUTS prevalence rises from the trough at the
onset of adolescence and continues to increase
throughout adolescence into adulthood. Our
reported prevalence of 25.8% in those participants
aged ≥18 years is in agreement with the trend in
young adults reported elsewhere.2 18 19
Little is known in the existing literature
regarding the trend of LUTS prevalence from
adolescence to adulthood. In a Finnish study of 594
subjects aged 4 to 26 years, the authors reported that
individuals aged 18 to 26 years had more urgency
than the other two age-groups of 8 to 12 years and
13 to 17 years.10 Although adolescence spans less
than a decade, it is unique with rapid physical,
psychological, and developmental changes. Reasons
for the increase in LUTS prevalence from adolescence
to young adulthood are largely unknown but likely
to be multifactorial. Changes in lifestyle, altered
micturition behaviour, habitual postponement of
micturition, unhealthy bowel habits, attitudes to
the use of school toilets, anxiety associated with
academic expectations, or worsening of relationships
with family may all contribute to newly developed
LUTS during adolescence. Further studies are
warranted to investigate this phenomenon.
Our findings that storage symptoms were more
prevalent than voiding symptoms are in agreement
with the reported results in young adults.2 18 19
Urinary frequency (10.2%) and nocturia ≥2 times
per night (4.4%) were the two most prevalent storage
symptoms among the control subjects. We
agree with others that nocturia once per night is
very common in the general population and using
the threshold of nocturia ≥2 times per night as LUTS
is more appropriate.2 17 18 19 Only 2.9% of our control
subjects had urgency suggestive of overactive bladder
(OAB) according to ICCS definitions,6 in contrast to
12% of Korean children aged 13 years.20 Children
with OAB may have urinary frequency in addition
to urgency. The much lower prevalence of urgency
than urinary frequency in our study suggests that
many of our study subjects had urinary frequency
unrelated to OAB. Glassberg et al21 found over
70% of their paediatric patients with dysfunctional
voiding (DV) and primary bladder neck dysfunction
(PBND) experienced urinary frequency; DV and
PBND are also associated with high residual urine
volume. Our finding that the feeling of incomplete
emptying (5.4%) was the second most prevalent
LUTS suggests that in some participants urinary
frequency was secondary to incomplete bladder
emptying associated with DV or PBND.
In our study, male non-substance users
experienced more voiding symptoms while females
had more daytime UI. Literature has consistently
found female gender to be a risk factor for daytime
UI in children.5 22 23 Our finding suggests that the
gender association with daytime UI extends from
childhood to adolescence. There are inconsistencies
in the paediatric literature with respect to gender
differences in voiding symptoms. Kyrklund et al10
found more voiding symptoms in boys than girls
only in the age-group of 4 to 7 years, while such
difference was not noted by others.8 24
Psychotropic substance use increased the risk
of LUTS in our adolescents. Notably, 60% of our
adolescents who abused ketamine had experienced at
least one LUTS and had high prevalence rates of 28%
to 47% in all areas of LUTS. Our finding that 2.7% of
our participants abused psychotropic substances is
consistent with the latest figure of 2.3% estimated by
our government in its survey conducted in 2011/12.15
Ketamine-associated uropathy has emerged as a
new clinical entity in our society since 2007.13 This
chemically induced cystitis as a result of the urinary
metabolites of ketamine is associated with severe
LUTS with the possible consequence of irreversible
bladder damage.12 25 Little information is available in the medical literature about the prevalence of LUTS
among ketamine users. An online survey conducted
in the UK reported a prevalence of 26.6% of at
least one LUTS in the last 12 months among 1285
participants who had illicitly used ketamine.26 The
LUTS prevalence is likely influenced by variation
in dose and frequency of ketamine use of the study
population. We have recently reported that both
the dose and frequency of ketamine use and female
gender are associated with the severity of the LUTS
at presentation among the young patients who
sought urological treatment for ketamine-associated
uropathy.25 In the present study, female ketamine
users were at a higher risk of developing LUTS than
males. This observation is in agreement with our
previous findings and our postulation that females
appear to be more susceptible to the chemically
induced injury following illicit use of ketamine for
unknown reasons.25
Non-ketamine substance users also experienced
more LUTSs than the control subjects in this
study although the prevalence was not as high as that
of ketamine users. Most recently Korean investigators
have reported a 77% prevalence rate of LUTS among
a group of young methamphetamine (also known
as ‘ice’) users, and suggested that a pathological
dopaminergic mechanism plays a predominant role
in methamphetamine-associated LUTS.27 There
has been a rising trend of using methamphetamine
in recent years and it is now the second most
popular psychotropic substance abused by youths
aged <21 years in our community.14 It would not be
surprising if we encountered more and more young
patients presenting with LUTS associated with
methamphetamine use in the foreseeable future.
Limitations of this study
There was potential bias in the sampling process as
almost two thirds of the schools that we selected
and approached refused to participate, the grades
of the participants were not randomly selected, and
non-response rate was approximately 20%. Young
participants of lower grades may not be able to
comprehend the LUTS questions that were designed
for adults. Nevertheless, our finding of 2.7% of
psychotropic substance use appears to be consistent
with the 2.3% reported by the 2011/12 government
survey in over 80 000 secondary school students.15
We did not study other potential risk factors that
may be associated with LUTS in adolescents such
as bowel function, urinary tract infection, stressful
events, lifestyle, and toilet environment. The 0.5%
to 2% missing data in each of the LUTS questions,
though small, may still affect the estimated
prevalence of each LUTS among our control subjects.
Although daytime UI was not a prevalent symptom,
the fact that less than half of the participants were
asked this question because of a printing error may
underestimate the overall prevalence of experiencing
at least one LUTS among different subgroups. The
4-week recall period only allowed crude assessment
of LUTS. A more-prevalent symptom may not
necessarily cause more inconvenience than a less-prevalent
symptom. How each individual LUTS
concerned the participant and how different the
substance abusers and non-substance abusers were
concerned by the LUTS were not investigated in this
study. Therefore individuals, particularly the non-substance
abusers, who reported the experience of
LUTS did not necessarily suffer from any established
lower urinary tract conditions that warranted
medical attention. The dose and frequency of illicit
psychotropic substance use would certainly have an
impact on the prevalence of LUTS but this was not
investigated in this survey.
Despite all these limitations, our study provides
important data on the prevalence of LUTS in
adolescents and the effect of psychotropic substance
use. The LUTSs are prevalent in the general
adolescent population. It is important for clinicians
to obtain a history about psychotropic substance
use when treating teenagers with LUTS as there is
a substantial possibility that the LUTSs are caused
by organic pathology associated with psychotropic
substance use and not functional voiding disorders.
Appendix
Additional material related to this article can be
found on the HKMJ website. Please go to <http://www.hkmj.org>, and search for the article.
Declaration
The study was supported by the Beat Drugs Fund
(BDF101012) of the Hong Kong SAR Government.
The funding source had no role in the study design,
data collection, data analysis, results interpretation,
writing of the manuscript, or the decision to submit
the manuscript for publication. All authors have no
conflicts of interest relevant to this article to disclose.
References
1. Irwin DE, Kopp ZS, Agatep B, Milsom I, Abrams P.
Worldwide prevalence estimates of lower urinary tract
symptoms, overactive bladder, urinary incontinence and
bladder outlet obstruction. BJU Int 2011;108:1132-8. Crossref
2. Irwin DE, Milsom I, Hunskaar S, et al. Population-based
survey of urinary incontinence, overactive bladder, and
other lower urinary tract symptoms in five countries:
results of the EPIC study. Eur Urol 2006;50:1306-14;
discussion 1314-5. Crossref
3. Koff AS, Wagner TT, Jayanthi VR. The relationship
among dysfunctional elimination syndromes, primary
vesicoureteral reflux and urinary tract infections in
children. J Urol 1998;160:1019-22. Crossref
4. Leonardo CR, Filgueiras MF, Vasconcelos MM, et al. Risk
factors for renal scarring in children and adolescents
with lower urinary tract dysfunction. Pediatr Nephrol
2007;22:1891-6. Crossref
5. Sureshkumar P, Jones M, Cumming R, Craig J. A population
based study of 2,856 school-age children with urinary
incontinence. J Urol 2009;181:808-15; discussion 815-6. Crossref
6. Nevéus T, von Gontard A, Hoebeke P, et al. The
standardization of terminology of lower urinary tract
function in children and adolescents: report from the
Standardization Committee of the International Children’s
Continence Society. J Urol 2006;176:314-24. Crossref
7. Yüksel S, Yurdakul AC, Zencir M, Cördük N. Evaluation
of lower urinary tract dysfunction in Turkish primary
schoolchildren: an epidemiological study. J Pediatr Urol
2014;10:1181-6. Crossref
8. Vaz GT, Vasconcelo MM, Oliveira EA, et al. Prevalence
of lower urinary tract symptoms in school-age children.
Pediatr Nephrol 2012;27:597-603. Crossref
9. Chung JM, Lee SD, Kang DI, et al. An epidemiologic
study of voiding and bowel habits in Korean children: a
nationwide multicenter study. Urology 2010;76:215-9. Crossref
10. Kyrklund K, Taskinen S, Rintala RJ, Pakarinen MP. Lower
urinary tract symptoms from childhood to adulthood: a
population based study of 594 Finnish individuals 4 to 26
years old. J Urol 2012;188:588-93. Crossref
11. Wood D, Cottrell A, Baker SC, et al. Recreational ketamine:
from pleasure to pain. BJU Int 2011;107:1881-4. Crossref
12. Chu PS, Ma WK, Wong SC, et al. The destruction of the
lower urinary tract by ketamine abuse: a new syndrome?
BJU Int 2008;102:1616-22. Crossref
13. Chu PS, Kwok SC, Lam KM, et al. ‘Street ketamine’–associated bladder dysfunction: a report of ten cases. Hong
Kong Med J 2007;13:311-3.
14. Central Registry of Drug Abuse Sixty-third Report 2004-2013. Narcotics Division, Security Bureau, The Government
of the Hong Kong Special Administrative Region. Available
from: http://www.nd.gov.hk/en/crda_63rd_report.htm.
Accessed Dec 2015.
15. The 2011/12 survey of drug use among students. Narcotics
Division, Security Bureau, The Government of the Hong
Kong Special Administrative Region. Available from:
http://www.nd.gov.hk/en/survey_of_drug_use_11-12.htm.
Accessed Dec 2015.
16. Yee CH, Li JK, Lam HC, Chan ES, Hou SS, Ng CF. The
prevalence of lower urinary tract symptoms in a Chinese
population, and the correlation with uroflowmetry and
disease perception. Int Urol Nephrol 2014;46:703-10. Crossref
17. Coyne KS, Sexton CC, Thompson CL, et al. The prevalence
of lower urinary tract symptoms (LUTS) in the USA, the
UK and Sweden: results from the Epidemiology of LUTS
(EpiLUTS) study. BJU Int 2009;104:352-60. Crossref
18. Zhang L, Zhu L, Xu T, et al. A population-based survey
of the prevalence, potential risk factors, and symptom-specific
bother of lower urinary tract symptoms in adult
Chinese women. Eur Urol 2015;68:97-112. Crossref
19. Wang Y, Hu H, Xu K, Wang X, Na Y, Kang X. Prevalence,
risk factors and the bother of lower urinary tract symptoms
in China: a population-based survey. Int Urogynecol J
2015;26:911-9. Crossref
20. Chung JM, Lee SD, Kang DI, et al. Prevalence and associated
factors of overactive bladder in Korean children 5-13 years
old: a nationwide multicenter study. Urology 2009;73:63-7;
discussion 68-9. Crossref
21. Glassberg KI, Combs AJ, Horowitz M. Nonneurogenic
voiding disorders in children and adolescents: clinical and
videourodynamic findings in 4 specific conditions. J Urol
2010;184:2123-7. Crossref
22. Kajiwara M, Inoue K, Usui A, Kurihara M, Usui T. The
micturition habits and prevalence of daytime urinary
incontinence in Japanese primary school children. J Urol
2004;171:403-7. Crossref
23. Hellström A, Hanson E, Hansson S, Hjälmås K, Jodal U.
Micturition habits and incontinence in 7-year-old Swedish
school entrants. Eur J Pediatr 1990;149:434-7. Crossref
24. Akil IO, Ozmen D, Cetinkaya AC. Prevalence of urinary
incontinence and lower urinary tract symptoms in school-age
children. Urol J 2014;11:1602-8.
25. Tam YH, Ng CF, Pang KK, et al. One-stop clinic for
ketamine-associated uropathy: report on service delivery
model, patients’ characteristics and non-invasive
investigations at baseline by a cross-sectional study in a
prospective cohort of 318 teenagers and young adults. BJU
Int 2014;114:754-60. Crossref
26. Winstock AR, Mitcheson L, Gillatt DA, Cottrell AM. The
prevalence and natural history of urinary symptoms
among recreational ketamine users. BJU Int 2012;110:1762-6. Crossref
27. Koo KC, Lee DH, Kim JH, et al. Prevalence and management
of lower urinary tract symptoms in methamphetamine
abusers: an under-recognized clinical identity. J Urol
2014;191:722-6. Crossref