Hong Kong Med J 2014;20(6):504–10 | Epub 15 Aug 2014
DOI: 10.12809/hkmj144219
© Hong Kong Academy of Medicine. CC BY-NC-ND 4.0
ORIGINAL ARTICLE
A validation study of the Hong Kong version of Montreal Cognitive Assessment (HK-MoCA) in Chinese older adults in Hong Kong
PY Yeung, MB, ChB, MRCP (UK)1; LL Wong, FHKCPsych, FHKAM (Psychiatry)2; CC Chan, MRCP (UK), FHKCP3; Jess LM Leung, MRCPsych, FHKCPsych2; CY Yung, FHKCP, FHKAM (Medicine)3
1 Department of Rehabilitation, Kowloon Hospital, 147A Argyle Street,
Hong Kong
2 Department of Psychiatry, United Christian Hospital, Kwun Tong, Hong
Kong
3 Department of Medicine and Geriatrics, United Christian Hospital, Kwun
Tong, Hong Kong
Corresponding author: Dr PY Yeung (yeunpy3@ha.org.hk)
Abstract
Objective: To validate the Hong Kong version of
Montreal Cognitive Assessment (HK-MoCA) in
identification of mild cognitive impairment and
dementia in Chinese older adults.
Design: Cross-sectional study.
Setting: Cognition clinic and memory clinic of a
public hospital in Hong Kong.
Participants: A total of 272 participants (dementia,
n=130; mild cognitive impairment, n=93; normal
controls, n=49) aged 60 years or above were
assessed using HK-MoCA. The HK-MoCA scores
were validated against expert diagnosis according
to the Diagnostic and Statistical Manual of Mental
Disorders (4th ed) criteria for dementia and
Petersen’s criteria for mild cognitive impairment.
Statistical analysis was performed using receiver
operating characteristic curve and regression
analyses. Additionally, comparison was made
with the Cantonese version of Mini-Mental State
Examination and Global Deterioration Scale.
Results: The optimal cutoff score for the HK-MoCA
to differentiate cognitive impaired persons (mild
cognitive impairment and dementia) from normal
controls was 21/22 after adjustment of education
level, giving a sensitivity of 0.928, specificity of 0.735,
and area under the curve of 0.920. Moreover, the
cutoff to detect mild cognitive impairment was 21/22
with a sensitivity of 0.828, specificity of 0.735, and
area under the curve of 0.847. Score of the Cantonese
version of the Mini-Mental State Examination to
detect mild cognitive impairment was 26/27 with
a sensitivity of 0.785, specificity of 0.816, and area
under the curve of 0.857. At the optimal cutoff of
18/19, HK-MoCA identified dementia from controls
with a sensitivity of 0.923, specificity of 0.918, and
area under the curve of 0.971.
Conclusion: The HK-MoCA is a useful cognitive
screening instrument for use in Chinese older adults
in Hong Kong. A score of less than 22 should prompt
further diagnostic assessment. It has comparable
sensitivity with the Cantonese version of Mini-Mental State Examination for detection of mild
cognitive impairment. It is brief and feasible to
conduct in the clinical setting, and can be completed
in less than 15 minutes. Thus, HK-MoCA provides
an attractive alternative screening instrument to
Mini-Mental State Examination which has ceiling
effect (ie may fail to detect mild/moderate cognitive
impairment in people with high education level or
premorbid intelligence) and needs to be purchased
due to copyright issues.
New knowledge added by this
study
- This study verified that the Hong Kong version of Montreal Cognitive Assessment (HK-MoCA) has high diagnostic accuracy for detection of dementia (sensitivity, 92.3%; specificity, 91.8%).
- It is a reasonably good screening tool for mild cognitive impairment with comparable efficacy with the Cantonese version of Mini-Mental State Examination (MMSE).
- HK-MoCA is brief and feasible to conduct in the clinical setting, and can be completed in less than 15 minutes.
- HK-MoCA provides an attractive screening instrument in place of MMSE which has ceiling effect and needs to be purchased due to copyright issues.
Introduction
Dementia implies decline in cognitive function
interfering with an individual’s life. Mild cognitive impairment (MCI) is a clinical transitional state in
which a person is cognitively impaired, typically
in the memory domain, which is greater than that expected for a person at the given age and education
level. A longitudinal study revealed that MCI patients
proceeded to overt dementia at a rate of 10% to 15%
per year, compared with a rate of 1% to 2% in control
subjects.1 This implies that MCI patients have high
risk of progressing to dementia. Prevalence of MCI
varies widely, depending on the diagnostic criteria
used and population studied. Its prevalence ranges
from 3% to 13% in people above 65 years of age.2
Hence, identification of MCI patients is important for
successful implementation of preventive strategies
and early interventions. In practice, cognitive
screening tools are used to detect persons with
cognitive impairment who then undergo a detailed
assessment process to ascertain the subtype, severity,
caregiver status, and the presence of behavioural and
psychological symptoms of dementia.
The Mini-Mental State Examination (MMSE)
is the most widely used screening tool introduced
by Folstein et al in 1975.3 It was originally designed
for screening Alzheimer’s disease and does not encompass all cognitive deficits. It has several
well-known drawbacks, including low level of task
difficulty, likelihood of ceiling effects (ie may fail
to detect mild/moderate cognitive impairment
in people with high education level or premorbid
intelligence), and narrow range of cognitive domains
assessed. Consequently, it has low sensitivity for
MCI patient detection. The Cantonese version of
Mini-Mental State Examination (CMMSE) was
translated and validated by Chiu et al in 19944 with
good sensitivity (97.5%) and specificity (97.3%)
to discriminate subjects with moderate-to-severe
dementia from normal subjects.
The Montreal Cognitive Assessment (MoCA)
is a brief and potentially useful screening tool
developed and validated by Nasreddine et al.5 It was
conceptualised in MCI patients performing within a
normal range on the MMSE. The MoCA is a one-page
test with a maximum score of 30. One point
is added if the person has 12 years of education or
less. A score of 23 to 26 represents MCI, 17 to 22
represents moderate impairment, and 16 or below
represents severe impairment suggesting dementia.6
The original validation study of MoCA reported a
sensitivity of 100% and specificity of 87% in detecting
mild Alzheimer’s disease using a cutoff score of 26.
It reported a sensitivity of 90% in detecting MCI.6
It was validated to detect cognitive impairment in
different clinical populations including those with
Parkinson’s disease, brain metastases and stroke,
and had established cross-cultural performance
in detecting MCI and dementia.7 8 9 10 11 12 However,
educational adjustment and cutoffs varied. In a
Korean study, a cutoff score of 22/23 yielded a
sensitivity of 89% and specificity of 84% for screening
MCI.7 A study in mainland China suggested 20/21 as
cutoff score (Xie He Hospital version) with 84.6%
sensitivity and 76% specificity.8 It was the same as
the Hong Kong version: 73% sensitivity and 75%
specificity for patients with small vessel disease
(SVD).13 Furthermore, the original MoCA has high
level of internal consistency (Cronbach’s alpha of 0.83)
and test-retest reliability (correlation coefficient=0.92, P<0.001).5
This study employed the Hong Kong version
of MoCA (HK-MoCA) which has been validated
in Chinese patients with cerebral SVD by Wong
et al.13 The primary objective was to evaluate the
HK-MoCA as a screening tool in identification of
MCI and dementia in Chinese older adults, and to
determine the corresponding optimal cutoff points. In
addition, the ability of HK-MoCA in discriminating
dementia subtypes was examined.
Methods
Participants
This was a cross-sectional validation study performed
from August 2011 to June 2013 to validate HK-MoCA as a cognitive screening instrument. It was conducted
at the Cognition Clinic of Department of Medicine
and Geriatrics and the Memory Clinic of Department
of Psychiatry in a general hospital (United Christian
Hospital) of Hong Kong. Cantonese-speaking
Chinese adults aged 60 years or above, who were
seen for suspected cognitive impairment and gave
consent, were recruited. They were divided into
three groups: subjects with dementia, subjects who
met the criteria for MCI, and cognitively normal
controls (NC). Besides, NC were recruited from
those who attended clinics of other subspecialties or
elderly vaccination programmes under Department
of Medicine and Geriatrics.
Patients were excluded if they had a
history, as documented in medical records, of
neurodegenerative disorders, central nervous
system infection, brain tumour, significant head
trauma, subdural haematoma, epilepsy, significant
psychiatric disorders (such as major depression or
schizophrenia), substance abuse, or alcoholism.
Besides, persons with inability to use a pen or
with communication barriers such as deafness or
significant language or speech problem were also
excluded. Last of all, advanced dementia patients
with Global Deterioration Scale (GDS) stage 6 or
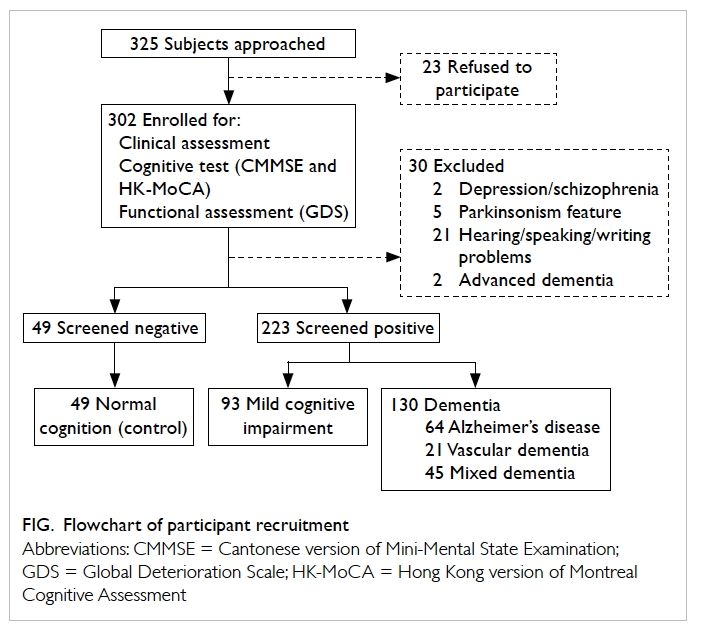
above were not recruited. The flowchart of participant recruitment is shown in the Figure. Ethical approval was
obtained from the Kowloon Central/Kowloon East
Cluster Clinical Research Ethics Committee of the
Hospital Authority.
Measurements
Clinical assessment
Basic demographic information (age, gender,
and education level), cardiovascular risk factors
(diabetes mellitus, hypertension, hyperlipidaemia,
coronary heart disease, and stroke) as well as
clinical information about drinking and smoking
habits was collected. Besides, a semi-structured
mental status examination and a comprehensive
neuropsychological battery (including biochemical
screening and cerebral imaging tests) were
performed for making a final cognitive diagnosis by
experienced geriatricians and psychogeriatricians
according to the DSM-IV (Diagnostic and Statistical Manual
of Mental Disorders, 4th edition) criteria14
for dementia and Petersen et al’s criteria for MCI.6 The
HK-MoCA scores were validated against expert
diagnoses.
Cognitive assessment
The CMMSE and HK-MoCA were administered
to each subject at the same consultation. The
administration was standardised and was done in
turn by two investigators. The HK-MoCA had more
questions and was harder than CMMSE. To avoid
frustration, CMMSE was administered before HK-MoCA. Fatigue may reduce attention span and
increase the likelihood of error. Hence, there was a
5-to-10-minute break to minimise the fatigue effect.
Functional decline of demented subjects was
determined using the GDS. It predicted a patient’s
ability to function, as reflected in activities of daily
living (ADL) and instrumental ADL as well as
psychiatric morbidity on the basis of progressive
cognitive delcine.9 It is composed of seven stages
defined by a set of clinical characteristics. Stages 1 to
3 are pre-dementia stages. Stages 4 to 7 are dementia
stages. Beginning in stage 5, an individual can no
longer survive without assistance.
Sample size calculation
Statistical software, MedCalc V12.3.0.0, was used for
sample size calculation. Based on a previous study,15
the estimated prevalence rates of MCI and dementia
were 8.5% and 12.5%, respectively. The overall sample
size was determined to be 138 (NC:MCI:dementia=1:1:1) with a power of 0.8 and a type I error of
0.05. In the receiver operating characteristic (ROC)
curve analyses of normal versus cognitive impaired
groups, a sample of 90 from the positive group
would achieve 90% power to detect a difference of
0.12 between the area under the curve (AUC)
of alternative hypothesis and an AUC under the null
hypothesis of 0.9000 (for MoCA) using a two-sided
z-test at a significance level of 0.05.
Statistical analyses
All statistical analyses were performed using the
Statistical Package for the Social Sciences (SPSS) version 17.0 and a difference with a P value of <0.05 was
regarded as statistically significant (two-tailed test).
Group differences in demographic characteristics
and various medical diseases were examined using
one-way analysis of variance or Chi squared test
for categorical data. Pairwise comparisons were
performed afterwards with the significance level
adjusted by the Bonferroni method. For differences
attaining statistical significance, multivariate linear
regression was performed to examine the influence
upon performance of the HK-MoCA total score.
Inter-rater reliability was reflected by intraclass
correlation coefficients with a sub-sample of 20
participants (persons with cognitive impairment and
NC) being tested 2 to 4 weeks apart. Convenience
sampling was employed with 10 participants by
each investigator. According to the optimal cutoff
points suggested in this study, five of them were NC,
four were persons with MCI, and 11 were demented
persons. Internal consistency was measured
using Cronbach’s alpha which measured pairwise
correlations between tested items. Criterion validity
was assessed using ROC analysis which gave the
sensitivity and specificity of HK-MoCA at different
cutoff points. With that, optimal cutoff scores
were chosen using highest Youden index (sensitivity
– [1 – specificity]). In case the indexes were very
close between the two scores, the one with higher
sensitivity would be chosen. The CMMSE and GDS
scores were used to test the concurrent validity.
The relationships between the performance of HK-MoCA,
CMMSE, and GDS were evaluated using
Pearson and Spearman’s rho correlation coefficients.
Finally, the discriminatory power of individual
cognitive domains was explored by examining any significant difference in scores among the three
groups.
Results
A total of 272 eligible subjects completed the
HK-MoCA screening in which 49 were NC, 93 were
MCI subjects, and 130 were demented subjects (99
with mild and 31 with moderate severity of dementia).
Of all the subjects with dementia, 49.2% had
Alzheimer’s disease, 16.2% had vascular dementia,
and 34.6% had dementia of mixed aetiology. The
administration time depended on the education
level and severity of cognitive impairment, and was
around 10 to 15 minutes.
Demographic and clinical characteristics
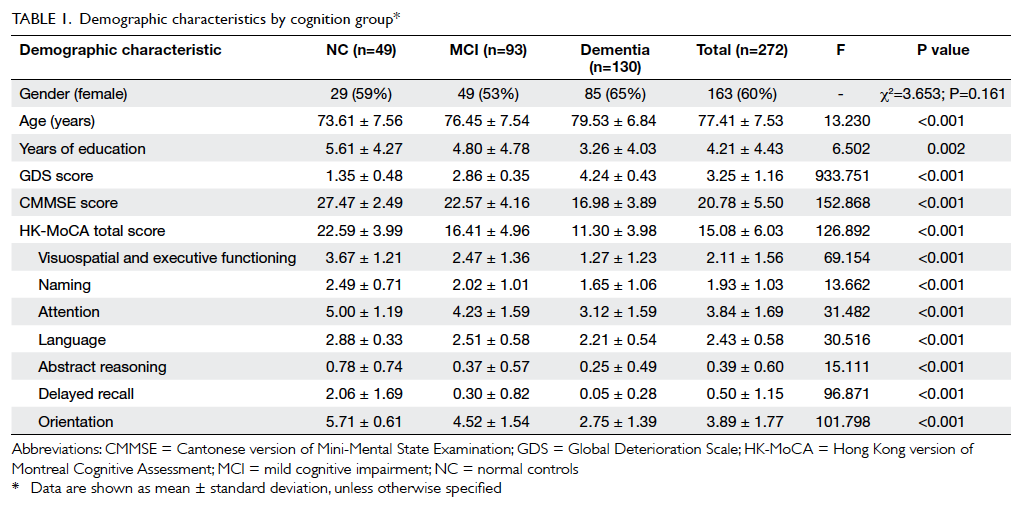
Sample characteristics are summarised in Table 1.
Overall, 60% of recruited subjects
were females with a mean (± standard deviation)
age of 77.41 ± 7.53 years. The level of education was
primary and below in 76% of the participants, with
a mean of 4.21 ± 4.43 years of education. Significant
differences among the three groups (NC, MCI,
and dementia) were found in the variables of age
(F [2,269]=13.230, P<0.001), years of education
(F [2,269]=6.502, P=0.002), HK-MoCA score
(F [2,269]=126.892, P<0.001), CMMSE score (F
[2,269]=152.868, P<0.001), and GDS score (F
[2,269]=933.751, P<0.001). There was no significant
difference among the three cognition groups by
gender (Chi squared test=3.653, P=0.161). Subjects
in the dementia group were significantly older (79.53 ± 6.84 years; P<0.001) and had less years of education
(3.26 ± 4.03 years; P=0.002) than those in the other two groups. In contrast, no significant differences
were demonstrated among the three subgroups of
dementia (Alzheimer’s disease, vascular dementia,
and mixed dementia) in terms of age (F [2,127]=2.873, P=0.060), years of education (F [2,127]=0.630, P=0.534), HK-MoCA score (F [2,127]=0.428,
P=0.653), CMMSE score (F [2,127]=0.322, P=0.725),
and GDS score (F [2,127]=0.161, P=0.851).
There was no significant difference between
those with dementia and NC in terms of drinking
and smoking habits, and various medical conditions,
except for stroke (P=0.031). The result was reasonable
as stroke is a known cause of dementia.
Score distribution of Hong Kong version of
Montreal Cognitive Assessment
From the original MoCA study, differences across
cognition groups were more pronounced using
MoCA than MMSE.5 This study did not reproduce
the wide dispersion of MoCA scores and, indeed,
mean scores of HK-MoCA of various groups were
lower in general. The results were justified by the
generally low education level of local Chinese older
adults.
The effect of age and education level
The appropriateness of original education adjustment
of MoCA total score was uncertain in local Chinese
older adults. This study examined the effect of
age and education level upon the performance
of HK-MoCA by regressing the unadjusted raw
score of HK-MoCA on age, years of education, and
clinical diagnosis using multivariate linear models.
The results are summarised in Table 2. There was
a positive relationship between years of education and performance on HK-MoCA (β, 0.318; P<0.001)
independent of age and clinical diagnosis. The effect
of age was not significant (P=0.530).
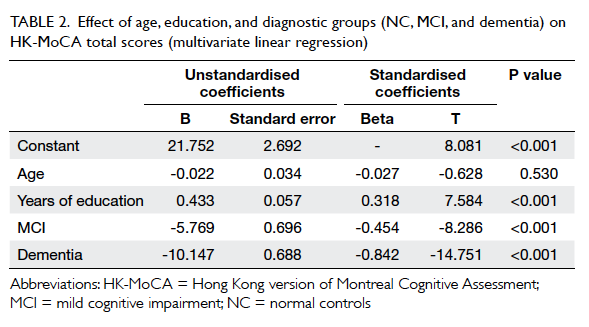
Table 2. Effect of age, education, and diagnostic groups (NC, MCI, and dementia) on HK-MoCA total scores (multivariate linear regression)
The original MoCA recommendation of adding
one point to subjects with 12 years of education or
less was probably unsuitable here. In this study, the
level of education in 76% of subjects was primary and
below. Only 11 (4%) subjects had more than 12 years
of education. For this reason, we adopted a lower
level of education adjustment to 6 years of education
which had been employed by studies in China,8 10
Korea,7 and Hong Kong.13 Using this adjustment
for education level, regression R2 coefficient was 0.587.
Thus, more than half of the variation in HK-MoCA was
explained by the model.
Psychometric properties of Hong Kong
version of Montreal Cognitive Assessment
Intraclass correlation coefficient for the inter-rater
reliability was 0.987 (P≤0.001). Besides, the
Cronbach’s alpha score was 0.767, indicating a high
level of internal consistency. Comparison of GDS, CMMSE, and HK-MoCA scores between NC, MCI,
and dementia groups showed that the severity level
of cognitive impairment graded by GDS score was
significantly correlated with HK-MoCA score with
Spearman’s rho correlation coefficient of -0.739
(P≤0.001). Similarly, Pearson correlation coefficient
of CMMSE with HK-MoCA scores was 0.894
(P≤0.001). Together, it supported high concurrent
validity of HK-MoCA.
Criterion validity of the adjusted HK-MoCA
score was examined using ROC analysis. Various
optimal cutoff scores are listed in Table 3. The optimal cutoff score for HK-MoCA to differentiate
persons with cognitive impairment (MCI and
dementia) from NC was 21/22, giving a sensitivity of
0.928, specificity of 0.735, and AUC of 0.920 (95%
confidence interval [CI], 0.881-0.959). Moreover, the cutoff to detect MCI was also 21/22 with a sensitivity of 0.828, specificity of 0.735, and AUC of 0.847 (95% CI, 0.778-0.902). For comparison, CMMSE score to detect MCI was 26/27 with a sensitivity of 0.785, specificity of 0.816, and AUC of 0.857. The optimal cutoff
score for HK-MoCA to detect dementia was 18/19 with a sensitivity of 0.923, specificity of 0.918, and AUC of 0.971 (95%
CI, 0.935-0.991).
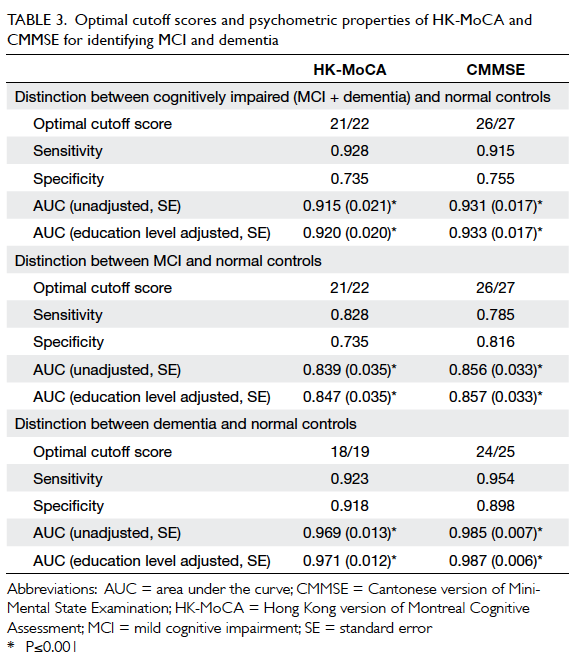
Table 3. Optimal cutoff scores and psychometric properties of HK-MoCA and CMMSE for identifying MCI and dementia
Discriminatory ability
The HK-MoCA total score and seven cognitive
domain scores discriminated NC, MCI, and
dementia groups in a stepwise fashion. In general,
demented participants performed most poorly,
followed by the MCI participants. Like the original
study, delayed recall task was the first and most
impaired domain in MCI participants. Besides,
sample participants with GDS score equal to 4
were used to examine the discriminatory ability in
differentiating subtypes of dementia; no significant
difference was demonstrated (P values ranged from
0.243 to 0.672). The generally low education level
and small dementia subgroup sample size might have
compromised the results.
Discussion
Validity and clinical utility of the Hong Kong
version of Montreal Cognitive Assessment
This study verified that HK-MoCA has high
diagnostic accuracy for detecting dementia subjects
(92.3% sensitivity, 91.8% specificity). It is reasonably
good and comparable with CMMSE in screening
for MCI. The original MoCA by Nasreddine et al5
in 2005 and other validation studies of MoCA in
different languages established the superiority to
MMSE. Explanations are that MMSE did not test
complex cognitive impairments in domains such
as visuospatial/executive function and abstract
reasoning. In addition, the attention and delayed
recall tasks are not as challenging as that in MoCA. In
practice, MoCA picks up more deficits in executive
function, attention, and delayed recall.16 This study
did not reproduce the superiority to MMSE and this
may be related to the low education level of Chinese
older adults. Due to the Chinese Civil War from 1927
to 1950, the majority of elderly Chinese individuals
did not receive much education and many were
illiterate. According to published data,17 the average
number of years of education for elderly Chinese
individuals is about 5 years, which is significantly
less than that of their western counterparts.
The validity of HK-MoCA is based on its non-inferiority to CMMSE. This study compared
HK-MoCA with CMMSE using a new cutoff point
derived from the same study and found comparable
sensitivity and specificity in detection of MCI. If
the CMMSE cutoff as suggested by Chiu et al4 was
utilised, HK-MoCA is definitely more sensitive.
As such, HK-MoCA is relatively easy to use (both
required less than 15 minutes to administer) and
incorporates important domains missed in CMMSE.
It is a clinically efficient and effective screening
instrument and can be generalised for use in Chinese
older adults with MCI or dementia. Customarily,
many memory clinics utilise MMSE as a screening
tool as it is convenient to use and available free of
charge. Considering the ceiling effect of MMSE due
to the low level of task difficulty and the copyright
fees introduced recently, validated HK-MoCA
provides an attractive alternative.
Psychometric properties of the Hong Kong
version of Montreal Cognitive Assessment
Montreal Cognitive Assessment is one of the
common cognition screening instruments used
locally and worldwide. It is commonly used to
discriminate cognitive impairment due to various
causes. In Hong Kong, there was only one validation
study conducted by Wong et al13 involving use of
HK-MoCA in patients with cerebral SVD. They
demonstrated that HK-MoCA differentiated SVD
patients from controls (AUC=0.81) with an optimal
cutoff at 21/22. This cutoff point was valid to
predict SVD patients with cognitive impairment
only, although in clinical practice, it was commonly
used to discriminate cognitive impairment of
various causes. This study successfully generalised
the validity of HK-MoCA for identifying MCI and
dementia in Chinese older adults, and determined
the optimal cutoff points of these conditions. The
optimal cutoff points yielded were similar to those in
previous studies in China8 10 and Korea7 using the same
descended educational adjustment. The HK-MoCA
is useful for detecting persons with cognitive
impairment in Chinese older adult population and
a score of below 22 should prompt detailed diagnostic
investigations. The results demonstrated good
intra-rater and inter-rater reliability and internal
consistency. It showed good convergent validity
with CMMSE and GDS scores as well. Besides, the
study investigated the effect of education on this
cognitive screening instrument with respect to the
low education level of Chinese older adults and
employed a descended education adjustment from
12 to 6 years of education. This descended education
adjustment is supported by studies conducted in
China8 and Korea.7
Limitations
There were several limitations to the HK-MoCA. This instrument required the participants to follow verbal
and written commands, hence the performance of
elderly with hearing or visual impairment would be
affected. Illiterate or poorly educated persons might
have difficulty in comprehending the instructions
and the cube and clock drawing tasks were too difficult.
Furthermore, stroke patients whose dominant hand
has been affected might not be able to perform the
drawing test.
In this study, subjects were recruited from
a local general hospital situated in a lower social
class residential area. Three quarters of the
participants received primary education or less. The
descended education adjustment from 12 to 6 years
of education should be subject to review with
respect to the trend of education received by older
adults. Besides, short break between CMMSE and
HK-MoCA administration might not totally relieve the
fatigue error. One might argue that geriatricians and
psychogeriatricians in this study were not blinded
from the HK-MoCA, CMMSE and GDS scores,
which might have introduced bias when they made the
final cognitive diagnosis. Furthermore, inter-rater
reliability established using convenience sampling of
20 participants being tested 2 to 4 weeks apart was
not an optimal way to determine the concordance
between the two co-investigators. Last but not the
least, the predictive values could not be ascertained
in this study as the patient groups and NC were
not recruited consecutively from a designated
population, leaving the true prevalence unknown.
Further study can explore the ability of HK-MoCA
to grade the severity of cognitive impairment and
predict long-term cognitive decline.
Conclusion
This study validated that HK-MoCA is a sensitive
screening instrument for use in Chinese older adults
in Hong Kong with MCI or dementia, irrespective of
the underlying aetiology. This validated HK-MoCA
is brief and feasible to conduct in the clinical setting,
and can be completed in less than 15 minutes. It is an
attractive alternative screening instrument to MMSE
which has ceiling effect and needs to be purchased
due to copyright issues. A score of less than 22
should prompt further diagnostic assessment.
Acknowledgement
The authors thank Dr Ziad S Nasreddine for
his permission to use the Hong Kong version of
Montreal Cognitive Assessment test.
Declaration
No conflicts of interest were declared by authors.
References
1. Lam LC, Tam CW, Lui VW, et al. Prevalence of very mild
and mild dementia in community-dwelling older Chinese
people in Hong Kong. Int Psychogeriatr 2008;20:135-48. CrossRef
2. Damian AM, Jacobson SA, Hentz JG, et al. The Montreal
Cognitive Assessment and the mini-mental state
examination as screening instruments for cognitive
impairment: item analyses and threshold scores. Dement
Geriatr Cogn Disord 2011;31:126-31. CrossRef
3. Folstein MF, Folstein SE, McHugh PR. “Mini-mental state”.
A practical method for grading the cognitive state of
patients for the clinician. J Psychiatr Res 1975;12:189-98. CrossRef
4. Chiu HF, Lee HC, Chung WS, Kwong PK. Reliability and
validity of the Cantonese version of mini-mental state
examination—a preliminary study. J Hong Kong Col
Psychiatrists 1994;4:25-8.
5. Nasreddine ZS, Phillips NA, Bédirian V, et al. The Montreal
Cognitive Assessment, MoCA: a brief screening tool for
mild cognitive impairment. J Am Geriatr Soc 2005;53:695-9. CrossRef
6. Petersen RC, Smith GE, Waring SC, Ivnik RJ, Tangalos
EG, Kokmen E. Mild cognitive impairment: clinical
characterization and outcome. Arch Neurol 1999;56:303-8. CrossRef
7. Lee JY, Lee DW, Cho SJ, et al. Brief screening for mild
cognitive impairment in elderly outpatient clinic:
validation of the Korean version of the Montreal Cognitive
Assessment. J Geriatr Psychiatry Neurol 2008;21:104-10. CrossRef
8. Wang YP, Xu GL, Yang SQ, Liu XM, Deng XY. Value of
Montreal Cognitive Assessment in identifying patients
with mild vascular cognitive impairment after first stroke.
Chinese Journal of Neuromedicine 2010;9:503-7.
9. Reisberg B, Ferris SH, de Leon MJ, Crook T. The Global
Deterioration Scale for assessment of primary degenerative
dementia. Am J Psychiatry 1982;139:1136-9.
10. Tu QY, Jin H, Ding BR, et al. Reliability, validity, and
optimal cutoff score of the Montreal Cognitive Assessment
(Changsha version) in ischemic cerebrovascular disease
patients of Hunan Province, China. Dement Geriatr Cogn
Dis Extra 2013;3:25-36. CrossRef
11. Rahman TT, El Gaafary MM. Montreal Cognitive
Assessment Arabic version: reliability and validity
prevalence of mild cognitive impairment among elderly
attending geriatric clubs in Cairo. Geriatr Gerontol Int
2009;9:54-61. CrossRef
12. Fujiwara Y, Suzuki H, Yasunaga M, et al. Brief screening
tool for mild cognitive impairment in older Japanese:
validation of the Japanese version of the Montreal Cognitive
Assessment. Geriatr Gerontol Int 2010;10:225-32. CrossRef
13. Wong A, Xiong YY, Kwan PW, et al. The validity, reliability
and clinical utility of the Hong Kong Montreal Cognitive
Assessment (HK-MoCA) in patients with cerebral small
vessel disease. Dement Geriatr Cogn Disord 2009;28:81-7. CrossRef
14. American Psychiatric Association. Diagnostic and
Statistical Manual of Mental Disorders, 4th ed. Washington,
DC: American Psychiatric Association; 1994.
15. Elderly Commission: Prevalence of dementia in
Hong Kong, 2006. Available from: http://www.elderlycommission.gov.hk/en/meeting/47.html. Accessed Jun 2011.
16. Wong A, Kwan P, Chan AY, et al. The validity, reliability and
utility of the Cantonese Montreal Cognitive Assessment
(MoCA) in Chinese patients with confluent white matter
lesions. Hong Kong Med J 2008;14(Suppl 6):7S.
17. Chan AS, Choi A, Chiu H, Lam L. Clinical validity of
the Chinese version of Mattis Dementia Rating Scale in
differentiating dementia of Alzheimer’s type in Hong
Kong. J Int Neuropsychol Soc 2003;9:45-55. CrossRef