Hong Kong Med J 2014 Oct;20(5):386–92 | Epub 11 Apr 2014
DOI: 10.12809/hkmj134066
© Hong Kong Academy of Medicine. CC BY-NC-ND 4.0
ORIGINAL ARTICLE
Laparoscopic liver resection: lessons learnt after 100 cases
Fiona KM Chan, MB, BS; KC Cheng, MB, BS, FHKAM (Surgery); YP Yeung, MB, BS, FHKAM (Surgery)
Department of Surgery, Kwong Wah Hospital, 25 Waterloo Road, Kowloon, Hong Kong
Corresponding author: Dr FKM Chan (ckmfiona@hotmail.com)
Abstract
Objective: To share our institutional experience
in laparoscopic liver resection and our learning
curve after the first 100 cases of laparoscopic liver
resection.
Design: Case series with internal comparison.
Setting: A regional hospital in Hong Kong.
Patients: Our institution started performing
laparoscopic liver resection since 2006. All patients
who underwent laparoscopic liver resections from
March 2006 to October 2012 were identified in a
prospectively collected database. The demographic
data and operative outcomes of these patients were
extracted, and results of the early (from March 2006
to May 2010) and late (from June 2010 to October
2012) study periods were compared.
Results: Between March 2006 and October 2012, 100
laparoscopic liver resections were performed for 98
patients in the Department of Surgery, Kwong Wah
Hospital, Hong Kong. They were 69 (70%) males and
29 (30%) females, and the median age was 65 years.
The final histological diagnoses were as follows:
hepatocellular carcinoma (n=72), colorectal liver metastases (n=14), intrahepatic cholangiocarcinoma
(n=4), and benign disease (n=10). There were more
anatomical resections, major hepatectomies as well
as resections of more anatomically challenging
right-sided and posterosuperior lesions in the late
versus the early period; however, operative outcomes
remained comparable in both periods.
Conclusion: Laparoscopic hepatectomies are
feasible with growing experience. Bearing in mind
the diversity in the level of operative techniques with
various types of laparoscopic liver resections, more
experience is needed to overcome the learning curve.
New knowledge added by this
study
- Laparoscopic hepatectomies are feasible in our hospital where operative skills and techniques improved over time with experience.
- Laparoscopic hepatectomies should be carried out in high-volume centres. Favourable results can be achieved with adequate experience, considering the steepness of the learning curve. The wide range of operative techniques for various types of laparoscopic hepatectomies implies that further exploration in training and learning curve effect is needed.
Introduction
Since its inception in 1992, laparoscopic liver
resection (LLR) has been increasingly employed as
the new alternative to open liver resection.1 Over
3000 cases of LLRs have been reported worldwide.2
The safety and efficacy of the procedure have been
shown in recent evidence to be comparable with
open surgery. The advantages of LLR over traditional
open surgery are less analgesic requirements,
less operative blood loss, shorter hospital stay,
accompanied with a low operative mortality
and morbidity of 0.3% and 11%, respectively.3 4 5 6
Laparoscopic left lateral sectionectomies and wedge resections are now considered standardised
operations performed routinely in dedicated
centres.7
Laparoscopic hepatectomy is considered to be
a complicated laparoscopic procedure. The surgeon
should be experienced in both laparoscopic and liver
surgery. Most of the studies on the learning curve
effect show improved outcome with experience.8 9 10 11
However, LLR is not a single procedure and the
complexity of operation ranges from wedge resections
to major hepatectomies involving anatomical
resection of three or more segments. Experience
in the complexity of procedure performed has not been adequately studied. In Kwong Wah Hospital,
our experience with laparoscopic liver surgery
commenced in 2006, and since then the procedure
has been performed in an increasing number of
patients. Here we report our experience with the
first 100 cases of LLR and the learning curve effect
through the series of cases.
Methods
All data including patient demographics, tumour
characteristics, operative procedures, and outcomes
were prospectively collected. All patients underwent
chest radiography and contrast computed tomography
(CT) of the abdomen. Magnetic resonance imaging
and lipiodol arteriogram were performed in selected
patients. Patients’ preoperative liver function was
assessed according to the Child-Pugh classification,12
and with indocyanine green retention test and CT
volumetric analysis, if necessary. Patients were
selected for laparoscopic liver surgery if they were
medically fit for the major operation, Child-Pugh
class A or B liver cirrhosis with adequate liver
remnant after resection. Our centre adopted a less
stringent criterion in terms of patient selection;
hence, patients were included if LLR was considered
technically feasible after evaluating patients’ history
of surgical operations, tumour size, and location.
In the later years, indications were expanded to
include resection of more benign pathologies and cholangiocarcinoma, wherein resection was
anticipated to be more difficult.13 14
Operative techniques
All patients were operated on by specialist hepatobiliary
surgeons with expertise in laparoscopic surgery.
Hand-assisted or laparoscopic-assisted approaches
were employed in the earlier period; however, the
approach changed into total laparoscopy in the later
period. Patient was put in Lloyd-Davis position for
left hepatectomies and in semi-left lateral or left
lateral position for right-sided lesion. Intra-operative
laparoscopic ultrasound was used routinely. Five
ports were used and placed according to tumour
location. Parenchymal dissection was performed by
Cavitron ultrasonic surgical aspirator (Valleylab Inc,
Boulder [CO], US), Harmonic Scalpel (Harmonic
ACE; Ethicon Endosurgery, Johnson & Johnson,
Langhorne [PA], US) or LigaSure (Valleylab).
Methods employed for haemostasis included bipolar
diathermy, metal clips, Hem-o-lok (Weck Surgical
Instruments, Teleflex Medical, Durham [NC], US)
or endovascular staplers; the Pringle manoeuvre was
not used routinely. Specimen was put inside a plastic
bag and retrieved via a Pfannenstiel incision if the
specimen was large or by extension of one of the port
sites if the specimen was small. Pneumoperitoneum
was re-established after specimen retrieval at a
pressure of 6 to 8 mm Hg to check for haemostasis.
Tissue glue (Tisseel; Baxter, Vienna, Austria) was
applied selectively. Abdominal drains were inserted
as needed.
Statistical analyses
Patients who underwent laparoscopic hepatectomies
were divided chronologically into two periods for
comparison. Those performed from March 2006 to
May 2010 were classified into the ‘early group’ while
those performed from June 2010 to October 2012
were classified into the ‘late group’. All data including
patient demographics, operative and postoperative
parameters were retrieved from a prospectively
collected database. Operative parameters included
operation type, conversion, operating time, blood
loss, transfusion requirement, duration of Pringle
manoeuvre, and intra-operative complications.
The postoperative parameters included resection
margin, staging, medical and surgical complications,
length of hospital stay, and operative mortality.
Complications were recorded and classified
according to the Clavien-Dindo classification.15
Postoperative survival was measured using Kaplan-Meier estimates.
Statistical analyses were performed with the
Statistical Package for the Social Sciences (Windows
version 16.0; SPSS Inc, Chicago [IL], US). Numerical
data were expressed as the median value. Mann-Whitney U test was used for comparing continuous variables. Chi squared test and Fisher’s exact test
were used for comparing categorical variables.
Statistical significance was set at a P value of less
than 0.05.
Results
From March 2006 to October 2012, our unit
performed a total of 212 hepatectomies. A
laparoscopic approach was employed in 98 patients.
The proportion of LLRs performed increased from
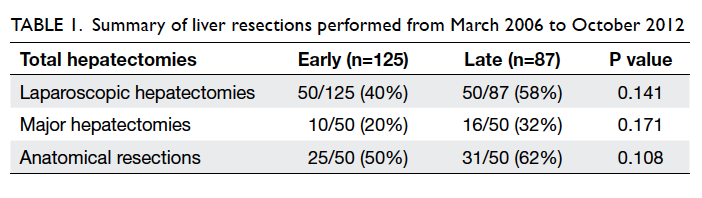
40% in the early group to 58% in the late group. There
was an increasing proportion of laparoscopic major
hepatectomies and anatomical resections in the late
group versus the early group (Table 1).
A total of 98 patients who underwent LLRs
were recruited during this study period. There were
69 (70%) male and 29 (30%) female patients; the
median age was 65 years. Of the 98 patients, two
underwent a second LLR, giving a total of 100 LLRs.
Some of these patients had previously undergone
conventional hepatectomy. The Eastern Cooperative
Oncology Group status was 0 for all patients. The
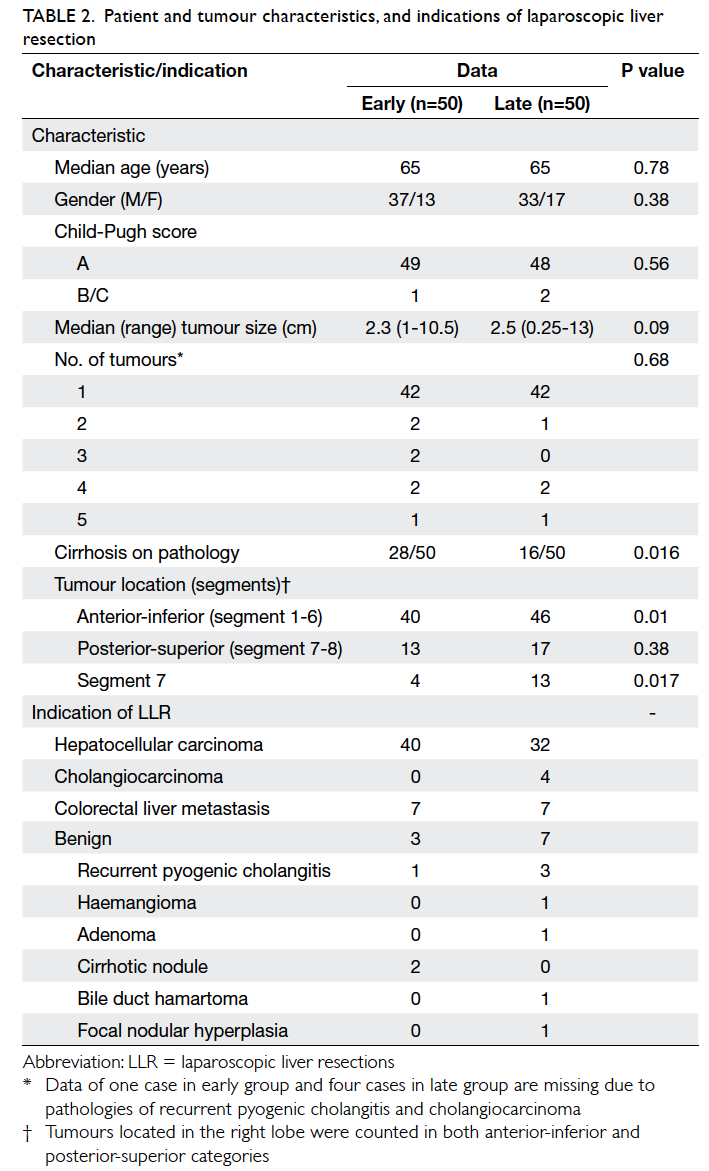
demographic data and tumour characteristics of
the two groups are shown in Table 2. There were
significantly more patients with cirrhosis in the early
group (P=0.016), and more patients had segment 7
tumour in the late group (P=0.017).
Indications for liver resection are shown
in Table 2. Overall, 72% of LLRs were performed
for hepatocellular carcinoma, whereas benign
pathologies accounted for 10% of all LLRs. There was
an increase in the number of LLRs performed for
benign pathologies and cholangiocarcinomas in the
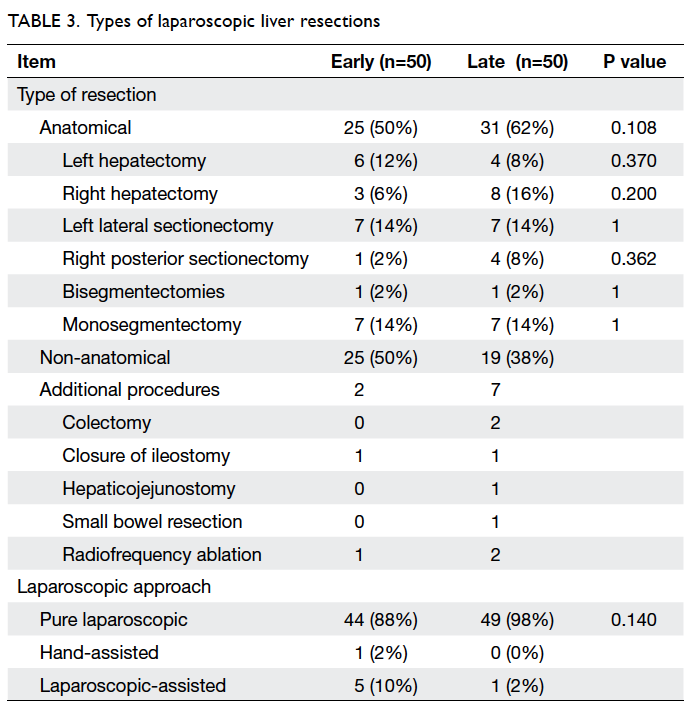
late versus the early period. The types of resection
performed are listed in Table 3. The proportion of
anatomical resections increased from 50% in the
early period to 62% in the late period, including
predominantly right hepatectomies (6% vs 16%)
and right posterior sectionectomies (2% vs 8%).
An increasing proportion of major hepatectomies,
including right and left hepatectomies, as well as
right posterior sectionectomies, were performed
in the late period (20% vs 32%). In addition, more
resections involving the posterosuperior segments
(including segments 7 and 8) were performed
in the late period (34% vs 26%); these are considered
to be anatomically more difficult resections. Pure
laparoscopic approach was employed in the majority
of LLRs, and more LLRs were performed with a pure
laparoscopic approach in the late period than in the
early period (98% vs 88%) [Table 3].
A number of procedures were performed
alongside with LLRs. These included two laparoscopic
colectomies, two closures of ileostomies, one
hepaticojejunostomy, one small bowel resection, and
three radiofrequency ablation–assisted LLRs.
Table 4 shows the intra-operative results,
postoperative complications, status of margin involvement, and hospital stay. Conversion rates
were higher in the late period than in the early
period (14% vs 2%) but did not reach statistical
significance. Among the operations that required
conversion to a standard approach (n=8), three were
due to haemorrhage, and the rest were due to poor exposure, dense adhesions with resultant small bowel
injury, anatomical limitations at posterior segment,
and doubtful tumour margin during resection.
There was no mortality in the early group and one
in the late group. Complications were classified
according to the Clavien-Dindo classification and
are shown in Table 4. One patient in the early group
who had situs inversus experienced complications
in the form of bile leakage from a segment 4 branch
after an anatomical right hepatectomy; this patient
required laparotomy with T-tube insertion. One
patient in the late group was found to have extensive
bowel ischaemia on postoperative day 2 after a
laparoscopic right hepatectomy; this patient required
reoperation but did not survive. Two patients, one
each from the early and late groups, developed
bile leak postoperatively after laparoscopic right
hepatectomies; they were managed with image-guided
drainage and endobiliary stenting.
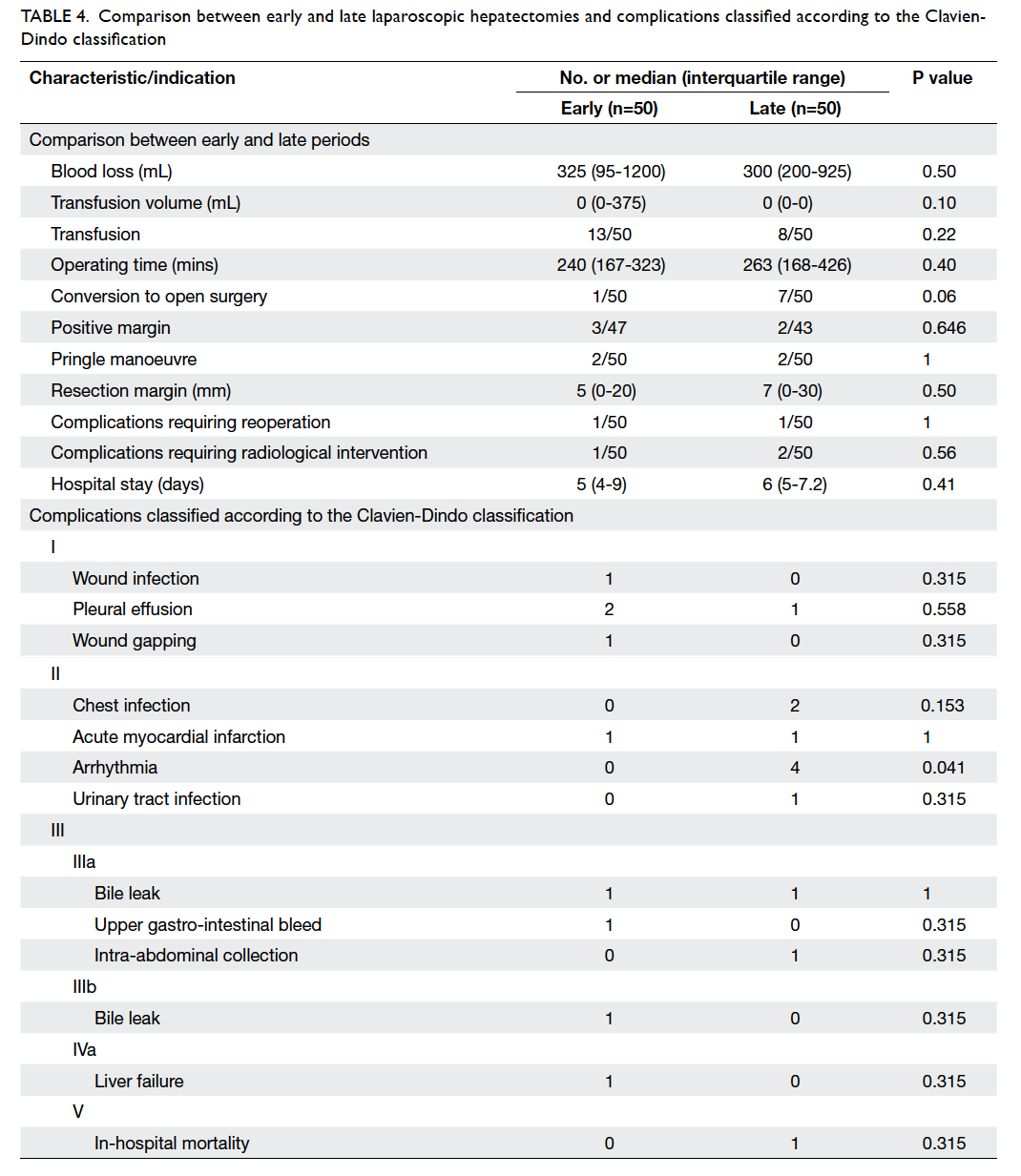
Table 4. Comparison between early and late laparoscopic hepatectomies and complications classified according to the Clavien-Dindo Classification
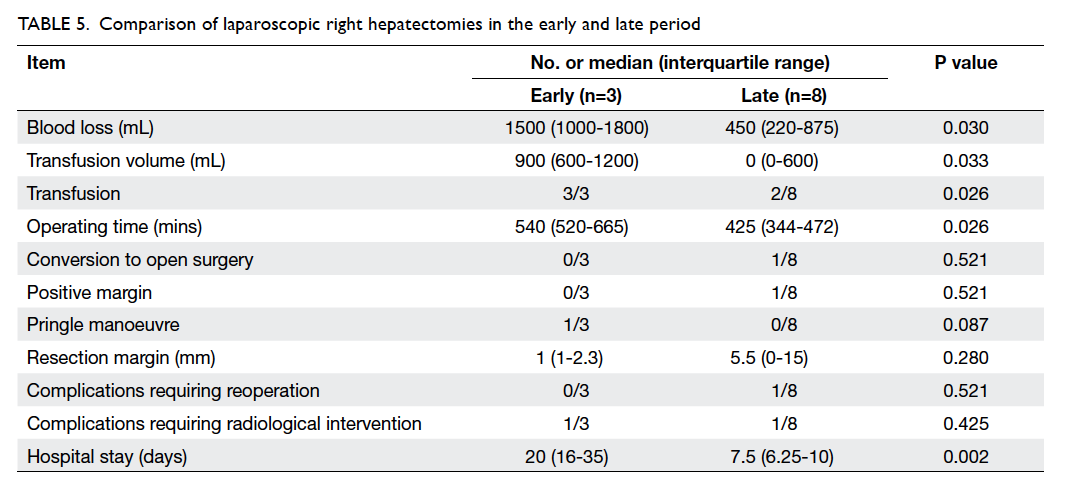
A subgroup analysis was conducted for patients
receiving laparoscopic right hepatectomies. Between
the early and late period, a total of 11 laparoscopic
right hepatectomies were performed. Table 5 shows
the peri-operative results of these patients. With
increasing experience, the operating time, blood
loss, transfusion rate and volume, as well as duration
of hospital stay were significantly reduced.
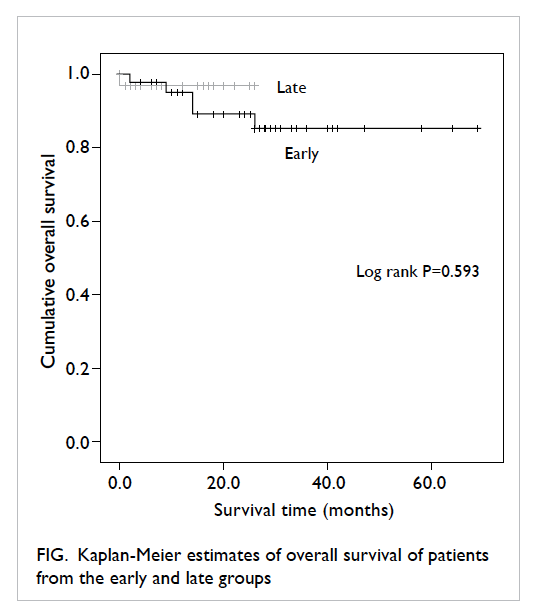
The 2-year survival, according to Kaplan-Meier survival analysis, showed an overall survival of 89.1% in the early group versus 96.9% in the late
group (log rank P=0.593; Fig). Since the majority of
the study population were recruited after 2008, 5-year
survival data from this main bulk of patients were
not available for this analysis.
Discussion
Laparoscopic hepatectomies are technically
demanding.16 17 The difficulty lies in parenchymal
transection with limited exposure and traction, thus
requiring proficiency in both laparoscopic and liver
surgery. The reproducibility and feasibility of the
procedure have been questioned, preventing the
procedure from being widely employed. Our current
study demonstrated that, with growing experience,
we could perform LLR safely, as demonstrated by
the favourable overall outcome of LLR. The rates
of overall mortality and major morbidity were 1%
and 5%, respectively. Reoperation was required
in two (2%) patients. For malignant indications,
R0 resection rate (complete resection with no
microscopic residual tumour) was 94% (85/90). The
overall results are in accordance with reports in the
literature.2 5
Blood transfusion was required in 21% of our
patients and the conversion rate was 8%. We did
not use the Pringle manoeuvre frequently because
most of the bleeding occurred from hepatic veins. Among these eight patients requiring conversion, three quarters were related to bleeding from branches of the hepatic vein. We preferred a
pure laparoscopic approach because the use of a
hand port caused interference with laparoscopic
trocars and instruments.18 It has been suggested
that hand-assisted or hybrid approach offers
speedy haemostasis but there is no solid evidence
to support which single method is superior. We did
not consider conversion to be a failure and hence,
a higher conversion rate (14% vs 2%, P=0.06) was
observed with a lower blood transfusion rate (16%
vs 26%, P=0.22) in the late versus the early period.
No strict transfusion criteria were implemented.
The decision of blood transfusion was mostly made
by individual anaesthetist intra-operatively. Early in
our series, we tended to initiate transfusion early
because we anticipated bleeding during LLR to be
more difficult to control. With gaining experience,
transfusion was given more judiciously. Thus, with
similar median blood loss, there was a trend towards
lower transfusion rate in the late group as compared
with the early group.
We further analysed the outcomes of the
LLRs performed in the early and late periods. The
overall outcome parameters were comparable with
no significant learning curve effect observed. We
observed a slight increase in operating time (263 vs
240 mins, P=0.40) and duration of hospital stay (6 vs 5
days, P=0.41) during the later period, and we believe that this was probably related to the increasing
number of laparoscopic major hepatectomies and
anatomical resections of right-sided lesions, as well
as posterior segment LLRs performed in the later
period.
With increasing experience in performing
LLRs, we extended our indications of LLR from
peripherally located tumours to posterosuperior
lesions and from wedge resections to major
resections, all reflecting an improvement in our techniques of performing LLR. However, we believe
that we are still on the learning curve for the more
difficult LLRs because the operative outcomes did
not improve much. We managed to perform more
anatomical resections with time in order to secure
oncological safety. However, 5-year survival and
recurrence results of our patients are not available
for comparison between these two study groups.
The subject of learning curve effect of
laparoscopic hepatectomy has been investigated by several authors in the literature. Many studies
attempted to identify the number of hepatectomies
required to overcome the learning curve effect.8 9 10 11 The
12-year experience of Vigano et al9 demonstrated
that after performing 60 consecutive cases of
laparoscopic hepatectomies, operative outcomes in
three consecutive periods in terms of conversion
rate (15.5%, 10.3%, and 3.4%; P<0.05), operating
time (210, 180, and 150 mins; P<0.05), blood loss
(300, 200, and 200 mL; P<0.05), and morbidity
(17.2%, 22.4%, and 3.4%; P<0.05) improved. They
reported a steady increase in the proportion of LLRs and a statistically significant increment in major
and right hepatectomies in the later period of the
study. The cumulative analysis of conversion rates
in minor hepatectomies showed that at the 60th
consecutive case, the conversion rate reached the
average value and improved thereafter. A Korean
group examined the results from their first 100
cases of laparoscopic liver surgery.8 Their mean
operating time was 220 minutes and the overall
morbidity was 11%. They demonstrated a decrease
in the volume of blood transfusion in the latter half
of patients operated with a malignant pathology.
Kluger et al11 investigated the learning curve effect
in laparoscopic major hepatectomy. Dividing their
study results chronologically into three phases, they
showed a steady increase in the proportion of major
LLRs (1% vs 9%, P<0.05) and malignant lesions being
resected at a later stage in the study period. Median
operating time (150 vs 210 mins, P<0.05), blood
loss (200 vs 300 mL, P<0.05), and clamping time (20
vs 45 mins, P<0.05) were significantly lower in the
later study period. Morbidity rates also improved
significantly with time (3% vs 17%, P<0.05). Their
group concluded that a learning curve existed for
both the operator and the institution, and a high-volume
environment enables overcoming of the
learning curve. The latest experience in the attempt
to identify a learning curve came from a UK group.10
Analysing their 37 LLRs, the researchers concluded
that their results followed a learning curve whereby
more complicated procedures could be performed
in the latter part of their experience. They also
emphasised the importance of achieving proficiency
in laparoscopic hepatectomies via simulation and
wet laboratories. From our experience, we agree
that we could safely expand our indications from
wedge resection of small tumours at anterior and superior liver segments to major resections and
posterosuperior lesions. However, the technical
demand and learning path for wedge resections
are entirely different from those of anatomical
hemihepatectomies or monosegmentectomies. The
training for LLR and learning curve issue is still
an important unresolved topic that needs to be
investigated further.
Conclusion
Laparoscopic hepatectomies are feasible and safe
with favourable patient outcomes. A learning curve
is present and could be overcome with increasing
experience. However, the long-term outcomes
associated with the procedure require further study
with longer follow-up.
References
1. Gagner M, Rheault M, Dubuc J. Laparoscopic partial hepatectomy for liver tumor [abstract]. Surg Endosc 1992;6:99.
2. Nguyen KT, Gamblin TC, Geller DA. World review of laparoscopic liver resection—2,804 patients. Ann Surg 2009;250:831-41. CrossRef
3. Buell JF, Cherqui D, Geller DA, et al. The international position on laparoscopic liver surgery: the Louisville Statement, 2008. Ann Surg 2009;250:825-30. CrossRef
4. Simillis C, Constantinides VA, Tekkis PP, et al. Laparoscopic versus open hepatic resections for benign and malignant neoplasms—a meta-analysis. Surgery 2007;141:203-11. CrossRef
5. Koffron AJ, Auffenberg G, Kung R, Abecassis M. Evaluation of 300 minimally invasive liver resections at a single institution: less is more. Ann Surg 2007;246:385-92. CrossRef
6. Croome KP, Yamashita MH. Laparoscopic vs open hepatic resection for benign and malignant tumors: an updated meta-analysis. Arch Surg 2010;145:1109-18. CrossRef
7. Chang S, Laurent A, Tayar C, Karoui M, Cherqui D. Laparoscopy as a routine approach for left lateral sectionectomy. Br J Surg 2007;94:58-63. CrossRef
8. Lee MR, Kim YH, Roh YH, et al. Lessons learned from 100 initial cases of laparoscopic liver surgery. J Korean Surg Soc 2011;80:334-41. CrossRef
9. Vigano L, Laurent A, Tayar C, Tomatis M, Ponti A, Cherqui D. The learning curve in laparoscopic liver resection: improved feasibility and reproducibility. Ann Surg 2009;250:772-82. CrossRef
10. Robinson SM, Hui KY, Amer A, Manas DM, White SA. Laparoscopic liver resection: is there a learning curve? Dig Surg 2012;29:62-9. CrossRef
11. Kluger MD, Vigano L, Barroso R, Cherqui D. The learning curve in laparoscopic major liver resection. J Hepatobiliary Pancreat Sci 2013;20:131-6. CrossRef
12. Pugh RN, Murray-Lyon IM, Dawson JL, Pietroni MC, Williams R. Transection of the oesophagus for bleeding oesophageal varices. Br J Surg 1973;60:646-9. CrossRef
13. Cheng KC, Yeung YP, Hui J, Ho KM, Yip AW. Laparoscopic resection of hepatocellular carcinoma at segment 7: the posterior approach to anatomic resection. Surg Endosc 2011;25:3437. CrossRef
14. Yeung YP. Laparoscopic anatomic monosegmentectomy of hepatocellular carcinoma of the right hepatic lobe. Surg Laparosc Endosc Percutan Tech 2012;22:e259-62. CrossRef
15. Dindo D, Demartines N, Clavien PA. Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg 2004;240:205-13. CrossRef
16. Buell JF, Thomas MJ, Doty TC, et al. An initial experience and evolution of laparoscopic hepatic resectional surgery. Surgery 2004;136:804-11. CrossRef
17. Morino M, Morra I, Rosso E, Miglietta C, Garrone C. Laparoscopic vs open hepatic resection: a comparative study. Surg Endosc 2003;17:1914-8. CrossRef
18. Cardinal JS, Reddy SK, Tsung A, Marsh JW, Geller DA. Laparoscopic major hepatectomy: pure laparoscopic approach versus hand-assisted technique. J Hepatobiliary Pancreat Sci 2013;20:114-9. CrossRef