Hong Kong Med J 2014;20:158–60 | Number 2, April 2014
DOI: 10.12809/hkmj133881
© Hong Kong Academy of Medicine. CC BY-NC-ND 4.0
CASE REPORT
Pseudohyperkalaemia with acute leukaemia:
association with pneumatic tube transport of blood specimens
Albert SW Ku, FHKAM (Paediatrics), FHKAM
(Anaesthesiology); Robin HS Chen, FHKAM (Paediatrics), FHKCPaed;
Rocky LK Law, MB, BS
Department of Paediatrics and
Adolescent Medicine, Pamela Youde
Nethersole Eastern Hospital, Chai Wan, Hong Kong
Corresponding author: Dr ASW Ku (alku1298@hkstar.com)
Abstract
Falsely elevated serum or plasma potassium level can
be the result of mechanical injury to blood cells. We
describe pseudohyperkalaemia caused by pneumatic
tube transport of blood specimens from a patient
with leukaemia. Clinicians should be aware of this
possibility when interpreting the clinical significance
of hyperkalaemia. In leukaemic patients, pneumatic
tube transport of blood specimens for potassium
analysis should be avoided.
Case report
A 10-year-old boy was referred from another
hospital for malaise and on-and-off fever for 2 weeks
in August 2010. There were no respiratory, urinary,
gastro-intestinal, or neurological symptoms. Physical
examination revealed a febrile child with pallor.
There were multiple bruises over the lower limbs,
multiple cervical and inguinal lymph nodes, and
hepatosplenomegaly. The potassium level checked
in the referring hospital was 4.3 mmol/L.
Laboratory investigations in our hospital
revealed a white blood cell (WBC) count of
340.6 x 109 /L with blast cell count of 282.69 x 109
/L
(83%). The neutrophil count was 45.41 x 109 /L
(13.3%), with 1.14 x 109 /L (0.3%) metamyelocytes.
The haemoglobin level was 71 g/L and platelet
count was 68 x 109 /L. Blood biochemistry revealed
the following serum levels: potassium 6.0 mmol/L,
sodium 137 mmol/L, urea 5.0 mmol/L, creatinine
61 μmol/L, calcium 2.33 mmol/L, phosphate 1.38
mmol/L, urate 0.4 mmol/L, and the serum lactate
dehydrogenase concentration was 3800 IU/L. The
potassium assay was repeated with blood specimen
drawn from a venepuncture into a serum specimen
bottle. The result was 7.4 mmol/L. It was suspected
to be a factitious result related to clotting of the
specimen or haemolysis. Another venous sample
was collected into a heparinised bottle for plasma
potassium level assay and was sent to laboratory by
pneumatic tube for urgent analysis. The result was
8.2 mmol/L.
In view of the rapidly rising potassium
level
in a patient at risk of tumour lysis syndrome,
despite cytotoxic chemotherapy not having been
commenced, the patient was transferred to the
paediatric intensive care unit. Electrocardiography revealed a
sinus tachycardia of 125 beats/min with
no features to suggest hyperkalaemic change. An
arterial specimen drawn from an arterial line into
a heparinised bottle was sent to the laboratory by
special messenger. Treatment of hyperkalaemia
was commenced before availability of the result in
view of the perceived rapidly rising potassium level
from 6.0 mmol/L to 8.2 mmol/L in less than 5 hours.
He was given a dose of calcium gluconate, insulin
with dextrose, and a dose of calcium polystyrene
sulphonate resin. Hyperhydration with intravenous
fluid 2.5 L/m2/day together with allopurinol,
ceftazidime and amikacin were also commenced.
The plasma potassium level in the pre-treatment
arterial specimen was 4.2 mmol/L.
Potassium level was measured again after
dextrose-insulin using an arterial heparinised
sample. The specimen was sent to laboratory by
pneumatic tube for urgent analysis. The result was
9.2 mmol/L, which was suspected to be factitious.
Therefore paired serum and plasma specimens
were sent to the laboratory by special messenger
for careful, urgent processing. The resulting plasma
potassium level was 4.1 mmol/L and the serum
sample showed interference from potassium leakage.
As the condition of pseudohyperkalaemia became
evident, no further therapy for hyperkalaemia
was given. The patient was then transferred to a
paediatric oncology centre, where the diagnosis of
acute T-cell lymphoblastic leukaemia was confirmed.
His electrocardiogram and blood glucose level
were normal all along; the plasma potassium level
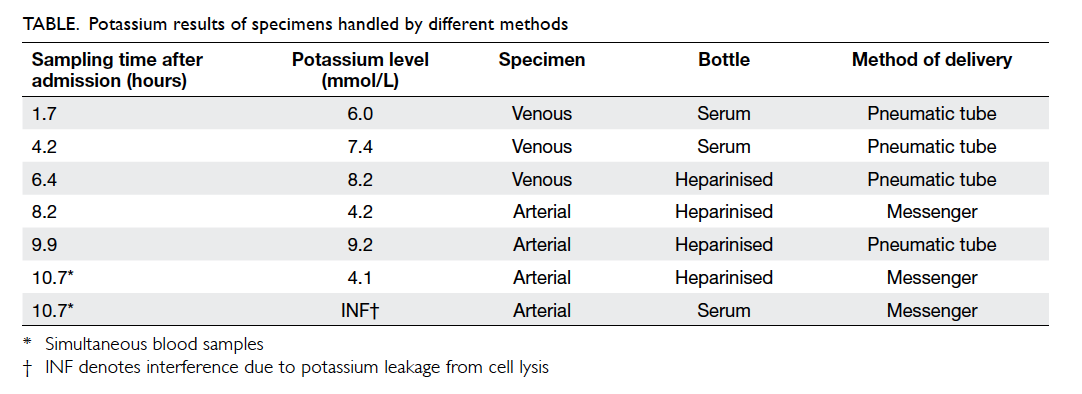
checked before transfer was 4.6 mmol/L. The Table
shows a summary of serum and plasma potassium
level results with reference to the sampling time
after hospital admission and processing techniques. Arterial line
samples drawn into heparinised bottles
with delivery by messenger showed more reliable
and consistent results.
Discussion
Hyperkalaemia is a potentially
life-threatening
condition for which emergency treatment may
be necessary. However, pseudohyperkalaemia is a
common laboratory artefact and, if unrecognised,
may lead to a dilemma.1 2 Workup for falsely
elevated
potassium levels may lead to delay in treatment
and waste of resources. On the other hand,
aggressive treatment including renal dialysis may
be unnecessarily commenced.3
Therefore, clinicians
should be aware of the conditions that may give rise
to falsely elevated potassium levels.
In most cases, pseudohyperkalaemia occurs
during the collection process, transport, or storage
of specimens.4 The artefact
is due to leaching of
potassium from cytosols during clotting or storage
of the sample.1 Leakage of
potassium from blood
cells can occur as an in-vitro phenomenon during
blood coagulation.5 6 7 This
phenomenon is usually
encountered in serum and not plasma. Serum
potassium levels have been reported to be higher
than plasma levels, with a mean difference of 0.36 ± 0.18 mmol/L
in samples with a normal number
of blood cells.8 Therefore
anticoagulated plasma
samples provide more accurate measurement of
the true potassium level.1
Lithium heparin is the
recommended anticoagulant for this purpose.4
Potassium release from red blood cells by
in-vitro haemolysis is a well-recognised cause of
spurious results. Conditions that induce in-vitro
haemolysis include fist clenching during phlebotomy,
drawing blood into an evacuated tube, use of small-gauge
needles, use of tourniquets, cold storage, delay
in sample processing, mechanical trauma during
vigorous mixing, or hard centrifugation.1
3 There
is a rare genetic condition, familial pseudohyperkalaemia,
which is an autosomal dominant disorder associated with excessive
leakage of potassium
across red cell membranes.9
Pseudohyperkalaemia can also be the result
of pre-existing pathological conditions resulting in
cellular potassium leakage. Such conditions include
acute leukaemia, chronic myeloproliferative disorders
(chronic myeloid leukaemia, polycythaemia vera,
essential thrombocythaemia), chronic lymphocytic
leukaemia, and reactive thrombocytosis.1
10 The large
number of blood cells may exaggerate the effects of
potassium leakage from coagulation and further
increase the discrepancy between serum and plasma
potassium levels. Unphysiological conditions and
shortage of metabolic fuels leading to impaired
sodium/potassium adenosine triphosphatase
activity may contribute to release of potassium
from large numbers of white cells.7
The abnormal
fragility of malignant leukocytes also makes them
susceptible to mechanical stress. Colussi11
reported
that the minor mechanical stress of drawing blood
into vacuum tubes or syringe shaking induced lysis
of leukaemic lymphocytes that appeared in blood
smears as lymphocytic ghosts called “basket cells”.
Kellerman and Thornbery3
reported the occurrence
of pseudohyperkalaemia due to pneumatic tube
transport in a leukaemic patient with a WBC count
of 290 x 109 /L, but they did not find any significant
differences in potassium values between walked
and tube-transported specimens in control patients with normal WBC
counts. The effect on potassium
resulting from pneumatic tube transport is likely due
to both WBC number and fragility.3
Chawla et al12
also reported a case of pseudohyperkalaemia due
to mechanical disruption of leukocytes in a patient
with chronic lymphocytic leukaemia and proposed
to designate this phenomenon as pneumatic tube
“pseudo tumour lysis syndrome”. Ruddy et al13 also
reported a chronic lymphocytic leukaemia patient
with venous potassium levels spuriously higher than
arterial potassium levels. The authors hypothesised
that this was likely due to a greater opportunity for
lysis of white blood cells in the venous blood related
to differences in mechanical stressors between
venous and arterial blood draw techniques.13 In
our patient, the possible contributing factors for
pseudohyperkalaemia include high WBC count,
fragile leukaemic blast cells, clotting of serum
specimens, and mechanical trauma secondary to
pneumatic tube transport.
Pseudohyperkalaemia is characterised by an
elevation of serum potassium levels in the absence
of clinical evidence of electrolyte imbalance,10 and
should be suspected when there are no other clinical
features of hyperkalaemia, such as peaked T waves
and QRS widening on the electrocardiogram.3 The
potassium level should therefore be interpreted
together with the clinical context and other
investigation results. Hyperkalaemia is exceptionally
unlikely if renal indices are normal and there are no
predisposing factors, such as intake of potassium
supplements and/or drugs that raise potassium
levels.4 In our patient,
the possibility of a spurious
result was suspected at an early stage. Treatment
was commenced because potassium levels showed
a rising trend in a patient at risk of tumour lysis
syndrome. Ultimately, the pseudohyperkalaemia
was confirmed by elimination of the possible causes
of measurement error. In this patient, there were
hints to remind clinicians to consider the possibility
of factitious results as the electrocardiogram was
normal all along and the other expected biochemical
changes of tumour lysis syndrome were absent.
Besides pseudohyperkalaemia, factitious
hypokalaemia
may also be encountered in patients with
leukaemia with WBC counts higher than 100 x 109 /L
when blood samples are allowed to stand at room
temperature.1 This
phenomenon is related to
transcellular potassium shift into leukaemic cells.1
Even in normal specimens, pseudohypokalaemia
may also be noted if sample analysis is delayed, and
is believed to be mediated by sodium-potassium-exchanging ATPase,14 while specimen
deterioration
due to long storage can lead to pseudohyperkalaemia.4
In conclusion, when considering
investigation
and treatment, clinicians should be aware of
the potential causes of pseudohyperkalaemia in
leukaemic patients. Extreme care in handling blood
samples is very important. The use of pneumatic
tube transport for potassium analysis should be
avoided in leukaemic patients.
References
1. Dalal BI, Brigden ML. Factitious
biochemical measurements resulting from haematologic conditions.
Am J Clin Pathol 2009;131:195-204. CrossRef
2. Brigden ML, Dalal BI. Spurious
and artifactual test results, II: morphologic abnormalities,
pseudosyndromes and spurious test results. Lab Med
1999;30:397-405.
3. Kellerman PS, Thornbery JM.
Pseudohyperkalaemia due to pneumatic tube transport in a leukaemic
patient. Am J Kidney Dis 2005;46:746-8. CrossRef
4. Smellie WS. Spurious
hyperkalaemia. BMJ 2007;334:693-5. CrossRef
5. Hartmann RC, Auditore JV,
Jackson DP. Studies on thrombocytosis. Hyperkalaemia due to
release of potassium from platelet during coagulation. J Clin
Invest 1958;37:699-707. CrossRef
6. Bronson WR, DeVita VT, Carbone
PP, Cotlove E. Pseudohyperkalaemia due to release of potassium
from white blood cells during clotting. N Engl J Med
1966;274:369-75. CrossRef
7. Colussi G, Cipriani D.
Pseudohyperkalaemia in extreme leukocytosis. Am J Nephrol
1995;15:450-2. CrossRef
8. Nijsten MW, de Smet BJ,
Dofferhoff AS. Pseudohyperkalemia and platelet counts. N Engl J
Med 1991;325:1107. CrossRef
9. Iolascon A, Stewart GW,
Ajetunmobi JF, et al. Familial pseudohyperkalaemia maps to the
same locus as dehydration hereditary stomatocytosis (hereditary
xerocytosis). Blood 1999;93:3120-3.
10. Sevastos N, Theodossiades G,
Efstathiou S, Papatheodoridis GV, Manesis E, Archimandritis AJ.
Pseudohyperkalaemia in serum: the phenomenon and its clinical
magnitude. J Lab Clin Med 2006;147:139-44. CrossRef
11. Colussi G. Pseudohyperkalaemia
in leukaemias. Am J Kidney Dis 2006;47:373. CrossRef
12. Chawla NR, Shapiro J, Sham RL.
Pneumatic tube "pseudo tumour lysis syndrome" in chronic
lymphocytic leukaemia. Am J Hematol 2009;84:613-4. CrossRef
13. Ruddy KJ, Wu D, Brown JR.
Pseudohyperkalaemia in chronic lymphocytic leukaemia. J Clin Oncol
2008;26:2781-2. CrossRef
14. Sodi R, Davison AS, Holmes E,
Hine TJ, Roberts NB. The phenomenon of seasonal pseudohypokalemia:
effects of ambient temperature, plasma glucose and role for
sodium-potassium-exchanging-ATPase. Clin Biochem 2009;42:813-8. CrossRef